Abstract
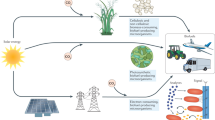
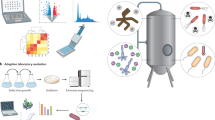
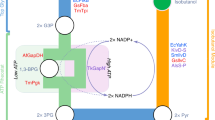
Advanced biofuels produced by microorganisms have similar properties to petroleum-based fuels, and can 'drop in' to the existing transportation infrastructure. However, producing these biofuels in yields high enough to be useful requires the engineering of the microorganism's metabolism. Such engineering is not based on just one specific feedstock or host organism. Data-driven and synthetic-biology approaches can be used to optimize both the host and pathways to maximize fuel production. Despite some success, challenges still need to be met to move advanced biofuels towards commercialization, and to compete with more conventional fuels.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Stephanopoulos, G. Challenges in engineering microbes for biofuels production. Science 315, 801–804 (2007).
Kerr, R. Global warming is changing the world. Science 316, 188–190 (2007).
Fortman, J. L. et al. Biofuel alternatives to ethanol: pumping the microbial well. Trends Biotechnol. 26, 375–381 (2008).
Atsumi, S., Hanai, T. & Liao, J. C. Non-fermentative pathways for synthesis of branched-chain higher alcohols as biofuels. Nature 451, 86–89 (2008). This paper describes the rerouting of the E. coli amino-acid biosynthetic pathway for the overproduction of a variety of long-chain or higher alcohols.
Keasling, J. D. Manufacturing molecules through metabolic engineering. Science 330, 1355–1358 (2010).
Nielsen, J. & Keasling, J. D. Synergies between synthetic biology and metabolic engineering. Nature Biotechnol. 29, 693–695 (2011).
Lee, S. K. et al. Metabolic engineering of microorganisms for biofuels production: from bugs to synthetic biology to fuels. Curr. Opin. Biotechnol. 19, 556–563 (2008).
Peralta-Yahya, P. P. & Keasling, J. D. Advanced biofuel production in microbes. Biotechnol. J. 5, 147–162 (2010).
Lee, S. Y. et al. Fermentative butanol production by Clostridia. Biotechnol. Bioeng. 101, 209–228 (2008).
Qureshi, N. & Blaschek, H. P. Production of acetone butanol ethanol (ABE) by a hyper-producing mutant strain of Clostridium beijerinckii BA101 and recovery by pervaporation. Biotechnol. Prog. 15, 594–602 (1999).
Ezeji, T. C., Qureshi, N. & Blaschek, H. P. Production of acetone butanol (AB) from liquefied corn starch, a commercial substrate, using Clostridium beijerinckii coupled with product recovery by gas stripping. J. Ind. Microbiol. Biotechnol. 34, 771–777 (2007).
Malaviya, A., Jang, Y. S. & Lee, S. Y. Continuous butanol production with reduced byproducts formation from glycerol by a hyper producing mutant of Clostridium pasteurianum. Appl. Microbiol. Biotechnol. 93, 1485–1494 (2011).
Kopke, M. et al. Clostridium ljungdahlii represents a microbial production platform based on syngas. Proc. Natl Acad. Sci. USA 107, 13087–13092 (2010).
Ezeji, T. et al. Achievements and perspectives to overcome the poor solvent resistance in acetone and butanol-producing microorganisms. Appl. Microbiol. Biotechnol. 85, 1697–1712 (2009).
Lutke-Eversloh, T. & Bahl, H. Metabolic engineering of Clostridium acetobutylicum: recent advances to improve butanol production. Curr. Opin. Biotechnol. 22, 634–647 (2011).
Atsumi, S. et al. Metabolic engineering of Escherichia coli for 1-butanol production. Metab. Eng. 10, 305–311 (2008).
Steen, E. J. et al. Metabolic engineering of Saccharomyces cerevisiae for the production of n-butanol. Microb. Cell Fact. 7, 36 (2008).
Nielsen, D. R. et al. Engineering alternative butanol production platforms in heterologous bacteria. Metab. Eng. 11, 262–273 (2009).
Berezina, O. V. et al. Reconstructing the clostridial n-butanol metabolic pathway in Lactobacillus brevis. Appl. Microbiol. Biotechnol. 87, 635–646 (2010).
Lan, E. I. & Liao, J. C. ATP drives direct photosynthetic production of 1-butanol in cyanobacteria. Proc. Natl Acad. Sci. USA 109, 6018–6023 (2012).
Bond-Watts, B. B., Bellerose, R. J. & Chang, M. C. Enzyme mechanism as a kinetic control element for designing synthetic biofuel pathways. Nature Chem. Biol. 7, 222–227 (2011). In this study, the authors assemble a synthetic butanol pathway in E. coli by choosing enzymes from different organisms on the basis of their biochemical properties.
Shen, C. R. et al. Driving forces enable high-titer anaerobic 1-butanol synthesis in Escherichia coli. Appl. Environ. Microbiol. 77, 2905–2915 (2011).
Yan, Y. & Liao, J. C. Engineering metabolic systems for production of advanced fuels. J. Ind. Microbiol. Biotechnol. 36, 471–479 (2009).
Smith, K. M., Cho, K. M. & Liao, J. C. Engineering Corynebacterium glutamicum for isobutanol production. Appl. Microbiol. Biotechnol. 87, 1045–1055 (2010).
Chen, X. et al. Increased isobutanol production in Saccharomyces cerevisiae by overexpression of genes in valine metabolism. Biotechnol. Biofuels 4, 21 (2011).
Higashide, W. et al. Metabolic engineering of Clostridium cellulolyticum for production of isobutanol from cellulose. Appl. Environ. Microbiol. 77, 2727–2733 (2011).
Atsumi, S., Higashide, W. & Liao, J. C. Direct photosynthetic recycling of carbon dioxide to isobutyraldehyde. Nature Biotechnol. 27, 1177–1180 (2009).
Huo, Y. X. et al. Conversion of proteins into biofuels by engineering nitrogen flux. Nature Biotechnol. 29, 346–351 (2011).
Li, H. et al. Integrated electromicrobial conversion of CO2 to higher alcohols. Science 335, 1596 (2012). This paper presents the production of isobutanol in Ralstonia from CO 2 by using electricity.
Kirby, J. & Keasling, J. D. Biosynthesis of plant isoprenoids: perspectives for microbial engineering. Annu. Rev. Plant Biol. 60, 335–355 (2009).
Harvey, B. G., Wright, M. E. & Quintana, R. L. High-density renewable fuels based on the selective dimerization of pinenes. Energy Fuels 24, 267–273 (2010).
Kuzuyama, T. Mevalonate and nonmevalonate pathways for the biosynthesis of isoprene units. Biosci. Biotechnol. Biochem. 66, 1619–1627 (2002).
Banerjee, A. et al. Botryococcus braunii: a renewable source of hydrocarbons and other chemicals. Crit. Rev. Biotechnol. 22, 245–279 (2002).
Tsukahara, K. et al. Treatment of liquid fraction separated from liquidized food waste in an upflow anaerobic sludge blanket reactor. J. Biosci. Bioeng. 87, 554–556 (1999).
Martin, V. J. et al. Engineering a mevalonate pathway in Escherichia coli for production of terpenoids. Nature Biotechnol. 21, 796–802 (2003).
Ro, D. K. et al. Production of the antimalarial drug precursor artemisinic acid in engineered yeast. Nature 440, 940–943 (2006).
Tsuruta, H. et al. High-level production of amorpha-4,11-diene, a precursor of the antimalarial agent artemisinin, in Escherichia coli. PLoS ONE 4, e4489 (2009).
Westfall, P. J. et al. Production of amorphadiene in yeast, and its conversion to dihydroartemisinic acid, precursor to the antimalarial agent artemisinin. Proc. Natl Acad. Sci. USA 109, E111–E118 (2012).
Leonard, E. et al. Combining metabolic and protein engineering of a terpenoid biosynthetic pathway for overproduction and selectivity control. Proc. Natl Acad. Sci. USA 107, 13654–13659 (2010).
Ajikumar, P. K. et al. Isoprenoid pathway optimization for Taxol precursor overproduction in Escherichia coli. Science 330, 70–74 (2010).
Renninger, N. & McPhee, D. Fuel compositions comprising farnesane and farnesane derivatives and method of making and using same. US patent 7,399,323 (2008).
Rude, M. A. & Schirmer, A. New microbial fuels: a biotech perspective. Curr. Opin. Microbiol. 12, 274–281 (2009).
Ubersax, J. & Platt, D. Genetically modified microbes producing isoprenoids patent world 1,414,52 A1 (2010).
Peralta-Yahya, P. P. et al. Identification and microbial production of a terpene-based advanced biofuel. Nature Commun. 2, 483 (2011). In this paper, the authors identify bisabolane as a biosynthetic diesel and overproduce its immediate precursor, the isoprenoid bisabolene, in E. coli and S. cerevisiae.
McAndrew, R. P. et al. Structure of a three-domain sesquiterpene synthase: a prospective target for advanced biofuels production. Structure 19, 1876–1884 (2011).
Withers, S. T. et al. Identification of isopentenol biosynthetic genes from Bacillus subtilis by a screening method based on isoprenoid precursor toxicity. Appl. Environ. Microbiol. 73, 6277–6283 (2007).
Bokinsky, G. et al. Synthesis of three advanced biofuels from ionic liquid-pretreated switchgrass using engineered Escherichia coli. Proc. Natl Acad. Sci. USA 108, 19949–19954 (2011). This paper presents the production of fatty-acid ethyl esters, butanol and the isoprenoid pinene in E. coli using ionic-liquid-treated switchgrass as the sole carbon source.
Steen, E. J. et al. Microbial production of fatty-acid-derived fuels and chemicals from plant biomass. Nature 463, 559–562 (2010). This study describes several pathways leading to the bioproduction of fatty acids, fatty alcohols and fatty-acid ethyl esters in E. coli in high titres.
Lennen, R. M. et al. A process for microbial hydrocarbon synthesis: overproduction of fatty acids in Escherichia coli and catalytic conversion to alkanes. Biotechnol. Bioeng. 106, 193–202 (2010).
Liu, T., Vora, H. & Khosla, C. Quantitative analysis and engineering of fatty-acid biosynthesis in E. coli. Metab. Eng. 12, 378–386 (2010).
Handke, P., Lynch, S. A. & Gill, R. T. Application and engineering of fatty acid biosynthesis in Escherichia coli for advanced fuels and chemicals. Metab. Eng. 13, 28–37 (2011).
Reiser, S. & Somerville, C. Isolation of mutants of Acinetobacter calcoaceticus deficient in wax ester synthesis and complementation of one mutation with a gene encoding a fatty acyl coenzyme A reductase. J. Bacteriol. 179, 2969–2975 (1997).
Tan, X. et al. Photosynthesis driven conversion of carbon dioxide to fatty alcohols and hydrocarbons in cyanobacteria. Metab. Eng. 13, 169–176 (2011).
Goh, E. B. et al. Engineering of bacterial methyl ketone synthesis for biofuels. Appl. Environ. Microbiol. 78, 70–80 (2011).
Zhang, F., Carothers, J. M. & Keasling, J. D. Design of a dynamic sensor-regulator system for production of chemicals and fuels derived from fatty acids. Nature Biotechnol. 30, 354–359 (2012). In this study, the authors developed a dynamic regulatory system that allows the host cell to sense the concentration of a key intermediate and automatically regulate a heterologous pathway.
Kalscheuer, R., Stolting, T. & Steinbuchel, A. Microdiesel: Escherichia coli engineered for fuel production. Microbiology 152, 2529–2536 (2006).
Nawabi, P. et al. Engineering Escherichia coli for biodiesel production utilizing a bacterial fatty acid methyltransferase. Appl. Environ. Microbiol. 77, 8052–8061 (2011).
Schirmer, A. et al. Microbial biosynthesis of alkanes. Science 329, 559–562 (2010). This paper presents the discovery of two cyanobacterial genes responsible for the biosynthesis of alkanes.
Rude, M. A. et al. Terminal olefin (1-alkene) biosynthesis by a novel p450 fatty acid decarboxylase from Jeotgalicoccus species. Appl. Environ. Microbiol. 77, 1718–1727 (2011).
Mendez-Perez, D., Begemann, M. B. & Pfleger, B. F. Modular synthase-encoding gene involved in alpha-olefin biosynthesis in Synechococcus sp. strain PCC 7002. Appl. Environ. Microbiol. 77, 4264–4267 (2011).
Beller, H. R., Goh, E. B. & Keasling, J. D. Genes involved in long-chain alkene biosynthesis in Micrococcus luteus. Appl. Environ. Microbiol. 76, 1212–1223 (2009).
Sukovich, D. J. et al. Widespread head-to-head hydrocarbon biosynthesis in bacteria and role of OleA. Appl. Environ. Microbiol. 76, 3850–3862 (2010).
Meades, G. Jr et al. A tale of two functions: enzymatic activity and translational repression by carboxyltransferase. Nucleic Acids Res. 38, 1217–1227 (2009).
Davis, M. S. & Cronan, J. E. Jr. Inhibition of Escherichia coli acetyl coenzyme A carboxylase by acyl-acyl carrier protein. J. Bacteriol. 183, 1499–1503 (2001).
Seregina, T. A. et al. Construction of a butyrate-producing E.coli strain without the use of heterologous genes. Appl. Biochem. Microbiol. 46, 745–754 (2010).
Sun, J., Pharkya, P. & Burgard, A. Primary alcohol producing organisms. US patent 0,040,426 A1 (2012).
Dellomonaco, C. et al. Engineered reversal of the beta-oxidation cycle for the synthesis of fuels and chemicals. Nature 476, 355–359 (2011). This paper describes a unique pathway leading to the biosynthesis of alcohols and fatty-acid derivatives using reverse beta-oxidation cycles.
Yuzawa, S. et al. Heterologous production of polyketides by modular type I polyketide synthases in Escherichia coli. Curr. Opin. Biotechnol. Preprint at http://dx.doi.org/10.1016/j.copbio.2011.12.029 (2012).
Pfeifer, B. A. & Khosla, C. Biosynthesis of polyketides in heterologous hosts. Microbiol. Mol. Biol. Rev. 65, 106–118 (2001).
Menzella, H. G. et al. Combinatorial polyketide biosynthesis by de novo design and rearrangement of modular polyketide synthase genes. Nature Biotechnol. 23, 1171–1176 (2005).
Jacobsen, J. R. et al. Precursor-directed biosynthesis of erythromycin analogs by an engineered polyketide synthase. Science 277, 367–369 (1997).
Yuzawa, S., Kim, L., Katz, L., et al. Heterologous production ofpolyketides by modular type I polyketide synthases in Escherichia coli. Curr Opin Biotechnol. http://dx.doi.org/10.1016/j.copbio.2011.12.029
Redding-Johanson, A. M. et al. Targeted proteomics for metabolic pathway optimization: application to terpene production. Metab. Eng. 13, 194–203 (2011).
Asadollahi, M. A. et al. Enhancing sesquiterpene production in Saccharomyces cerevisiae through in silico driven metabolic engineering. Metab. Eng. 11, 328–334 (2009).
Dunlop, M. J. et al. Engineering microbial biofuel tolerance and export using efflux pumps. Mol. Syst. Biol. 7, 487 (2011).
Tyo, K. E., Ajikumar, P. K. & Stephanopoulos, G. Stabilized gene duplication enables long-term selection-free heterologous pathway expression. Nature Biotechnol. 27, 760–765 (2009).
Jones, K. L., Kim, S. W. & Keasling, J. D. Low-copy plasmids can perform as well as or better than high-copy plasmids for metabolic engineering of bacteria. Metab. Eng. 2, 328–338 (2000).
De Mey, M. et al. Construction and model-based analysis of a promoter library for E. coli: an indispensable tool for metabolic engineering. BMC Biotechnol. 7, 34 (2007).
Lee, T. S. et al. BglBrick vectors and datasheets: a synthetic biology platform for gene expression. J. Biol. Eng. 5, 12 (2011).
d'Aubenton Carafa, Y., Brody, E. & Thermes, C. Prediction of rho-independent Escherichia coli transcription terminators. A statistical analysis of their RNA stem-loop structures. J. Mol. Biol. 216, 835–858 (1990).
Pfleger, B. F. et al. Combinatorial engineering of intergenic regions in operons tunes expression of multiple genes. Nature Biotechnol. 24, 1027–1032 (2006).
Smolke, C. D., Carrier, T. A. & Keasling, J. D. Coordinated, differential expression of two genes through directed mRNA cleavage and stabilization by secondary structures. Appl. Environ. Microbiol. 66, 5399–5405 (2000).
Salis, H. M., Mirsky, E. A. & Voigt, C. A. Automated design of synthetic ribosome binding sites to control protein expression. Nature Biotechnol. 27, 946–950 (2009).
Griffith, K. L. & Grossman, A. D. Inducible protein degradation in Bacillus subtilis using heterologous peptide tags and adaptor proteins to target substrates to the protease ClpXP. Mol. Microbiol. 70, 1012–1025 (2008).
Holtz, W. J. & Keasling, J. D. Engineering static and dynamic control of synthetic pathways. Cell 140, 19–23 (2010).
Michener, J. K. et al. Applications of genetically-encoded biosensors for the construction and control of biosynthetic pathways. Metab. Eng. 14, 212–222 (2011).
Primak, Y. A. et al. Characterization of a feedback-resistant mevalonate kinase from the archaeon Methanosarcina mazei. Appl. Environ. Microbiol. 77, 7772–7778 (2011).
Dueber, J. E. et al. Synthetic protein scaffolds provide modular control over metabolic flux. Nature Biotechnol. 27, 753–759 (2009). In this paper, a synthetic protein scaffold was used to spatially assemble metabolic-pathway enzymes to increase the flux through a heterologous metabolic pathway.
Buelter, T. et al. Engineered microorganisms capable of producing target compounds under anaerobic conditions. US patent 0,058,532 A1 (2012).
Donaldson, G. K. et al. Fermentative production of four carbon alcohols. US Patent 0,313,206 A1 (2007).
Hong, K. K. & Nielsen, J. Metabolic engineering of Saccharomyces cerevisiae: a key cell factory platform for future biorefineries. Cell Mol. Life Sci. http://dx.doi.org/10.1007/s00018-012-0945-1 (2012).
de Jong, B., Siewers, V. & Nielsen, J. Systems biology of yeast: enabling technology for development of cell factories for production of advanced biofuels. Curr. Opin. Biotechnol. http://dx.doi.org/10.1016/j.copbio.2011.11.021 (2011).
Donaldson, G. K. et al. Fermentative production of four carbon alcohols. US patent 0,313,206 A1 (2011).
Festel, G. et al. Fermentative production of isobutanol in yeast. US patent 0,053,235 A1 (2011).
Knothe, G. Biodiesel and renewable diesel: a comparison. Prog. Energy Combust. Sci. 36, 364–373 (2010).
Trimbur, D. et al. Production of oil in microorganisms. US patent 0,047,863 A1 (2011).
Yu, X. et al. In vitro reconstitution and steady-state analysis of the fatty acid synthase from Escherichia coli. Proc. Natl Acad. Sci. USA 108, 18643–18648 (2011).
Katz, L., Fortman, J. L. & Keasling, J. D. Producing biofuels using polyketide synthases. US patent CN102014623 (2011).
Betancor, L. et al. Improved catalytic activity of a purified multienzyme from a modular polyketide synthase after coexpression with Streptomyces chaperonins in Escherichia coli. ChemBioChem 9, 2962–2966 (2008).
Tracewell, C. A. & Arnold, F. H. Directed enzyme evolution: climbing fitness peaks one amino acid at a time. Curr. Opin. Chem. Biol. 13, 3–9 (2009).
Acknowledgements
P. P. P.-Y. and F. Z. contributed equally to this work. This work was supported in part by the Joint BioEnergy Institute, which is funded by the U.S. Department of Energy, Office of Science, Office of Biological and Environmental Research, through contract DE-AC02-05CH11231, and by the Synthetic Biology Engineering Research Center, which is funded by National Science Foundation Award No. 0540879.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
J.D.K. has a financial interest in Amyris, LS9 and Lygos. S.B.d. has a financial interest in LS9. The remaining authors declare no competing financial interest.
Additional information
Reprints and permissions information is available at www.nature.com/reprints
Rights and permissions
About this article
Cite this article
Peralta-Yahya, P., Zhang, F., del Cardayre, S. et al. Microbial engineering for the production of advanced biofuels. Nature 488, 320–328 (2012). https://doi.org/10.1038/nature11478
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11478
This article is cited by
-
Lipid membrane remodeling and metabolic response during isobutanol and ethanol exposure in Zymomonas mobilis
Biotechnology for Biofuels and Bioproducts (2024)
-
The logic of carbon substitution: from fossilised life to “cell factories”
Review of Agricultural, Food and Environmental Studies (2024)
-
Tighter Bounds on Transient Moments of Stochastic Chemical Systems
Journal of Optimization Theory and Applications (2024)
-
Mastering targeted genome engineering of GC-rich oleaginous yeast for tailored plant oil alternatives for the food and chemical sector
Microbial Cell Factories (2023)
-
Mapping competitive pathways to terpenoid biosynthesis in Synechocystis sp. PCC 6803 using an antisense RNA synthetic tool
Microbial Cell Factories (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.