Abstract
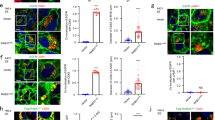
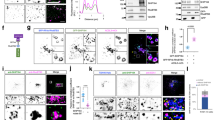
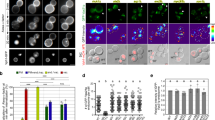
An outstanding question is how cells control the number and size of membrane organelles. The small GTPase Rab5 has been proposed to be a master regulator of endosome biogenesis. Here, to test this hypothesis, we developed a mathematical model of endosome dependency on Rab5 and validated it by titrating down all three Rab5 isoforms in adult mouse liver using state-of-the-art RNA interference technology. Unexpectedly, the endocytic system was resilient to depletion of Rab5 and collapsed only when Rab5 decreased to a critical level. Loss of Rab5 below this threshold caused a marked reduction in the number of early endosomes, late endosomes and lysosomes, associated with a block of low-density lipoprotein endocytosis. Loss of endosomes caused failure to deliver apical proteins to the bile canaliculi, suggesting a requirement for polarized cargo sorting. Our results demonstrate for the first time, to our knowledge, the role of Rab5 as an endosome organizer in vivo and reveal the resilience mechanisms of the endocytic system.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Mellman, I. Endocytosis and molecular sorting. Annu. Rev. Cell Dev. Biol. 12, 575–625 (1996)
Bonifacino, J. S. & Glick, B. S. The mechanisms of vesicle budding and fusion. Cell 116, 153–166 (2004)
Robinson, M. S. Adaptable adaptors for coated vesicles. Trends Cell Biol. 14, 167–174 (2004)
Jahn, R. & Scheller, R. H. SNAREs–engines for membrane fusion. Nature Rev. Mol. Cell Biol. 7, 631–643 (2006)
Pfeffer, S. R. Unsolved mysteries in membrane traffic. Annu. Rev. Biochem. 76, 629–645 (2007)
Gruenberg, J. & Maxfield, F. R. Membrane transport in the endocytic pathway. Curr. Opin. Cell Biol. 7, 552–563 (1995)
Rink, J., Ghigo, E., Kalaidzidis, Y. & Zerial, M. Rab conversion as a mechanism of progression from early to late endosomes. Cell 122, 735–749 (2005)
Poteryaev, D., Datta, S., Ackema, K., Zerial, M. & Spang, A. Identification of the switch in early-to-late endosome transition. Cell 141, 497–508 (2010)
Vonderheit, A. & Helenius, A. Rab7 associates with early endosomes to mediate sorting and transport of Semliki forest virus to late endosomes. PLoS Biol. 3, e233 (2005)
Gorvel, J. P., Chavrier, P., Zerial, M. & Gruenberg, J. rab5 controls early endosome fusion in vitro . Cell 64, 915–925 (1991)
Rybin, V. et al. GTPase activity of Rab5 acts as a timer for endocytic membrane fusion. Nature 383, 266–269 (1996)
Skjeldal, F. M. et al. The fusion of early endosomes induces molecular motor-driven tubule formation and fission. J . Cell Sciencehttp://dx.doi.org/10.1242/jcs.092569 (22 February 2012)
Futter, C. E., Connolly, C. N., Cutler, D. F. & Hopkins, C. R. Newly synthesized transferrin receptors can be detected in the endosome before they appear on the cell surface. J. Biol. Chem. 270, 10999–11003 (1995)
Harsay, E. & Schekman, R. A subset of yeast vacuolar protein sorting mutants is blocked in one branch of the exocytic pathway. J. Cell Biol. 156, 271–286 (2002)
Ang, A. L. et al. Recycling endosomes can serve as intermediates during transport from the Golgi to the plasma membrane of MDCK cells. J. Cell Biol. 167, 531–543 (2004)
Weisz, O. A. & Rodriguez-Boulan, E. Apical trafficking in epithelial cells: signals, clusters and motors. J. Cell Sci. 122, 4253–4266 (2009)
Golachowska, M. R., Hoekstra, D. & van IJzendoorn, S. C. D. Recycling endosomes in apical plasma membrane domain formation and epithelial cell polarity. Trends Cell Biol. 20, 618–626 (2010)
Ihrke, G. et al. Apical plasma membrane proteins and endolyn-78 travel through a subapical compartment in polarized WIF-B hepatocytes. J. Cell Biol. 141, 115–133 (1998)
Cresawn, K. O. et al. Differential involvement of endocytic compartments in the biosynthetic traffic of apical proteins. EMBO J. 26, 3737–3748 (2007)
Nokes, R. L., Fields, I. C., Collins, R. N. & Folsch, H. Rab13 regulates membrane trafficking between TGN and recycling endosomes in polarized epithelial cells. J. Cell Biol. 182, 845–853 (2008)
Farr, G. A., Hull, M., Mellman, I. & Caplan, M. J. Membrane proteins follow multiple pathways to the basolateral cell surface in polarized epithelial cells. J. Cell Biol. 186, 269–282 (2009)
Zerial, M. & McBride, H. Rab proteins as membrane organizers. Nature Rev. Mol. Cell Biol. 2, 107–117 (2001)
Ohya, T. et al. Reconstitution of Rab- and SNARE-dependent membrane fusion by synthetic endosomes. Nature 459, 1091–1097 (2009)
Bucci, C. et al. Co-operative regulation of endocytosis by three Rab5 isoforms. FEBS Lett. 366, 65–71 (1995)
Wucherpfennig, T., Wilsch-Brauninger, M. & Gonzalez-Gaitan, M. Role of Drosophila Rab5 during endosomal trafficking at the synapse and evoked neurotransmitter release. J. Cell Biol. 161, 609–624 (2003)
Morrison, H. A. et al. Regulation of early endosomal entry by the Drosophila tumor suppressors Rabenosyn and Vps45. Mol. Biol. Cell 19, 4167–4176 (2008)
Bucci, C. et al. The small GTPase rab5 functions as a regulatory factor in the early endocytic pathway. Cell 70, 715–728 (1992)
Akinc, A. et al. Targeted delivery of RNAi therapeutics with endogenous and exogenous ligand-based mechanisms. Mol. Ther. 18, 1357–1364 (2010)
Del Conte-Zerial, P. et al. Membrane identity and GTPase cascades regulated by toggle and cut-out switches. Mol. Syst. Biol. 4, 206 (2008)
Heinrich, R. & Rapoport, T. A. Generation of nonidentical compartments in vesicular transport systems. J. Cell Biol. 168, 271–280 (2005)
Serio, G. et al. Small GTPase Rab5 participates in chromosome congression and regulates localization of the centromere-associated protein CENP-F to kinetochores. Proc. Natl Acad. Sci. USA 108, 17337–17342 (2011)
Akinc, A. et al. A combinatorial library of lipid-like materials for delivery of RNAi therapeutics. Nature Biotechnol. 26, 561–569 (2008)
Love, K. T. et al. Lipid-like materials for low-dose, in vivo gene silencing. Proc. Natl Acad. Sci. USA 107, 1864–1869 (2010)
Christoforidis, S., McBride, H. M., Burgoyne, R. D. & Zerial, M. The Rab5 effector EEA1 is a core component of endosome docking. Nature 397, 621–625 (1999)
Collinet, C. et al. Systems survey of endocytosis by multiparametric image analysis. Nature 464, 243–249 (2010)
Chen, P. I., Kong, C., Su, X. & Stahl, P. D. Rab5 isoforms differentially regulate the trafficking and degradation of epidermal growth factor receptors. J. Biol. Chem. 284, 30328–30338 (2009)
Mukherjee, S., Ghosh, R. N. & Maxfield, F. R. Endocytosis. Physiol. Rev. 77, 759–803 (1997)
Schroeder, B. M. M. in The Liver: Biology and Pathobiology 5th edn (eds Arias, I. et al.) Ch. 7 107–123 (Wiley-Blackwell, 2009)
Wolfsdorf, J. I. & Weinstein, D. A. Glycogen storage diseases. Rev. Endocr. Metab. Disord. 4, 95–102 (2003)
van der Bliek, A. M. et al. Mutations in human dynamin block an intermediate stage in coated vesicle formation. J. Cell Biol. 122, 553–563 (1993)
Shen, H. et al. Constitutive activated Cdc42-associated kinase (Ack) phosphorylation at arrested endocytic clathrin-coated pits of cells that lack dynamin. Mol. Biol. Cell 22, 493–502 (2011)
Kipp, H., Pichetshote, N. & Arias, I. M. Transporters on demand: intrahepatic pools of canalicular ATP binding cassette transporters in rat liver. J. Biol. Chem. 276, 7218–7224 (2001)
Wang, L. & Boyer, J. L. The maintenance and generation of membrane polarity in hepatocytes. Hepatology 39, 892–899 (2004)
Wakabayashi, Y., Dutt, P., Lippincott-Schwartz, J. & Arias, I. M. Rab11a and myosin Vb are required for bile canalicular formation in WIF-B9 cells. Proc. Natl Acad. Sci. USA 102, 15087–15092 (2005)
Lawe, D. C. et al. Sequential roles for phosphatidylinositol 3-phosphate and Rab5 in tethering and fusion of early endosomes via their interaction with EEA1. J. Biol. Chem. 277, 8611–8617 (2002)
Renaud, G., Hamilton, R. L. & Havel, R. J. Hepatic metabolism of colloidal gold-low-density lipoprotein complexes in the rat: evidence for bulk excretion of lysosomal contents into bile. Hepatology 9, 380–392 (1989)
Damke, H., Baba, T., van der Bliek, A. M. & Schmid, S. L. Clathrin-independent pinocytosis is induced in cells overexpressing a temperature-sensitive mutant of dynamin. J. Cell Biol. 131, 69–80 (1995)
Koenig, J. H. & Ikeda, K. Transformational process of the endosomal compartment in nephrocytes of Drosophila melanogaster . Cell Tissue Res. 262, 233–244 (1990)
Wakabayashi, Y., Lippincott-Schwartz, J. & Arias, I. M. Intracellular trafficking of bile salt export pump (ABCB11) in polarized hepatic cells: constitutive cycling between the canalicular membrane and rab11-positive endosomes. Mol. Biol. Cell 15, 3485–3496 (2004)
Acknowledgements
We acknowledge K. Simons, E. Knust, C. Fetzer, T. Galvez, G. O’Sullivan and M. P. McShane for discussions and comments on the manuscript. We thank W. John and A. Muench-Wuttke from the Biomedical Services Facility for mouse care and injections. We acknowledge J. Peychl for the management of the Light Microscopy Facility and K. Manygoats as well as J.-M. Verbavatz for their support. We thank B. Bettencourt and J. Hettinger for siRNA design and serum biochemistry assays, respectively. This work was financially supported by the Virtual Liver initiative (http://www.virtual-liver.de), funded by the German Federal Ministry of Research and Education (BMBF), the Max Planck Society (MPG) and the DFG. A.Z. was supported by a grant from Marie Curie Action, Intra-European Fellowship (fp7-people-ief-2008) and J.G. from an EMBO long-term fellowship.
Author information
Authors and Affiliations
Contributions
M.Z. and V.K. conceived the project and M.Z. directed it. A.Z. designed and directed the animal injection experiments, and performed the stainings and imaging of liver sections. J.G. and A.Z. co-developed the staining procedures for the tissue sections. J.G.H. helped A.Z. to establish the hepatocyte isolation technique. A.Z. established the primary culture, developed the endocytosis assays, staining protocols and the knockdown technique with LNPs in primary hepatocytes. J.G. performed the electron microscopy analysis and quantifications, and the sectioning of liver tissue. R.L.B. selected siRNAs and validated their efficacy and specificity in vitro and in vivo, and designed, performed and analysed some in vivo experiments. H.E.-B. prepared the siRNAs into lipidoid-based formulations and analysed them. S.K. and C.G.P. performed the synthesis and analysis of siRNAs. V.M.R. designed, performed and analysed the 5′RACE assay. H.N. performed the RT–PCR in primary hepatocytes. S.S. under the supervision of A.Z. and J.G. performed the western blot analysis and performed the hepatocyte isolation and primary culturing under the supervision of A.Z.; G.M. under the supervision of Y.K. adapted the QMPIA for primary hepatocytes and liver tissue and performed the image analysis. Y.K. and P.dC.-Z. developed the mathematical model. M.Z., Y.K., A.Z. and J.G. wrote the manuscript, R.L.B. and V.K. participated in the editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-11, Supplementary Methods, Supplementary Model Descriptions, a Supplementary Discussion and Supplementary References. (PDF 2944 kb)
Supplementary Data 1
This zipped fie contains Supplementary Data. (ZIP 12403 kb)
Supplementary Data 2
This zipped fie contains Supplementary Data. (ZIP 38 kb)
Rights and permissions
About this article
Cite this article
Zeigerer, A., Gilleron, J., Bogorad, R. et al. Rab5 is necessary for the biogenesis of the endolysosomal system in vivo. Nature 485, 465–470 (2012). https://doi.org/10.1038/nature11133
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11133
This article is cited by
-
The Role of RIN3 Gene in Alzheimer’s Disease Pathogenesis: a Comprehensive Review
Molecular Neurobiology (2023)
-
BAR domain is essential for early endosomal trafficking and dynamics in Ascochyta rabiei
3 Biotech (2023)
-
Mesenchymal stem cell-derived extracellular vesicles for immunomodulation and regeneration: a next generation therapeutic tool?
Cell Death & Disease (2022)
-
Phosphoinositides as membrane organizers
Nature Reviews Molecular Cell Biology (2022)
-
The ESCRT-III molecules regulate the apical targeting of bile salt export pump
Journal of Biomedical Science (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.