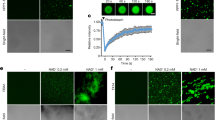
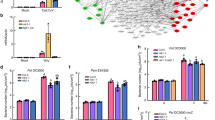
Abstract
Plant innate immunity is activated on the detection of pathogen-associated molecular patterns (PAMPs) at the cell surface, or of pathogen effector proteins inside the plant cell1,2,3,4. Together, PAMP-triggered immunity and effector-triggered immunity constitute powerful defences against various phytopathogens. Pathogenic bacteria inject a variety of effector proteins into the host cell to assist infection or propagation. A number of effector proteins have been shown to inhibit plant immunity5, but the biochemical basis remains unknown for the vast majority of these effectors. Here we show that the Xanthomonas campestris pathovar campestris type III effector AvrAC enhances virulence and inhibits plant immunity by specifically targeting Arabidopsis BIK1 and RIPK, two receptor-like cytoplasmic kinases known to mediate immune signalling6,7,8. AvrAC is a uridylyl transferase that adds uridine 5′-monophosphate to and conceals conserved phosphorylation sites in the activation loop of BIK1 and RIPK, reducing their kinase activity and consequently inhibiting downstream signalling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ausubel, F. M. Are innate immune signaling pathways in plants and animals conserved? Nature Immunol. 6, 973–979 (2005)
Chisholm, S. T., Coaker, G., Day, B. & Staskawicz, B. J. Host microbe interactions: shaping the evolution of the plant immune response. Cell 124, 803–814 (2006)
Jones, J. D. & Dangl, J. L. The plant immune system. Nature 444, 323–329 (2006)
Boller, T. & Felix, G. A renaissance of elicitors: perception of microbe-associated molecular patterns and danger signals by pattern-recognition receptors. Annu. Rev. Plant Biol. 60, 379–406 (2009)
Block, A. & Alfano, J. R. Plant targets for Pseudomonas syringae type III effectors: virulence targets or guarded decoys? Curr. Opin. Microbiol. 14, 39–46 (2011)
Lu, D. et al. A receptor-like cytoplasmic kinase, BIK1, associates with a flagellin receptor complex to initiate plant innate immunity. Proc. Natl Acad. Sci. USA 107, 496–501 (2010)
Zhang, J. et al. Receptor-like cytoplasmic kinases integrate signaling from multiple plant immune receptors and are targeted by a Pseudomonas syringae effector. Cell Host Microbe 7, 290–301 (2010)
Liu, J., Elmore, J. M., Lin, Z.-J. D. & Coaker, G. A receptor-like cytoplasmic kinase phosphorylates the host target RIN4, leading to the activation of a plant innate immune receptor. Cell Host Microbe 9, 137–146 (2011)
Xu, R.-Q. et al. AvrACXcc8004, a type III effector with a leucine-rich-repeat domain from Xanthomonas campestris pathovar campestris confers avirulence in vascular tissues of Arabidopsis thaliana ecotype Col-0. J. Bacteriol. 190, 343–355 (2008)
He, P. et al. Specific bacterial suppressors of MAMP signaling upstream of MAPKKK in Arabidopsis innate immunity. Cell 125, 563–575 (2006)
Segonzac, C. & Zipfel, C. Activation of plant pattern-recognition receptors by bacteria. Curr. Opin. Microbiol. 14, 54–61 (2011)
Chinchilla, D. et al. A flagellin-induced complex of the receptor FLS2 and BAK1 initiates plant defence. Nature 448, 497–500 (2007)
Heese, A. et al. The receptor-like kinase SERK3/BAK1 is a central regulator of innate immunity in plants. Proc. Natl Acad. Sci. USA 104, 12217–12222 (2007)
Chung, E.-H. et al. Specific threonine phosphorylation of a host target by two unrelated type III effectors activates a host innate immune receptor in plants. Cell Host Microbe 9, 125–136 (2011)
Woolery, A. R., Luong, P., Broberg, C. A. & Orth, K. AMPylation: something old is new again. Front. Microbiol. 1, 113 (2010)
Mattoo, S. et al. Comparative analysis of Histophilus Somni IbpA with other FIC enzymes reveals differences in substrate and nucleotide specificities. J. Biol. Chem. 286, 32834–32842 (2011)
Yarbrough, M. L. et al. AMPylation of Rho GTPases by Vibrio VopS disrupts effector binding and downstream signaling. Science 323, 269–272 (2009)
Worby, C. A. et al. The Fic domain: regulation of cell signaling by adenylylation. Mol. Cell 34, 93–103 (2009)
Muller, M. P. et al. The Legionella effector protein DrrA AMPylates the membrane traffic regulator Rab1b. Science 329, 946–949 (2010)
Mukherjee, S. et al. Modulation of Rab GTPase function by a protein phosphocholine transferase. Nature 477, 103–106 (2011)
Laluk, K. et al. Biochemical and genetic requirements for function of the immune response regulator BOTRYTIS-INDUCED KINASE1 in plant growth, ethylene signaling, and PAMP-triggered immunity in Arabidopsis. Plant Cell 23, 2831–2849 (2011)
Luong, P. et al. Kinetic and structural insights into the mechanism of AMPylation by VopS Fic domain. J. Biol. Chem. 285, 20155–20163 (2010)
Xiao, J. et al. Structural basis of Fic mediated adenylylation. Nature Struct. Mol. Biol. 17, 1004–1010 (2010)
Sun, W., Dunning, M. F., Pfund, C., Weingarten, R. & Bent, A. F. Within-species flagellin polymorphism in Xanthomonas campestris pv campestris and its impact on elicitation of Arabidopsis FLAGELLIN SENSING2-dependent defenses. Plant Cell 18, 764–779 (2006)
Veronese, P. et al. The membrane-anchored BOTRYTIS-INDUCED KINASE1 plays distinct roles in Arabidopsis resistance to necrotrophic and biotrophic pathogens. Plant Cell 18, 257–273 (2006)
White, F. F., Potnis, N., Jones J. B & Koebnik, R. The type III effectors of Xanthomonas. Mol. Plant Pathol. 10, 749–766 (2009)
Kinch, L. N., Yarbrough, M. L., Orth, K. & Grishin, N. V. Fido, a novel AMPylation domain common to Fic, Doc, and AvrB. PLoS ONE 4, e5818 (2009)
Mukherjee, S. et al. Yersinia YopJ acetylates and inhibits kinase activation by blocking phosphorylation. Science 312, 1211–1214 (2006)
Wang, L. F., Tang, X. Y. & He, C. Z. The bifunctional effector AvrXccC of Xanthomonas campestris pv. campestris requires plasma membrane-anchoring for host recognition. Mol. Plant Pathol. 8, 491–501 (2007)
Yuan, J. & He, S. Y. The Pseudomonas syringae Hrp regulation and secretion system controls the production and secretion of multiple extracellular proteins. J. Bacteriol. 178, 6399–6402 (1996)
Li, X. et al. Flagellin induces innate immunity in nonhost interactions that is suppressed by Pseudomonas syringae effectors. Proc. Natl Acad. Sci. USA 102, 12990–12995 (2005)
Schäfer, A. et al. Small mobilizable multi-purpose cloning vectors derived from the Escherichia coli plasmids pK18 and pK19: selection of defined deletions in the chromosome of Corynebacterium glutamicum. Gene 145, 69–73 (1994)
Zuo, J., Niu, Q. W. & Chua, N. H. An estrogen receptor-based transactivator XVE mediates highly inducible gene expression in transgenic plants. Plant J. 24, 265–273 (2000)
Zhang, J. et al. A Pseudomonas syringae effector inactivates MAPKs to suppress PAMP-induced immunity in plants. Cell Host Microbe 1, 175–185 (2007)
Schwessinger, B. et al. Phosphorylation-dependent differential regulation of plant growth, cell death, and innate immunity by the regulatory receptor-like kinase BAK1. PLoS Genet. 7, e1002046 (2011)
Macho, A. P. et al. Competitive index in mixed infections: a sensitive and accurate assay for the genetic analysis of Pseudomonas syringae–plant interactions. Mol. Plant Pathol. 8, 437–450 (2007)
Dow, J. M. et al. Biofilm dispersal in Xanthomonas campestris is controlled by cell-cell signaling and is required for full virulence to plants. Proc. Natl Acad. Sci. USA 100, 10995–11000 (2003)
Schechter, L. M. et al. Pseudomonas syringae type III secretion system targeting signals and novel effectors studied with a Cya translocation reporter. J. Bacteriol. 186, 543–555 (2004)
Cheng, W. et al. The AvrPtoB-BAK1 complex reveals two structurally similar kinase-interacting domains in a single type III effector. Cell Host Microbe 10, 616–626 (2011)
Acknowledgements
The authors thank J. Chai for sharing plasmids before publication, A. Bent for the XccB186 strain, S. Y. He and F. White for helpful comments. J.-M.Z. was supported by grants from the Chinese Ministry of Science and Technology (2011CB100700; 2010CB835301). S.C. was supported by a grant from the Chinese Ministry of Science and Technology (2010CB835204) C.H. was supported by Funds from Hainan University.
Author information
Authors and Affiliations
Contributions
S.C., C.H. and J.-M.Z. conceived and designed the experiments. F.F., F.Y., W.R., X.W. and J.Z. performed the experiments. F.F., F.Y., S.C., C.H. and J.-M.Z. analysed the data. F.F., S.C., C.H. and J.-M.Z. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1-16. (PDF 1597 kb)
Rights and permissions
About this article
Cite this article
Feng, F., Yang, F., Rong, W. et al. A Xanthomonas uridine 5′-monophosphate transferase inhibits plant immune kinases. Nature 485, 114–118 (2012). https://doi.org/10.1038/nature10962
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10962
This article is cited by
-
Plant immunity research in China
Phytopathology Research (2023)
-
Plant Immunity: A Plastic System Operated Through Cell-Fate Transition
Journal of Plant Biology (2023)
-
Evasion of plant immunity by microbial pathogens
Nature Reviews Microbiology (2022)
-
A secreted effector with a dual role as a toxin and as a transcriptional factor
Nature Communications (2022)
-
Regulation of plant responses to biotic and abiotic stress by receptor-like cytoplasmic kinases
Stress Biology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.