Abstract
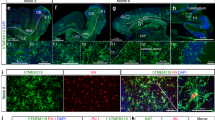
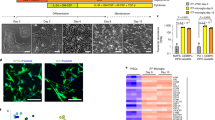
Rett syndrome is an X-linked autism spectrum disorder. The disease is characterized in most cases by mutation of the MECP2 gene, which encodes a methyl-CpG-binding protein1,2,3,4,5. Although MECP2 is expressed in many tissues, the disease is generally attributed to a primary neuronal dysfunction6. However, as shown recently, glia, specifically astrocytes, also contribute to Rett pathophysiology. Here we examine the role of another form of glia, microglia, in a murine model of Rett syndrome. Transplantation of wild-type bone marrow into irradiation-conditioned Mecp2-null hosts resulted in engraftment of brain parenchyma by bone-marrow-derived myeloid cells of microglial phenotype, and arrest of disease development. However, when cranial irradiation was blocked by lead shield, and microglial engraftment was prevented, disease was not arrested. Similarly, targeted expression of MECP2 in myeloid cells, driven by Lysmcre on an Mecp2-null background, markedly attenuated disease symptoms. Thus, through multiple approaches, wild-type Mecp2-expressing microglia within the context of an Mecp2-null male mouse arrested numerous facets of disease pathology: lifespan was increased, breathing patterns were normalized, apnoeas were reduced, body weight was increased to near that of wild type, and locomotor activity was improved. Mecp2+/− females also showed significant improvements as a result of wild-type microglial engraftment. These benefits mediated by wild-type microglia, however, were diminished when phagocytic activity was inhibited pharmacologically by using annexin V to block phosphatydilserine residues on apoptotic targets, thus preventing recognition and engulfment by tissue-resident phagocytes. These results suggest the importance of microglial phagocytic activity in Rett syndrome. Our data implicate microglia as major players in the pathophysiology of this devastating disorder, and suggest that bone marrow transplantation might offer a feasible therapeutic approach for it.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Van den Veyver, I. B. & Zoghbi, H. Y. Mutations in the gene encoding methyl-CpG-binding protein 2 cause Rett syndrome. Brain Dev. 23 (suppl. 1). S147–S151 (2001)
Van den Veyver, I. B. & Zoghbi, H. Y. Genetic basis of Rett syndrome. Ment. Retard. Dev. Disabil. Res. Rev. 8, 82–86 (2002)
Amir, R. E. et al. Rett syndrome is caused by mutations in X-linked MECP2, encoding methyl-CpG-binding protein 2. Nature Genet. 23, 185–188 (1999)
Guy, J., Hendrich, B., Holmes, M., Martin, J. E. & Bird, A. A mouse Mecp2-null mutation causes neurological symptoms that mimic Rett syndrome. Nature Genet. 27, 322–326 (2001)
Nan, X. & Bird, A. The biological functions of the methyl-CpG-binding protein MeCP2 and its implication in Rett syndrome. Brain Dev. 23 (suppl. 1). S32–S37 (2001)
Luikenhuis, S., Giacometti, E., Beard, C. F. & Jaenisch, R. Expression of MeCP2 in postmitotic neurons rescues Rett syndrome in mice. Proc. Natl Acad. Sci. USA 101, 6033–6038 (2004)
Ballas, N., Lioy, D. T., Grunseich, C. & Mandel, G. Non-cell autonomous influence of MeCP2-deficient glia on neuronal dendritic morphology. Nature Neurosci. 12, 311–317 (2009)
Maezawa, I., Swanberg, S., Harvey, D., LaSalle, J. M. & Jin, L. W. Rett syndrome astrocytes are abnormal and spread MeCP2 deficiency through gap junctions. J. Neurosci. 29, 5051–5061 (2009)
Lioy, D. T. et al. A role for glia in the progression of Rett’s syndrome. Nature 475, 497–500 (2011)
Maezawa, I. & Jin, L. W. Rett syndrome microglia damage dendrites and synapses by the elevated release of glutamate. J. Neurosci. 30, 5346–5356 (2010)
Ginhoux, F. et al. Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science 330, 841–845 (2010)
Ajami, B., Bennett, J. L., Krieger, C., Tetzlaff, W. & Rossi, F. M. Local self-renewal can sustain CNS microglia maintenance and function throughout adult life. Nature Neurosci. 10, 1538–1543 (2007)
Mildner, A. et al. Microglia in the adult brain arise from Ly-6ChiCCR2+ monocytes only under defined host conditions. Nature Neurosci. 10, 1544–1553 (2007)
Boissonneault, V. et al. Powerful beneficial effects of macrophage colony-stimulating factor on β-amyloid deposition and cognitive impairment in Alzheimer’s disease. Brain 132, 1078–1092 (2009)
Chen, S. K. et al. Hematopoietic origin of pathological grooming in Hoxb8 mutant mice. Cell 141, 775–785 (2010)
Hoogerbrugge, P. M. et al. Donor-derived cells in the central nervous system of twitcher mice after bone marrow transplantation. Science 239, 1035–1038 (1988)
Simard, A. R., Soulet, D., Gowing, G., Julien, J. P. & Rivest, S. Bone marrow-derived microglia play a critical role in restricting senile plaque formation in Alzheimer’s disease. Neuron 49, 489–502 (2006)
Shechter, R. et al. Infiltrating blood-derived macrophages are vital cells playing an anti-inflammatory role in recovery from spinal cord injury in mice. PLoS Med. 6, e1000113 (2009)
Chen, R. Z., Akbarian, S., Tudor, M. & Jaenisch, R. Deficiency of methyl-CpG binding protein-2 in CNS neurons results in a Rett-like phenotype in mice. Nature Genet. 27, 327–331 (2001)
Tropea, D. et al. Partial reversal of Rett syndrome-like symptoms in MeCP2 mutant mice. Proc. Natl Acad. Sci. USA 106, 2029–2034 (2009)
Willemen, H. L. et al. Microglial/macrophage GRK2 determines duration of peripheral IL-1β-induced hyperalgesia: contribution of spinal cord CX3CR1, p38 and IL-1 signaling. Pain 150, 550–560 (2010)
Nijboer, C. H. et al. Cell-specific roles of GRK2 in onset and severity of hypoxic-ischemic brain damage in neonatal mice. Brain Behav. Immun. 24, 420–426 (2010)
Cho, I. H. et al. Role of microglial IKKβ in kainic acid-induced hippocampal neuronal cell death. Brain 131, 3019–3033 (2008)
Lu, Z. et al. Phagocytic activity of neuronal progenitors regulates adult neurogenesis. Nature Cell Biol. 13, 1076–1083 (2011)
Zhang, X. et al. A minimally invasive, translational biomarker of ketamine-induced neuronal death in rats: microPET imaging using 18F-annexin V. Toxicol. Sci. 111, 355–361 (2009)
Oldfors, A. et al. Rett syndrome: cerebellar pathology. Pediatr. Neurol. 6, 310–314 (1990)
McGraw, C. M., Samaco, R. C. & Zoghbi, H. Y. Adult neural function requires MeCP2. Science 333, 186 (2011)
Park, D. et al. BAI1 is an engulfment receptor for apoptotic cells upstream of the ELMO/Dock180/Rac module. Nature 450, 430–434 (2007)
Guy, J., Gan, J., Selfridge, J., Cobb, S. & Bird, A. Reversal of neurological defects in a mouse model of Rett syndrome. Science 315, 1143–1147 (2007)
Drorbaugh, J. E. & Fenn, W. O. A barometric method for measuring ventilation in newborn infants. Pediatrics 16, 81–87 (1955)
Acknowledgements
We thank S. Smith for editing the manuscript. We thank the members of the Kipnis laboratory as well as the members of the University of Virginia Neuroscience Department for their comments during multiple discussions of this work. We also thank S. Feldman for injection of neonatal mice, I. Smirnov for tail vein injections, and B. Tomlin and J. Jones for their animal care. N.C.D. is a recipient of a Hartwell Foundation post-doctoral fellowship. This work was primarily supported by a grant from the Rett Syndrome Research Trust (to J.K.) and in part by HD056293 and AG034113 (to J.K).
Author information
Authors and Affiliations
Contributions
N.C.D. performed most of the experiments, analysed the data and prepared it for presentation, and contributed to experimental design and manuscript writing. J.C.C. performed the experiments with phagocytic activity of microglia in vivo and assisted with additional immunohistochemistry experiments along with data analysis and presentation, and contributed to experimental design and manuscript editing. Z.L. assisted with in vitro phagocytic activity experiments. E.X. assisted with animal behaviour scoring. S.B.G.A. assisted with plethysmography experiments and data analysis. P.G.G. assisted with plethysmography experimental design, data analysis and presentation of plethysmography-related data, and contributed to manuscript editing. J.K. designed the study, assisted with data analysis and presentation, and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1-6. (PDF 576 kb)
Supplementary Movie 1
Representative wild type and Mecp2−/y mice at ~7 weeks of age. Notice reduced size and activity of Mecp2−/y littermate. Representative appearance, tremors and clasping are shown in Mecp2−/y at 60 days of age. (MOV 17028 kb)
Supplementary Movie 2
Representative transplanted mice (wild-type → Mecp2−/yand wild-type → wild-type) are shown at 18 weeks of age (14 weeks post bone marrow transplantation). Note improved appearance, activity, body size, and lack of visible tremors in wild-type → Mecp2−/y mice. (MOV 10604 kb)
Supplementary Movie 3
Representative movie of wild-type → Mecp2−/y mouse at 40 weeks of age (4- to 5-fold increase in lifespan). (MOV 9627 kb)
Supplementary Movie 4
Genetic approach: Mecp2lox–stop mice were bred to LysmCre mice and their progeny ( Mecp2lox–stop/yLysmCre mice) are Mecp2 -null mice that express wild-type Mecp2 protein in myeloid cells (including microglia). Representative movie of these mice is shown. Note the body size and activity at 23 weeks of age. (MOV 18028 kb)
Rights and permissions
About this article
Cite this article
Derecki, N., Cronk, J., Lu, Z. et al. Wild-type microglia arrest pathology in a mouse model of Rett syndrome. Nature 484, 105–109 (2012). https://doi.org/10.1038/nature10907
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10907
This article is cited by
-
Role of Maternal Immune Factors in Neuroimmunology of Brain Development
Molecular Neurobiology (2023)
-
Strategies for Manipulating Microglia to Determine Their Role in the Healthy and Diseased Brain
Neurochemical Research (2023)
-
Methyl-CpG-Binding Protein 2 Emerges as a Central Player in Multiple Sclerosis and Neuromyelitis Optica Spectrum Disorders
Cellular and Molecular Neurobiology (2023)
-
Novel Microglia-based Therapeutic Approaches to Neurodegenerative Disorders
Neuroscience Bulletin (2023)
-
Thirty-year clinical outcomes after haematopoietic stem cell transplantation in neuronopathic Gaucher disease
Orphanet Journal of Rare Diseases (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.