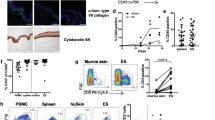
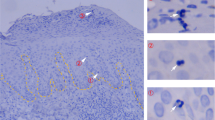
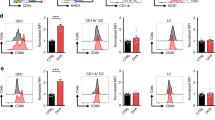
Abstract
Protective T-cell memory has long been thought to reside in blood and lymph nodes, but recently the concept of immune memory in peripheral tissues mediated by resident memory T (TRM) cells has been proposed1,2,3,4,5. Here we show in mice that localized vaccinia virus (VACV) skin infection generates long-lived non-recirculating CD8+ skin TRM cells that reside within the entire skin. These skin TRM cells are potent effector cells, and are superior to circulating central memory T (TCM) cells at providing rapid long-term protection against cutaneous re-infection. We find that CD8+ T cells are rapidly recruited to skin after acute VACV infection. CD8+ T-cell recruitment to skin is independent of CD4+ T cells and interferon-γ, but requires the expression of E- and P-selectin ligands by CD8+ T cells. Using parabiotic mice, we further show that circulating CD8+ TCM and CD8+ skin TRM cells are both generated after skin infection; however, CD8+ TCM cells recirculate between blood and lymph nodes whereas TRM cells remain in the skin. Cutaneous CD8+ TRM cells produce effector cytokines and persist for at least 6 months after infection. Mice with CD8+ skin TRM cells rapidly cleared a subsequent re-infection with VACV whereas mice with circulating TCM but no skin TRM cells showed greatly impaired viral clearance, indicating that TRM cells provide superior protection. Finally, we show that TRM cells generated as a result of localized VACV skin infection reside not only in the site of infection, but also populate the entire skin surface and remain present for many months. Repeated re-infections lead to progressive accumulation of highly protective TRM cells in non-involved skin. These findings have important implications for our understanding of protective immune memory at epithelial interfaces with the environment, and suggest novel strategies for vaccines that protect against tissue tropic organisms.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Liu, L. et al. Epidermal injury and infection during poxvirus immunization is crucial for the generation of highly protective T cell-mediated immunity. Nature Med. 16, 224–227 (2010)
Clark, R. A. et al. Skin effector memory T cells do not recirculate and provide immune protection in alemtuzumab-treated CTCL patients. Sci. Transl. Med. 4, 117 (2012)
Sheridan, B. S. & Lefrançois, L. Regional and mucosal memory T cells. Nature Immunol. 131, 485–491 (2011)
Boyman, O. et al. Spontaneous development of psoriasis in a new animal model shows an essential role for resident T cells and tumor necrosis factor-α. J. Exp. Med. 199, 731–736 (2004)
Conrad, C. et al. α1β1 integrin is crucial for accumulation of epidermal T cells and the development of psoriasis. Nature Med. 13, 836–842 (2007)
Bevan, M. J. Helping the CD8+ T-cell response. Nature Rev. Immunol. 4, 595–602 (2004)
Antia, R., Ganusov, V. V. & Ahmed, R. The role of models in understanding CD8+ T-cell memory. Nature Rev. Immunol. 5, 101–111 (2005)
Kaech, S. M. & Wherry, E. J. Heterogeneity and cell-fate decisions in effector and memory CD8+ T cell differentiation during viral infection. Immunity 27, 393–405 (2007)
Lefrançois, L. & Obar, J. J. Once a killer, always a killer: from cytotoxic T cell to memory cell. Immunol. Rev. 235, 206–218 (2010)
Freyschmidt, E. J. et al. Skin inflammation arising from cutaneous regulatory T cell deficiency leads to impaired viral immune responses. J. Immunol. 185, 1295–1302 (2010)
Sanz, P. & Moss, B. Identification of a transcription factor, encoded by two vaccinia virus early genes, that regulates the intermediate stage of viral gene expression. Proc. Natl Acad. Sci. USA 96, 2692–2697 (1999)
Nakanishi, Y., Lu, B., Gerard, C. & Iwasaki, A. CD8+ T lymphocyte mobilization to virus-infected tissue requires CD4+ T-cell help. Nature 462, 510–513 (2009)
Wakim, L. M., Waithman, J., van Rooijen, N., Heath, W. R. & Carbone, F. R. Dendritic cell-induced memory T cell activation in nonlymphoid tissues. Science 319, 198–202 (2008)
Masopust, D., Vezys, V., Marzo, A. L. & Lefrançois, L. Preferential localization of effector memory cells in nonlymphoid tissue. Science 291, 2413–2417 (2001)
Lefrançois, L. Development, trafficking, and function of memory T-cell subsets. Immunol. Rev. 211, 93–103 (2006)
Klonowski, K. D. et al. Dynamics of blood-borne CD8 memory T cell migration in vivo. Immunity 20, 551–562 (2004)
Gebhardt, T. et al. Different patterns of peripheral migration by memory CD4+ and CD8+ T cells. Nature 477, 216–219 (2011)
Chong, B. F., Murphy, J. E., Kupper, T. S. & Fuhlbrigge, R. C. E-selectin, thymus- and activation-regulated chemokine/CCL17, and intercellular adhesion molecule-1 are constitutively coexpressed in dermal microvessels: a foundation for a cutaneous immunosurveillance system. J. Immunol. 172, 1575–1581 (2004)
Weninger, W. et al. Specialized contributions by α(1,3)-fucosyltransferase-IV and FucT-VII during leukocyte rolling in dermal microvessels. Immunity 12, 665–676 (2000)
Jiang, X., Campbell, J. J. & Kupper, T. S. Embryonic trafficking of γδ T cells to skin is dependent on E/P selectin ligands and CCR4. Proc. Natl Acad. Sci. USA 107, 7443–7448 (2010)
Gebhardt, T. et al. Memory T cells in nonlymphoid tissue that provide enhanced local immunity during infection with herpes simplex virus. Nature Immunol. 10, 524–530 (2009)
Masopust, D. et al. Dynamic T cell migration program provides resident memory within intestinal epithelium. J. Exp. Med. 207, 553–564 (2010)
Wakim, L. M., Woodward-Davis, A. & Bevan, M. J. Memory T cells persisting within the brain after local infection show functional adaptations to their tissue of residence. Proc. Natl Acad. Sci. USA 107, 17872–17879 (2010)
Clark, R. A. et al. The vast majority of CLA+ T cells are resident in normal skin. J. Immunol. 176, 4431–4439 (2006)
Purwar, R. et al. Resident memory T cells (TRM) are abundant in human lung: diversity, function, and antigen specificity. PLoS ONE 6, e16245 (2011)
Lund, J. M., Hsing, L., Pham, T. T. & Rudensky, A. Y. Coordination of early protective immunity to viral infection by regulatory T cells. Science 320, 1220–1224 (2008)
Román, E. et al. CD4 effector T cell subsets in the response to influenza: heterogeneity, migration, and function. J. Exp. Med. 196, 957–968 (2002)
Romani, L. Immunity to fungal infections. Nature Rev. Immunol. 11, 275–288 (2011)
Liu, L., Fuhlbrigge, R. C., Karibian, K., Tian, T. & Kupper, T. S. Dynamic programming of CD8+ T cell trafficking after live viral immunization. Immunity 25, 511–520 (2006)
Wagers, A. J., Sherwood, R. I., Christensen, J. L. & Weissman, I. L. Little evidence for developmental plasticity of adult hematopoietic stem cells. Science 297, 2256–2259 (2002)
Acknowledgements
We thank T. Tian, R. Purwar and Q. Zhan for technical assistance. We thank J. J. Campbell for discussion of the project. This work was supported by National Institutes of Health (NIH) grants R01AI041707, R37AI025082 and TR01 AI097128 to T.S.K.
Author information
Authors and Affiliations
Contributions
X.J. and T.S.K. designed research; X.J. performed research; L.L. helped to establish the VACV skin scarification model; A.J.W. helped to create parabiotic mice; X.J., R.A.C., R.C.F., L.L. and T.S.K. analysed data; and X.J., R.A.C. and T.S.K. wrote the paper.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1-5 with legends. (PDF 328 kb)
Rights and permissions
About this article
Cite this article
Jiang, X., Clark, R., Liu, L. et al. Skin infection generates non-migratory memory CD8+ TRM cells providing global skin immunity. Nature 483, 227–231 (2012). https://doi.org/10.1038/nature10851
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10851
This article is cited by
-
3D sheep rumen epithelial structures driven from single cells in vitro
Veterinary Research (2023)
-
Transient regulatory-T-cell interruption promotes skin-resident memory T cells mediated tumor protection
Scientific Reports (2023)
-
Assessing the generation of tissue resident memory T cells by vaccines
Nature Reviews Immunology (2023)
-
Unique properties of tissue-resident memory T cells in the lungs: implications for COVID-19 and other respiratory diseases
Nature Reviews Immunology (2023)
-
A virtual memory CD8+ T cell-originated subset causes alopecia areata through innate-like cytotoxicity
Nature Immunology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.