Abstract
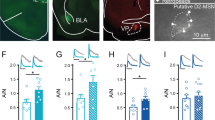
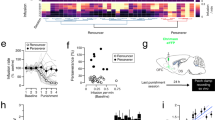
Drug-evoked synaptic plasticity is observed at many synapses and may underlie behavioural adaptations in addiction1. Mechanistic investigations start with the identification of the molecular drug targets. Cocaine, for example, exerts its reinforcing2 and early neuroadaptive effects3 by inhibiting the dopamine transporter, thus causing a strong increase in mesolimbic dopamine. Among the many signalling pathways subsequently engaged, phosphorylation of the extracellular signal-regulated kinase (ERK) in the nucleus accumbens4 is of particular interest because it has been implicated in NMDA-receptor and type 1 dopamine (D1)-receptor-dependent synaptic potentiation5 as well as in several behavioural adaptations6,7,8. A causal link between drug-evoked plasticity at identified synapses and behavioural adaptations, however, is missing, and the benefits of restoring baseline transmission have yet to be demonstrated. Here we find that cocaine potentiates excitatory transmission in D1-receptor-expressing medium-sized spiny neurons (D1R-MSNs) in mice via ERK signalling with a time course that parallels locomotor sensitization. Depotentiation of cortical nucleus accumbens inputs by optogenetic stimulation in vivo efficiently restored normal transmission and abolished cocaine-induced locomotor sensitization. These findings establish synaptic potentiation selectively in D1R-MSNs as a mechanism underlying a core component of addiction, probably by creating an imbalance between distinct populations of MSNs in the nucleus accumbens. Our data also provide proof of principle that reversal of cocaine-evoked synaptic plasticity can treat behavioural alterations caused by addictive drugs and may inspire novel therapeutic approaches involving deep brain stimulation or transcranial magnetic stimulation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lüscher, C. & Malenka, R. C. Drug-evoked synaptic plasticity in addiction: from molecular changes to circuit remodeling. Neuron 69, 650–663 (2011)
Chen, R. et al. Abolished cocaine reward in mice with a cocaine-insensitive dopamine transporter. Proc. Natl Acad. Sci. USA 103, 9333–9338 (2006)
Brown, M. T. et al. Drug-driven AMPA receptor redistribution mimicked by selective dopamine neuron stimulation. PLoS ONE 5, e15870 (2010)
Valjent, E., Pages, C., Herve, D., Girault, J. A. & Caboche, J. Addictive and non-addictive drugs induce distinct and specific patterns of ERK activation in mouse brain. Eur. J. Neurosci. 19, 1826–1836 (2004)
Pascoli, V. et al. Cyclic adenosine monophosphate-independent tyrosine phosphorylation of NR2B mediates cocaine-induced extracellular signal-regulated kinase activation. Biol. Psychiatry 69, 218–227 (2011)
Lu, L., Koya, E., Zhai, H., Hope, B. T. & Shaham, Y. Role of ERK in cocaine addiction. Trends Neurosci. 29, 695–703 (2006)
Girault, J. A., Valjent, E., Caboche, J. & Herve, D. ERK2: a logical AND gate critical for drug-induced plasticity? Curr. Opin. Pharmacol. 7, 77–85 (2007)
Thomas, M. J., Kalivas, P. W. & Shaham, Y. Neuroplasticity in the mesolimbic dopamine system and cocaine addiction. Br. J. Pharmacol. 154, 327–342 (2008)
Bertran-Gonzalez, J. et al. Opposing patterns of signaling activation in dopamine D1 and D2 receptor-expressing striatal neurons in response to cocaine and haloperidol. J. Neurosci. 28, 5671–5685 (2008)
Kramer, P. F. et al. Dopamine D2 receptor overexpression alters behavior and physiology in Drd2-EGFP mice. J. Neurosci. 31, 126–132 (2011)
Lammel, S., Ion, D. I., Roeper, J. & Malenka, R. C. Projection-specific modulation of dopamine neuron synapses by aversive and rewarding stimuli. Neuron 70, 855–862 (2011)
Wolf, M. E. The Bermuda Triangle of cocaine-induced neuroadaptations. Trends Neurosci. 33, 391–398 (2010)
Kourrich, S., Rothwell, P. E., Klug, J. R. & Thomas, M. J. Cocaine experience controls bidirectional synaptic plasticity in the nucleus accumbens. J. Neurosci. 27, 7921–7928 (2007)
Boudreau, A. C. & Wolf, M. E. Behavioral sensitization to cocaine is associated with increased AMPA receptor surface expression in the nucleus accumbens. J. Neurosci. 25, 9144–9151 (2005)
Boudreau, A. C., Reimers, J. M., Milovanovic, M. & Wolf, M. E. Cell surface AMPA receptors in the rat nucleus accumbens increase during cocaine withdrawal but internalize after cocaine challenge in association with altered activation of mitogen-activated protein kinases. J. Neurosci. 27, 10621–10635 (2007)
English, J. D. & Sweatt, J. D. A requirement for the mitogen-activated protein kinase cascade in hippocampal long term potentiation. J. Biol. Chem. 272, 19103–19106 (1997)
Xie, G. Q. et al. Ethanol attenuates the HFS-induced, ERK-mediated LTP in a dose-dependent manner in rat striatum. Alcohol. Clin. Exp. Res. 33, 121–128 (2009)
Shen, W., Flajolet, M., Greengard, P. & Surmeier, D. J. Dichotomous dopaminergic control of striatal synaptic plasticity. Science 321, 848–851 (2008)
Zhu, J. J., Qin, Y., Zhao, M., Van Aelst, L. & Malinow, R. Ras and Rap control AMPA receptor trafficking during synaptic plasticity. Cell 110, 443–455 (2002)
Valjent, E. et al. Regulation of a protein phosphatase cascade allows convergent dopamine and glutamate signals to activate ERK in the striatum. Proc. Natl Acad. Sci. USA 102, 491–496 (2005)
Pierce, R. C., Pierce-Bancroft, A. F. & Prasad, B. M. Neurotrophin-3 contributes to the initiation of behavioral sensitization to cocaine by activating the Ras/mitogen-activated protein kinase signal transduction cascade. J. Neurosci. 19, 8685–8695 (1999)
Valjent, E. et al. Mechanisms of locomotor sensitization to drugs of abuse in a two-injection protocol. Neuropsychopharmacology 35, 401–415 (2010)
Brami-Cherrier, K., Roze, E., Girault, J. A., Betuing, S. & Caboche, J. Role of the ERK/MSK1 signalling pathway in chromatin remodelling and brain responses to drugs of abuse. J. Neurochem. 108, 1323–1335 (2009)
Sesack, S. R., Deutch, A. Y., Roth, R. H. & Bunney, B. S. Topographical organization of the efferent projections of the medial prefrontal cortex in the rat: an anterograde tract-tracing study with Phaseolus vulgaris leucoagglutinin. J. Comp. Neurol. 290, 213–242 (1989)
LaLumiere, R. T., Niehoff, K. E. & Kalivas, P. W. The infralimbic cortex regulates the consolidation of extinction after cocaine self-administration. Learn. Mem. 17, 168–175 (2010)
Lobo, M. K. et al. Cell type-specific loss of BDNF signaling mimics optogenetic control of cocaine reward. Science 330, 385–390 (2010)
Conrad, K. L. et al. Formation of accumbens GluR2-lacking AMPA receptors mediates incubation of cocaine craving. Nature 454, 118–121 (2008)
Anderson, S. M. et al. CaMKII: a biochemical bridge linking accumbens dopamine and glutamate systems in cocaine seeking. Nature Neurosci. 11, 344–353 (2008)
Robinson, T. E. & Kolb, B. Structural plasticity associated with exposure to drugs of abuse. Neuropharmacology 47 (suppl. 1). 33–46 (2004)
Vanderschuren, L. J. & Pierce, R. C. Sensitization processes in drug addiction. Curr. Top. Behav. Neurosci. 3, 179–195 (2010)
Mameli, M. et al. Cocaine-evoked synaptic plasticity: persistence in the VTA triggers adaptations in the nucleus accumbens. Nature Neurosci. 12, 1036–1041 (2009)
Gong, S. et al. Targeting Cre recombinase to specific neuron populations with bacterial artificial chromosome constructs. J. Neurosci. 27, 9817–9823 (2007)
Kombian, S. B. & Malenka, R. C. Simultaneous LTP of non-NMDA- and LTD of NMDA-receptor-mediated responses in the nucleus accumbens. Nature 368, 242–246 (1994)
Pennartz, C. M., Ameerun, R. F., Groenewegen, H. J. & Lopes da Silva, F. H. Synaptic plasticity in an in vitro slice preparation of the rat nucleus accumbens. Eur. J. Neurosci. 5, 107–117 (1993)
Acknowledgements
We thank P. Kalivas and the members of the Lüscher laboratory for suggestions on the manuscript. This work is supported by the Swiss National Science Foundation (C.L.) and “Synapsy”, a National Competence Center in Research (NCCR) of the Swiss Confederation on the synaptic basis of mental disorders.
Author information
Authors and Affiliations
Contributions
V.P. carried out all electrophysiology experiments and was helped by M.T. with the behavioural experiments. C.L. designed the study and wrote the manuscript together with V.P. and M.T.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
The file contains Supplementary Figures 1-14 with legends. (PDF 920 kb)
Rights and permissions
About this article
Cite this article
Pascoli, V., Turiault, M. & Lüscher, C. Reversal of cocaine-evoked synaptic potentiation resets drug-induced adaptive behaviour. Nature 481, 71–75 (2012). https://doi.org/10.1038/nature10709
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10709
This article is cited by
-
Potentiation of the lateral habenula-ventral tegmental area pathway underlines the susceptibility to depression in mice with chronic pain
Science China Life Sciences (2024)
-
Whole-brain tracking of cocaine and sugar rewards processing
Translational Psychiatry (2023)
-
Cell-type specific synaptic plasticity in dorsal striatum is associated with punishment-resistance compulsive-like cocaine self-administration in mice
Neuropsychopharmacology (2023)
-
Drug reinforcement impairs cognitive flexibility by inhibiting striatal cholinergic neurons
Nature Communications (2023)
-
High frequency DBS-like optogenetic stimulation of nucleus accumbens dopamine D2 receptor-containing neurons attenuates cocaine reinstatement in male rats
Neuropsychopharmacology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.