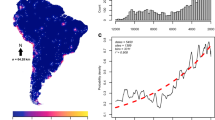
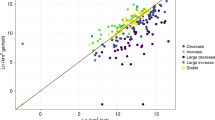
Abstract
Despite decades of research, the roles of climate and humans in driving the dramatic extinctions of large-bodied mammals during the Late Quaternary period remain contentious. Here we use ancient DNA, species distribution models and the human fossil record to elucidate how climate and humans shaped the demographic history of woolly rhinoceros, woolly mammoth, wild horse, reindeer, bison and musk ox. We show that climate has been a major driver of population change over the past 50,000 years. However, each species responds differently to the effects of climatic shifts, habitat redistribution and human encroachment. Although climate change alone can explain the extinction of some species, such as Eurasian musk ox and woolly rhinoceros, a combination of climatic and anthropogenic effects appears to be responsible for the extinction of others, including Eurasian steppe bison and wild horse. We find no genetic signature or any distinctive range dynamics distinguishing extinct from surviving species, emphasizing the challenges associated with predicting future responses of extant mammals to climate and human-mediated habitat change.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Barnosky, A. D., Koch, P. L., Feranec, R. S., Wing, S. L. & Shabel, A. B. Assessing the causes of Late Pleistocene extinctions on the continents. Science 306, 70–75 (2004)
Martin, P. S. in Quaternary Extinctions: A Prehistoric Revolution (eds Martin, P. S. & Klein, R. G. ) 364–403 (Univ. Arizona Press, 1984)
Alroy, J. A. multispecies overkill simulation of the end-Pleistocene megafaunal mass extinction. Science 292, 1893–1896 (2001)
Stuart, A. J., Kosintsev, P. A., Higham, T. F. G. & Lister, A. M. Pleistocene to Holocene extinction dynamics in giant deer and woolly mammoth. Nature 431, 684–689 (2004)
Koch, P. L. & Barnosky, A. D. Late Quaternary extinctions: state of the debate. Annu. Rev. Ecol. Evol. Syst. 37, 215–250 (2006)
Nogués-Bravo, D., Ohlemüller, R., Batra, P. & Araújo, M. B. Climate predictors of Late Quaternary extinctions. Evolution 64, 2442–2449 (2010)
Haile, J. et al. Ancient DNA reveals late survival of mammoth and horse in interior Alaska. Proc. Natl Acad. Sci. USA 106, 22363–22368 (2009)
Shapiro, B. et al. Rise and fall of the Beringian steppe bison. Science 306, 1561–1565 (2004)
Drummond, A. J., Rambaut, A., Shapiro, B. & Pybus, O. G. Bayesian coalescent inference of past population dynamics from molecular sequences. Mol. Biol. Evol. 22, 1185–1192 (2005)
Campos, P. F. et al. Ancient DNA analyses exclude humans as the driving force behind Late Pleistocene musk ox (Ovibos moschatus) population dynamics. Proc. Natl Acad. Sci. USA 107, 5675–5680 (2010)
Stiller, M. et al. Withering away—25,000 years of genetic decline preceded cave bear extinction. Mol. Biol. Evol. 27, 975–978 (2010)
Barnes, I. et al. Genetic structure and extinction of the woolly mammoth, Mammuthus primigenius . Curr. Biol. 17, 1–4 (2007)
Debruyne, R. et al. Out of America: ancient DNA evidence for a new world origin of Late Quaternary woolly mammoths. Curr. Biol. 18, 1320–1326 (2008)
Barnett, R., Yamaguchi, N., Barnes, I. & Cooper, A. The origin, current diversity, and future conservation of the modern lion (Panthera leo). Proc. R. Soc. B 273, 2159–2168 (2006)
Guthrie, R. D. Rapid body size decline in Alaskan Pleistocene horses before extinction. Nature 426, 169–171 (2003)
Grayson, D. K. Deciphering North American Pleistocene extinctions. J. Anthropol. Res. 63, 185–214 (2007)
Andrewartha, H. G. & Birch, L. C. The Distribution and Abundance of Animals (Univ. Chicago Press, 1954)
Borregaard, M. K. & Rahbek, C. Causality in the relationship between geographic distribution and species abundance. Q. Rev. Biol. 85, 3–25 (2010)
Minin, V. N., Bloomquist, E. W. & Suchard, M. A. Smooth skyride through a rough skyline: Bayesian coalescent-based inference of population dynamics. Mol. Biol. Evol. 25, 1459–1471 (2008)
Zazula, G. D. et al. Ice age steppe vegetation in east Beringia. Nature 423, 603 (2003)
Zazula, G. D., Froese, D. G., Elias, S. A., Kuzmina, S. & Mathewes, R. W. Arctic ground squirrels of the mammoth-steppe: paleoecology of Late Pleistocene middens (∼24 000–29 450 14C yr BP), Yukon Territory, Canada. Quat. Sci. Rev. 26, 979–1003 (2007)
Pitulko, V. V. et al. The Yana RHS site: humans in the Arctic before the last glacial maximum. Science 303, 52–56 (2004)
Goebel, T., Waters, M. R. & O’Rourke, D. H. The Late Pleistocene dispersal of modern humans in the Americas. Science 319, 1497–1502 (2008)
Stuart, A. J., Sulerzhitsky, L. D., Orlova, L. A., Kuzmin, Y. V. & Lister, A. M. The latest woolly mammoths (Mammuthus primigenius Blumenbach) in Europe and Asia: a review of the current evidence. Quat. Sci. Rev. 21, 1559–1569 (2002)
Vereshchagin, N. K. Prehistoric hunting and the extinction of Pleistocene mammals in the USSR. Proc. Zool. Inst. Russ. Acad. Sci. 69, 200–232 (1971)
Kuznetsova, T. V., Sulerzhitsky, L. D., Siegert, C. & Schirrmeister, L. (2001) in La Terra degli Elefanti (eds Cavarretta, G. Giola, P., Mussi, M. & Palombo, M. R. ) The World of Elephants, Proc. 1st Int. Congr. 289–292. (2001)
Wilson, R. J., Thomas, C. D., Fox, R., Roy, D. B. & Kunin, W. E. Spatial patterns in species distributions reveal biodiversity change. Nature 432, 393–396 (2004)
MacPhee, R. D. E. & Marx, P. A. in Natural Change and Human Impact in Madagascar (eds Goodman, S. M. & Patterson, B. D.) 169–217 (Smithsonian Institution Press, 1997)
Tener, J. S. Muskoxen in Canada: a biological and taxonomic review. Canadian Wildlife Service Monograph Series No. 2 (1965)
Ugan, A. & Byers, D. A global perspective on the spatiotemporal pattern of the Late Pleistocene human and woolly mammoth radiocarbon record. Quaternary Int. 191, 69–81 (2008)
Surovell, T. A. & Waguespack, N. M. How many elephant kills are 14? Clovis mammoth and mastodon kills in context. Quaternary Int. 191, 82–97 (2008)
Zielinski, G. A. & Mershon, G. R. Paleoenvironmental implications of the insoluble microparticle record in the GISP2 (Greenland) ice core during the rapidly changing climate of the Pleistocene–Holocene transition. Geol. Soc. Am. Bull. 109, 547–559 (1997)
Guthrie, R. D. New carbon dates link climatic change with human colonization and Pleistocene extinctions. Nature 441, 207–209 (2006)
Farnell, R. et al. Multidisciplinary investigations of alpine ice patches in southwest Yukon, Canada: paleoenvironmental and paleobiological investigations. Arctic 57, 247–259 (2004)
Williams, T. M. & Heard, D. C. World status of wild Rangifer tarandus population. Rangifer 1 (special issue). 19–28 (1986)
Joly, K., Klein, D. R., Verbyla, D. L., Rupp, T. S. & Chapin, F. S. Linkages between large-scale climate patterns and the dynamics of Arctic caribou populations. Ecography 34, 345–352 (2011)
Skogland, T. The effects of density-dependent resource limitation on the demography of wild reindeer. J. Anim. Ecol. 54, 359–374 (1985)
Leader-Williams, N. Reindeer on South Georgia Ch. 1, 3–18 (Cambridge Univ. Press, 1988)
Beaumont, M. in Simulations, Genetics and Human Prehistory (eds Matsumura, S. Forster, P. & Renfrew, C. ) 134–154 (McDonald Institute for Archaeological Research, 2008)
Reimer, P. J. et al. IntCal09 and Marine09 radiocarbon age calibration curves, 0–50,000 years cal BP. Radiocarbon 51, 1111–1150 (2009)
Nogués-Bravo, D., Rodríguez, J., Hortal, J., Batra, P. & Araújo, M. B. Climate change, humans, and the extinction of the woolly mammoth. PLoS Biol. 6, e79 (2008)
Drummond, A. J. & Rambaut, A. BEAST: Bayesian evolutionary analysis by sampling trees. BMC Evol. Biol. 7, 214 (2007)
Acknowledgements
This paper is in memory of our friend and colleague Andrei Sher, who was a contributor to this study. Dr Sher died unexpectedly, but his major contributions to the field of Quaternary science will be remembered and appreciated for many years. We are grateful to A. Lister and T. Stuart for guidance and discussions. We thank T. B. Brandt, B. Hockett and A. Telka for laboratory help and samples, and L. M. R. Thrane for his work on the megafauna locality database. Data taken from the Stage 3 project were partly funded by grant F/757/A from the Leverhulme Trust, and a grant from the McDonald Grants and Awards Fund. B.S. was supported by NSF ARC-0909456. We acknowledge the Danish National Research Foundation, the Lundbeck Foundation, the Danish Council for Independent Research and the US National Science Foundation for financial support.
Author information
Authors and Affiliations
Contributions
E.W. conceived and headed the overall project. C.R. headed the species distribution modelling and range measurements. E.D.L. and J.T.S. extracted, amplified and sequenced the reindeer DNA sequences. J.B. extracted, amplified and sequenced the woolly rhinoceros DNA sequences; M.H. generated part of the woolly rhinoceros data. J.W., K.-P.K., J.L. and R.K.W. generated the horse DNA sequences; A.C. generated part of the horse data. L.O., E.D.L. and B.S. analysed the genetic data, with input from R.N., K.M., M.A.S. and S.Y.W.H. Palaeoclimate simulations were provided by P.B., A.M.H, J.S.S. and P.J.V. The directly dated spatial latitudinal/longitudinal megafauna locality information was collected by E.D.L., K.A.M., D.N.-B., D.B. and A.U.; K.A.M. and D.N.-B. performed the species distribution modelling and range measurements. M.B. carried out the gene–climate correlation. A.U. and D.B. assembled the human Upper Palaeolithic sites from Eurasia. T.G. and K.E.G. assembled the archaeofaunal assemblages from Siberia. A.U. analysed the spatial overlap of humans and megafauna and the archaeofaunal assemblages. E.D.L., L.O., B.S., K.A.M., D.N.-B., M.K.B., A.U., T.G. and K.E.G. wrote the Supplementary Information. D.F., G.Z., T.W.S., K.A.-S., G.B., J.A.B., D.L.J., P.K., T.K., X.L., L.D.M., H.G.M., D.M., M.M., E.S., M.S., R.S.S., T.S., E.S., A.T., R.W. and A.C. provided the megafauna samples used for ancient DNA analysis. E.D.L. produced the figures. E.D.L, L.O. and E.W. wrote most of the manuscript, with input from B.S., M.H., D.N.-B., K.A.M., M.T.P.G., C.R., R.K.W, A.U. and the remaining authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
The file contains Supplementary Sections 1-6 (see page 1 for details), Supplementary Figures with legends, Supplementary Tables and additional references. (PDF 18259 kb)
Rights and permissions
About this article
Cite this article
Lorenzen, E., Nogués-Bravo, D., Orlando, L. et al. Species-specific responses of Late Quaternary megafauna to climate and humans. Nature 479, 359–364 (2011). https://doi.org/10.1038/nature10574
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10574
This article is cited by
-
Ancient mitogenomes reveal a high maternal genetic diversity of Pleistocene woolly rhinoceros in Northern China
BMC Ecology and Evolution (2023)
-
Whole-genome sequencing reveals adaptations of hairy-footed jerboas (Dipus, Dipodidae) to diverse desert environments
BMC Biology (2023)
-
Mitogenomes revealed the history of bison colonization of Northern Plains after the Last Glacial Maximum
Scientific Reports (2023)
-
Climate-driven habitat shifts of high-ranked prey species structure Late Upper Paleolithic hunting
Scientific Reports (2023)
-
The paleo-synanthropic niche: a first attempt to define animal’s adaptation to a human-made micro-environment in the Late Pleistocene
Archaeological and Anthropological Sciences (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.