Abstract
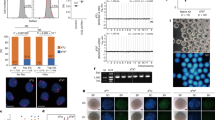
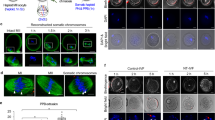
The exchange of the oocyte’s genome with the genome of a somatic cell, followed by the derivation of pluripotent stem cells, could enable the generation of specific cells affected in degenerative human diseases. Such cells, carrying the patient’s genome, might be useful for cell replacement. Here we report that the development of human oocytes after genome exchange arrests at late cleavage stages in association with transcriptional abnormalities. In contrast, if the oocyte genome is not removed and the somatic cell genome is merely added, the resultant triploid cells develop to the blastocyst stage. Stem cell lines derived from these blastocysts differentiate into cell types of all three germ layers, and a pluripotent gene expression program is established on the genome derived from the somatic cell. This result demonstrates the feasibility of reprogramming human cells using oocytes and identifies removal of the oocyte genome as the primary cause of developmental failure after genome exchange.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
Data deposits
Illumina array data have been deposited at GEO under accession number GSE28024.
References
Wilmut, I., Schnieke, A. E., McWhir, J., Kind, A. J. & Campbell, K. H. Viable offspring derived from fetal and adult mammalian cells. Nature 385, 810–813 (1997)
Thomson, J. A. et al. Embryonic stem cell lines derived from human blastocysts. Science 282, 1145–1147 (1998)
French, A. J. et al. Development of human cloned blastocysts following somatic cell nuclear transfer with adult fibroblasts. Stem Cells 26, 485–493 (2008)
Hall, V. J. et al. Developmental competence of human in vitro aged oocytes as host cells for nuclear transfer. Hum. Reprod. 22, 52–62 (2007)
Stojkovic, M. et al. Derivation of a human blastocyst after heterologous nuclear transfer to donated oocytes. Reprod. Biomed. Online 11, 226–231 (2005)
Cibelli, J. et al. Somatic cell nuclear transfer in humans: pronuclear and early embryonic development. J. Regen. Med. 2, 25–31 (2001)
Kennedy, D. Editorial retraction. Science 311, 335 (2006)
McElroy, S. L. et al. Developmental competence of immature and failed/abnormally fertilized human oocytes in nuclear transfer. Reprod. Biomed. Online 16, 684–693 (2008)
Chung, Y. et al. Reprogramming of human somatic cells using human and animal oocytes. Cloning Stem Cells 11, 213–223 (2009)
Heindryckx, B., De Sutter, P., Gerris, J., Dhont, M. & Van der Elst, J. Embryo development after successful somatic cell nuclear transfer to in vitro matured human germinal vesicle oocytes. Hum. Reprod. 22, 1982–1990 (2007)
Egli, D., Chen, A. E., Melton, D. & Eggan, K. Impracticality of egg donor recruitment in the absence of compensation. Cell Stem Cell 10.1016/j.stem.2011.08.002 (in the press)
Takahashi, K. et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 (2007)
Chin, M. H. et al. Induced pluripotent stem cells and embryonic stem cells are distinguished by gene expression signatures. Cell Stem Cell 5, 111–123 (2009)
Ghosh, Z. et al. Persistent donor cell gene expression among human induced pluripotent stem cells contributes to differences with human embryonic stem cells. PLoS ONE 5, e8975 (2010)
Doi, A. et al. Differential methylation of tissue- and cancer-specific CpG island shores distinguishes human induced pluripotent stem cells, embryonic stem cells and fibroblasts. Nature Genet. 41, 1350–1353 (2009)
Lister, R. et al. Hotspots of aberrant epigenomic reprogramming in human induced pluripotent stem cells. Nature 471, 68–73 (2011)
Hu, B. Y. et al. Neural differentiation of human induced pluripotent stem cells follows developmental principles but with variable potency. Proc. Natl Acad. Sci. USA 107, 4335–4340 (2010)
Gore, A. et al. Somatic coding mutations in human induced pluripotent stem cells. Nature 471, 63–67 (2011)
Hussein, S. M. et al. Copy number variation and selection during reprogramming to pluripotency. Nature 471, 58–62 (2011)
Mayshar, Y. et al. Identification and classification of chromosomal aberrations in human induced pluripotent stem cells. Cell Stem Cell 7, 521–531 (2010)
Revazova, E. S. et al. Patient-specific stem cell lines derived from human parthenogenetic blastocysts. Cloning Stem Cells 9, 432–449 (2007)
Egli, D., Chen, A. E., Melton, D. & Eggan, K. Reprogramming occurs within hours after mouse but not human nuclear transfer. Nature Commun. 10.1038/ncomms1503 (in the press)
Klitzman, R. & Sauer, M. V. Payment of egg donors in stem cell research in the USA. Reprod. Biomed. Online 18, 603–608 (2009)
Society for Assisted Reproductive Technology & the American Society for Reproductive Medicine . Assisted reproductive technology in the United States: 2001 results generated from the American Society for Reproductive Medicine/Society for Assisted Reproductive Technology registry. Fertil. Steril. 87, 1253–1266 (2007)
The Ethics Committee of the American Society for Reproductive Medicine . Financial compensation of oocyte donors. Fertil. Steril. 88, 305–309 (2007)
Daley, G. Q. et al. Ethics. The ISSCR guidelines for human embryonic stem cell research. Science 315, 603–604 (2007)
Mitalipov, S. M. et al. Reprogramming following somatic cell nuclear transfer in primates is dependent upon nuclear remodeling. Hum. Reprod. 22, 2232–2242 (2007)
Braude, P., Bolton, V. & Moore, S. Human gene expression first occurs between the four- and eight-cell stages of preimplantation development. Nature 332, 459–461 (1988)
Draper, J. S. et al. Recurrent gain of chromosomes 17q and 12 in cultured human embryonic stem cells. Nature Biotechnol. 22, 53–54 (2004)
Zhang, K. et al. Digital RNA allelotyping reveals tissue-specific and allele-specific gene expression in human. Nature Methods 6, 613–618 (2009)
Cowan, C. A., Atienza, J., Melton, D. A. & Eggan, K. Nuclear reprogramming of somatic cells after fusion with human embryonic stem cells. Science 309, 1369–1373 (2005)
Kim, K. et al. Epigenetic memory in induced pluripotent stem cells. Nature 467, 285–290 (2010)
Chen, A. E. et al. Optimal timing of inner cell mass isolation increases the efficiency of human embryonic stem cell derivation and allows generation of sibling cell lines. Cell Stem Cell 4, 103–106 (2009)
Cowan, C. A. et al. Derivation of embryonic stem-cell lines from human blastocysts. N. Engl. J. Med. 350, 1353–1356 (2004)
Dimos, J. T. et al. Induced pluripotent stem cells generated from patients with ALS can be differentiated into motor neurons. Science 321, 1218–1221 (2008)
Freberg, C. T., Dahl, J. A., Timoskainen, S. & Collas, P. Epigenetic reprogramming of OCT4 and NANOG regulatory regions by embryonal carcinoma cell extract. Mol. Biol. Cell 18, 1543–1553 (2007)
Imamura, M. et al. Transcriptional repression and DNA hypermethylation of a small set of ES cell marker genes in male germline stem cells. BMC Dev. Biol. 6, 34 (2006)
Pick, M. et al. Clone- and gene-specific aberrations of parental imprinting in human induced pluripotent stem cells. Stem Cells 27, 2686–2690 (2009)
Acknowledgements
We thank our research subjects for participating. We thank S. Solomon and K. Eggan for discussions and support, L. Bauer for help with blastocyst thawing, D. Kahler for cell sorting, M. Verbitsky and S. Kisselev for microarray hybridization, V. Miljkovic for Affymetrix SNP chip hybridization, C. LeDuc and Y. Ravussin for help with data analysis and mouse work, R. Maehr for DiPS H.1.5, C. Marshall and J. Safran for administrative support, S. Paull for cover art, Z. Hall and S. Chang for critical reading of the manuscript. This research was supported by a UCSD startup fund to K.Z., the New York Stem Cell Foundation primarily, and the Russell Berrie Foundation.
Author information
Authors and Affiliations
Contributions
R.G. and M.V.S. wrote IRB and consent documents, M.V.S., K.C.S., K.O. and R.P. consented oocyte donors and retrieved oocytes, D.E. and S.N. designed and performed experiments with oocytes, D.E., H.-L.F., A.G., H.M., D.P. and K.Z. characterized stem cell lines, M.F., E.G. and M.V.S. performed skin biopsies, D.E. performed skin cell isolation, soPS and iPS derivation, S.D. performed NYSCF1 derivation, D.E. and R.L.L. wrote the paper with input from all authors. All work with human oocytes and stem cells was performed at the NYSCF laboratory.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-10 with Legends, Supplementary Karyotypes and Fingerprints, Supplementary Tables 1-7, a Supplementary Discussion and Supplementary References. (PDF 19560 kb)
Rights and permissions
About this article
Cite this article
Noggle, S., Fung, HL., Gore, A. et al. Human oocytes reprogram somatic cells to a pluripotent state. Nature 478, 70–75 (2011). https://doi.org/10.1038/nature10397
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10397
This article is cited by
-
Using intracellular markers to identify a novel set of surface markers for live cell purification from a heterogeneous hIPSC culture
Scientific Reports (2018)
-
Compensating human subjects providing oocytes for stem cell research: 9-year experience and outcomes
Journal of Assisted Reproduction and Genetics (2018)
-
Genomic instability during reprogramming by nuclear transfer is DNA replication dependent
Nature Cell Biology (2017)
-
Derivation of Human Induced Pluripotent Stem Cell (iPSC) Lines and Mechanism of Pluripotency: Historical Perspective and Recent Advances
Stem Cell Reviews and Reports (2017)
-
Cellular reprogramming in farm animals: an overview of iPSC generation in the mammalian farm animal species
Journal of Animal Science and Biotechnology (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.