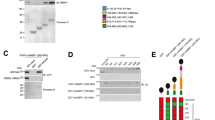
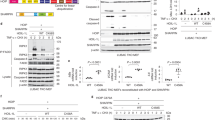
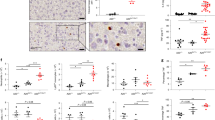
Abstract
Members of the tumour necrosis factor (TNF) receptor superfamily have important functions in immunity and inflammation. Recently linear ubiquitin chains assembled by a complex containing HOIL-1 and HOIP (also known as RBCK1 and RNF31, respectively) were implicated in TNF signalling, yet their relevance in vivo remained uncertain. Here we identify SHARPIN as a third component of the linear ubiquitin chain assembly complex, recruited to the CD40 and TNF receptor signalling complexes together with its other constituents, HOIL-1 and HOIP. Mass spectrometry of TNF signalling complexes revealed RIP1 (also known as RIPK1) and NEMO (also known as IKKγ or IKBKG) to be linearly ubiquitinated. Mutation of the Sharpin gene (Sharpincpdm/cpdm) causes chronic proliferative dermatitis (cpdm) characterized by inflammatory skin lesions and defective lymphoid organogenesis. Gene induction by TNF, CD40 ligand and interleukin-1β was attenuated in cpdm-derived cells which were rendered sensitive to TNF-induced death. Importantly, Tnf gene deficiency prevented skin lesions in cpdm mice. We conclude that by enabling linear ubiquitination in the TNF receptor signalling complex, SHARPIN interferes with TNF-induced cell death and, thereby, prevents inflammation. Our results provide evidence for the relevance of linear ubiquitination in vivo in preventing inflammation and regulating immune signalling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ware, C. F. The TNF superfamily. Cytokine Growth Factor Rev. 14, 181–184 (2003)
Bianchi, K. & Meier, P. A tangled web of ubiquitin chains: breaking news in TNF-R1 signaling. Mol. Cell 36, 736–742 (2009)
Silke, J. & Brink, R. Regulation of TNFRSF and innate immune signalling complexes by TRAFs and cIAPs. Cell Death Differ. 17, 35–45 (2010)
Wertz, I. E. & Dixit, V. M. Ubiquitin-mediated regulation of TNFR1 signaling. Cytokine Growth Factor Rev. 19, 313–324 (2008)
Haas, T. L. et al. Recruitment of the linear ubiquitin chain assembly complex stabilizes the TNF-R1 signaling complex and is required for TNF-mediated gene induction. Mol. Cell 36, 831–844 (2009)
Tokunaga, F. et al. Involvement of linear polyubiquitylation of NEMO in NF-κB activation. Nature Cell Biol. 11, 123–132 (2009)
Rahighi, S. et al. Specific recognition of linear ubiquitin chains by NEMO is important for NF-κB activation. Cell 136, 1098–1109 (2009)
Lo, Y. C. et al. Structural basis for recognition of diubiquitins by NEMO. Mol. Cell 33, 602–615 (2009)
Komander, D. et al. Molecular discrimination of structurally equivalent Lys 63-linked and linear polyubiquitin chains. EMBO Rep. 10, 466–473 (2009)
Xu, M., Skaug, B., Zeng, W. & Chen, Z. J. A ubiquitin replacement strategy in human cells reveals distinct mechanisms of IKK activation by TNFα and IL-1β. Mol. Cell 36, 302–314 (2009)
Cossu, F. et al. Structural basis for bivalent Smac-mimetics recognition in the IAP protein family. J. Mol. Biol. 392, 630–644 (2009)
Seymour, R. E. et al. Spontaneous mutations in the mouse Sharpin gene result in multiorgan inflammation, immune system dysregulation and dermatitis. Genes Immun. 8, 416–421 (2007)
HogenEsch, H. et al. A spontaneous mutation characterized by chronic proliferative dermatitis in C57BL mice. Am. J. Pathol. 143, 972–982 (1993)
HogenEsch, H., Janke, S., Boggess, D. & Sundberg, J. P. Absence of Peyer’s patches and abnormal lymphoid architecture in chronic proliferative dermatitis (cpdm/cpdm) mice. J. Immunol. 162, 3890–3896 (1999)
HogenEsch, H. et al. Increased expression of type 2 cytokines in chronic proliferative dermatitis (cpdm) mutant mice and resolution of inflammation following treatment with IL-12. Eur. J. Immunol. 31, 734–742 (2001)
Tay, S. et al. Single-cell NF-κB dynamics reveal digital activation and analogue information processing. Nature 466, 267–271 (2010)
Senftleben, U. et al. Activation by IKKα of a second, evolutionary conserved, NF-κB signaling pathway. Science 293, 1495–1499 (2001)
Gijbels, M. J., HogenEsch, H., Bruijnzeel, P. L., Elliott, G. R. & Zurcher, C. Maintenance of donor phenotype after full-thickness skin transplantation from mice with chronic proliferative dermatitis (cpdm/cpdm) to C57BL/Ka and nude mice and vice versa. J. Invest. Dermatol. 105, 769–773 (1995)
Liang, Y., Seymour, R. E. & Sundberg, J. P. Inhibition of NF-κB signaling retards eosinophilic dermatitis in SHARPIN-deficient mice. J. Invest. Dermatol. 131, 141–149 (2011)
Martin, M. U. & Wesche, H. Summary and comparison of the signaling mechanisms of the Toll/interleukin-1 receptor family. Biochim. Biophys. Acta 1592, 265–280 (2002)
Arend, W. P., Palmer, G. & Gabay, C. IL-1, IL-18, and IL-33 families of cytokines. Immunol. Rev. 223, 20–38 (2008)
Huang, H. et al. K33-linked polyubiquitination of T cell receptor-ζ regulates proteolysis-independent T cell signaling. Immunity 33, 60–70 (2010)
Matsumoto, M. L. et al. K11-linked polyubiquitination in cell cycle control revealed by a K11 linkage-specific antibody. Mol. Cell 39, 477–484 (2010)
Arimoto, K. et al. Polyubiquitin conjugation to NEMO by triparite motif protein 23 (TRIM23) is critical in antiviral defense. Proc. Natl Acad. Sci. USA 107, 15856–15861 (2010)
Ikeda, F., Crosetto, N. & Dikic, I. What determines the specificity and outcomes of ubiquitin signaling? Cell 143, 677–681 (2010)
Liu, S. & Chen, Z. J. Expanding role of ubiquitination in NF-κB signaling. Cell Res. 21, 6–21 (2011)
Laplantine, E. et al. NEMO specifically recognizes K63-linked poly-ubiquitin chains through a new bipartite ubiquitin-binding domain. EMBO J. 28, 2885–2895 (2009)
Dynek, J. N. et al. c-IAP1 and UbcH5 promote K11-linked polyubiquitination of RIP1 in TNF signalling. EMBO J. 29, 4198–4209 (2010)
Taylor, P. C. & Feldmann, M. Anti-TNF biologic agents: still the therapy of choice for rheumatoid arthritis. Nature Rev. Rheumatol. 5, 578–582 (2009)
Sacre, S. M., Andreakos, E., Taylor, P., Feldmann, M. & Foxwell, B. M. Molecular therapeutic targets in rheumatoid arthritis. Expert Rev. Mol. Med. 7, 1–20 (2005)
Mantovani, A., Allavena, P., Sica, A. & Balkwill, F. Cancer-related inflammation. Nature 454, 436–444 (2008)
Pikarsky, E. et al. NF-κB functions as a tumour promoter in inflammation-associated cancer. Nature 431, 461–466 (2004)
Grivennikov, S. I., Greten, F. R. & Karin, M. Immunity, inflammation, and cancer. Cell 140, 883–899 (2010)
Silke, J. et al. Determination of cell survival by RING-mediated regulation of inhibitor of apoptosis (IAP) protein abundance. Proc. Natl Acad. Sci. USA 102, 16182–16187 (2005)
Körner, H. et al. Distinct roles for lymphotoxin-α and tumor necrosis factor in organogenesis and spatial organization of lymphoid tissue. Eur. J. Immunol. 27, 2600–2609 (1997)
Diessenbacher, P. et al. NF-κB inhibition reveals differential mechanisms of TNF versus TRAIL-induced apoptosis upstream or at the level of caspase-8 activation independent of cIAP2. J. Invest. Dermatol. 128, 1134–1147 (2008)
Moolenbeek, C. & Ruitenberg, E. J. The ‘Swiss roll’: a simple technique for histological studies of the rodent intestine. Lab. Anim. 15, 57–59 (1981)
Acknowledgements
We thank M. Miasari for developing the cIAP2 antibody, M. Leverkus for advice on skin histology, J. Lokan for advice on liver histology, C. Rappl, N. Barboza, S. Kupka, D. Heinze, J. Zipprich, J. Corbin, S. Wiegelmann and A. Bankovacki for excellent technical assistance, M. Bolognesi and P. Seneci for SM-164, H. Koerner for Tnf−/− mice, and all past and present members of the Walczak and Silke laboratories for continuous stimulating scientific discussions and support. Research in the Walczak lab is supported by grants from Cancer Research UK, AICR, BBSRC (ERASysBio PLUS), Ovarian Cancer Action and the EU Marie Curie Research Training Network ApopTRAIN. Work in the Silke lab is supported by NHMRC grants 541901, 541902 and 602516. Work in the Purcell lab is supported by the NHMRC (Senior Research Fellowships for A.W.P. and a C. J. Martin Overseas Biomedical Fellowship to A.I.W.), and by grants from the NHMRC and ARC. U.W. is funded by HGF/SBCancer. E.R. and T.L.H. are ApopTRAIN fellows and U.N. is supported by the Schweizer Nationalfonds (SNF).
Author information
Authors and Affiliations
Contributions
T.L.H. and H.W. conceived the moTAP procedure. B.G., E.R., H.W. and U.W., and T.L.H. and U.W. determined the composition of the CD40- and TNF-RSC, respectively. B.G. performed all other experiments involving CD40L. A.C.S. and C.H.E. cloned and purified all recombinant proteins, determined the molecular interaction between the LUBAC components and performed the in vitro ubiquitination assays. B.G., S.M.C., A.C.S., C.H.E. and E.R. provided moTAP-purified TNF-RSCs for 2D-MRM analysis which was conceived by A.I.W. and H.W., performed by A.I.W., and analysed by A.I.W. and A.W.P.; J.S. and H.W. planned and J.A.R., H.A., U.N., W.W.-L.W., L.G., B.G., S.M.C. and E.R. performed the analyses of cells, tissues and blood samples obtained from all mouse strains used in this study. H.W. and J.S. wrote the manuscript assisted by B.G., S.M.C., A.C.S., C.H.E., E.R. and A.I.W.
Corresponding author
Ethics declarations
Competing interests
H.W. is founder, shareholder and scientific advisor of Apogenix GmbH, Heidelberg, Germany. J.S. is a consultant for TetraLogic Pharmaceuticals, Malvern, Pennsylvania, USA.
Supplementary information
Supplementary Figures
The file contains Supplementary Figures 1-22 with legends. (PDF 4057 kb)
Rights and permissions
About this article
Cite this article
Gerlach, B., Cordier, S., Schmukle, A. et al. Linear ubiquitination prevents inflammation and regulates immune signalling. Nature 471, 591–596 (2011). https://doi.org/10.1038/nature09816
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature09816
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.