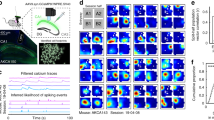
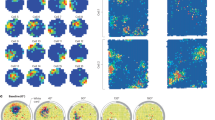
Abstract
During spatial exploration, hippocampal neurons show a sequential firing pattern in which individual neurons fire specifically at particular locations along the animal’s trajectory (place cells1,2). According to the dominant model of hippocampal cell assembly activity, place cell firing order is established for the first time during exploration, to encode the spatial experience, and is subsequently replayed during rest3,4,5,6 or slow-wave sleep7,8,9,10 for consolidation of the encoded experience11,12. Here we report that temporal sequences of firing of place cells expressed during a novel spatial experience occurred on a significant number of occasions during the resting or sleeping period preceding the experience. This phenomenon, which is called preplay, occurred in disjunction with sequences of replay of a familiar experience. These results suggest that internal neuronal dynamics during resting or sleep organize hippocampal cellular assemblies13,14,15 into temporal sequences that contribute to the encoding of a related novel experience occurring in the future.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
O’Keefe, J. & Nadel, L. The Hippocampus as a Cognitive Map (Oxford Univ. Press, 1978)
Wilson, M. A. & McNaughton, B. L. Dynamics of the hippocampal ensemble code for space. Science 261, 1055–1058 (1993)
Foster, D. J. & Wilson, M. A. Reverse replay of behavioural sequences in hippocampal place cells during the awake state. Nature 440, 680–683 (2006)
Diba, K. & Buzsaki, G. Forward and reverse hippocampal place-cell sequences during ripples. Nature Neurosci. 10, 1241–1242 (2007)
Karlsson, M. P. & Frank, L. M. Awake replay of remote experiences in the hippocampus. Nature Neurosci. 12, 913–918 (2009)
Davidson, T. J., Kloosterman, F. & Wilson, M. A. Hippocampal replay of extended experience. Neuron 63, 497–507 (2009)
Wilson, M. A. & McNaughton, B. L. Reactivation of hippocampal ensemble memories during sleep. Science 265, 676–679 (1994)
Skaggs, W. E. & McNaughton, B. L. Replay of neuronal firing sequences in rat hippocampus during sleep following spatial experience. Science 271, 1870–1873 (1996)
Nádasdy, Z., Hirase, H., Czurko, A., Csicsvari, J. & Buzsaki, G. Replay and time compression of recurring spike sequences in the hippocampus. J. Neurosci. 19, 9497–9507 (1999)
Lee, A. K. & Wilson, M. A. Memory of sequential experience in the hippocampus during slow wave sleep. Neuron 36, 1183–1194 (2002)
Buzsáki, G. Two-stage model of memory trace formation: a role for “noisy” brain states. Neuroscience 31, 551–570 (1989)
Nakashiba, T., Buhl, D. L., McHugh, T. J. & Tonegawa, S. Hippocampal CA3 output is crucial for ripple-associated reactivation and consolidation of memory. Neuron 62, 781–787 (2009)
Hebb, D. O. The Organization of Behavior: A Neuropsychological Theory (Wiley, 1949)
Harris, K. D., Csicsvari, J., Hirase, H., Dragoi, G. & Buzsaki, G. Organization of cell assemblies in the hippocampus. Nature 424, 552–556 (2003)
Dragoi, G. & Buzsaki, G. Temporal encoding of place sequences by hippocampal cell assemblies. Neuron 50, 145–157 (2006)
Thompson, L. T. & Best, P. J. Place cells and silent cells in the hippocampus of freely-behaving rats. J. Neurosci. 9, 2382–2390 (1989)
O’Neill, J., Senior, T. & Csicsvari, J. Place-selective firing of CA1 pyramidal cells during sharp wave/ripple network patterns in exploratory behavior. Neuron 49, 143–155 (2006)
Zhang, K., Ginzburg, I., McNaughton, B. L. & Sejnowski, T. J. Interpreting neuronal population activity by reconstruction: unified framework with application to hippocampal place cells. J. Neurophysiol. 79, 1017–1044 (1998)
Johnson, A. & Redish, A. D. Neural ensembles in CA3 transiently encode paths forward of the animal at a decision point. J. Neurosci. 27, 12176–12189 (2007)
Kudrimoti, H. S., Barnes, C. A. & McNaughton, B. L. Reactivation of hippocampal cell assemblies: effects of behavioral state, experience, and EEG dynamics. J. Neurosci. 19, 4090–4101 (1999)
Csicsvari, J., Hirase, H., Czurko, A., Mamiya, A. & Buzsaki, G. Oscillatory coupling of hippocampal pyramidal cells and interneurons in the behaving rat. J. Neurosci. 19, 274–287 (1999)
Dragoi, G., Harris, K. D. & Buzsaki, G. Place representation within hippocampal networks is modified by long-term potentiation. Neuron 39, 843–853 (2003)
Pastalkova, E., Itskov, V., Amarasingham, A. & Buzsaki, G. Internally generated cell assembly sequences in the rat hippocampus. Science 321, 1322–1327 (2008)
Black, J. E. & Greenough, W. T. Advances in Developmental Psychology (Lawrence Erlbaum, 1986)
Hassabis, D., Kumaran, D., Vann, S. D. & Maguire, E. A. Patients with hippocampal amnesia cannot imagine new experiences. Proc. Natl Acad. Sci. USA 104, 1726–1731 (2007)
Tse, D. et al. Schemas and memory consolidation. Science 316, 76–82 (2007)
Tsien, J. Z., Huerta, P. T. & Tonegawa, S. The essential role of hippocampal CA1 NMDA receptor-dependent synaptic plasticity in spatial memory. Cell 87, 1327–1338 (1996)
Schmitzer-Torbert, N., Jackson, J., Henze, D., Harris, K. & Redish, A. D. Quantitative measures of cluster quality for use in extracellular recordings. Neuroscience 131, 1–11 (2005)
Acknowledgements
We thank M. A. Wilson for assistance with data acquisition, discussions and comments on an earlier version of the manuscript; J. O’Keefe, A. Siapas, F. Kloosterman, D. L. Buhl for comments on earlier versions of the manuscript; and F. Kloosterman for providing assistance with the line detection for the Bayesian decoding. This work was supported by NIH grants R01-MH078821 and P50-MH58880 to S.T., who was an HHMI Investigator in an earlier part of this study.
Author information
Authors and Affiliations
Contributions
S.T. and G.D. conceived the project jointly. G.D. designed and performed the experiments and the analyses. G.D. and S.T. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
The file contains Supplementary Text, a Supplementary Table, additional references and Supplementary Figures 1-8 with legends. (PDF 3672 kb)
Rights and permissions
About this article
Cite this article
Dragoi, G., Tonegawa, S. Preplay of future place cell sequences by hippocampal cellular assemblies. Nature 469, 397–401 (2011). https://doi.org/10.1038/nature09633
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature09633
This article is cited by
-
Minute-scale oscillatory sequences in medial entorhinal cortex
Nature (2024)
-
The generative grammar of the brain: a critique of internally generated representations
Nature Reviews Neuroscience (2024)
-
Backbone spiking sequence as a basis for preplay, replay, and default states in human cortex
Nature Communications (2023)
-
Robust encoding of natural stimuli by neuronal response sequences in monkey visual cortex
Nature Communications (2023)
-
Differential replay of reward and punishment paths predicts approach and avoidance
Nature Neuroscience (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.