Abstract
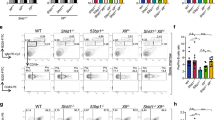
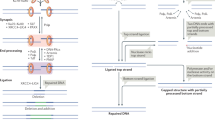
Classical non-homologous DNA end-joining (NHEJ) is a major mammalian DNA double-strand-break (DSB) repair pathway. Deficiencies for classical NHEJ factors, such as XRCC4, abrogate lymphocyte development, owing to a strict requirement for classical NHEJ to join V(D)J recombination DSB intermediates1,2. The XRCC4-like factor (XLF; also called NHEJ1) is mutated in certain immunodeficient human patients and has been implicated in classical NHEJ3,4,5,6; however, XLF-deficient mice have relatively normal lymphocyte development and their lymphocytes support normal V(D)J recombination5. The ataxia telangiectasia-mutated protein (ATM) detects DSBs and activates DSB responses by phosphorylating substrates including histone H2AX7. However, ATM deficiency causes only modest V(D)J recombination and lymphocyte developmental defects, and H2AX deficiency does not have a measurable impact on these processes7,8,9. Here we show that XLF, ATM and H2AX all have fundamental roles in processing and joining DNA ends during V(D)J recombination, but that these roles have been masked by unanticipated functional redundancies. Thus, combined deficiency of ATM and XLF nearly blocks mouse lymphocyte development due to an inability to process and join chromosomal V(D)J recombination DSB intermediates. Combined XLF and ATM deficiency also severely impairs classical NHEJ, but not alternative end-joining, during IgH class switch recombination. Redundant ATM and XLF functions in classical NHEJ are mediated by ATM kinase activity and are not required for extra-chromosomal V(D)J recombination, indicating a role for chromatin-associated ATM substrates. Correspondingly, conditional H2AX inactivation in XLF-deficient pro-B lines leads to V(D)J recombination defects associated with marked degradation of unjoined V(D)J ends, revealing that H2AX has a role in this process.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lieber, M. R. The mechanism of human nonhomologous DNA end joining. J. Biol. Chem. 283, 1–5 (2008)
Rooney, S., Chaudhuri, J. & Alt, F. W. The role of the non-homologous end-joining pathway in lymphocyte development. Immunol. Rev. 200, 115–131 (2004)
Ahnesorg, P., Smith, P. & Jackson, S. P. XLF interacts with the XRCC4-DNA ligase IV complex to promote DNA nonhomologous end-joining. Cell 124, 301–313 (2006)
Buck, D. et al. Cernunnos, a novel nonhomologous end-joining factor, is mutated in human immunodeficiency with microcephaly. Cell 124, 287–299 (2006)
Li, G. et al. Lymphocyte-specific compensation for XLF/Cernunnos end-joining functions in V(D)J recombination. Mol. Cell 31, 631–640 (2008)
Zha, S., Alt, F. W., Cheng, H. L., Brush, J. W. & Li, G. Defective DNA repair and increased genomic instability in Cernunnos-XLF-deficient murine ES cells. Proc. Natl Acad. Sci. USA 104, 4518–4523 (2007)
Bassing, C. H. & Alt, F. W. The cellular response to general and programmed DNA double strand breaks. DNA Repair (Amst.) 3, 781–796 (2004)
Bredemeyer, A. L. et al. ATM stabilizes DNA double-strand-break complexes during V(D)J recombination. Nature 442, 466–470 (2006)
Yin, B. et al. Histone H2AX stabilizes broken DNA strands to suppress chromosome breaks and translocations during V(D)J recombination. J. Exp. Med. 206, 2625–2639 (2009)
Bassing, C. H., Swat, W. & Alt, F. W. The mechanism and regulation of chromosomal V(D)J recombination. Cell 109 (Suppl.). S45–S55 (2002)
Bredemeyer, A. L. et al. DNA double-strand breaks activate a multi-functional genetic program in developing lymphocytes. Nature 456, 819–823 (2008)
Zha, S., Sekiguchi, J., Brush, J. W., Bassing, C. H. & Alt, F. W. Complementary functions of ATM and H2AX in development and suppression of genomic instability. Proc. Natl Acad. Sci. USA 105, 9302–9306 (2008)
Yan, C. T. et al. IgH class switching and translocations use a robust non-classical end-joining pathway. Nature 449, 478–482 (2007)
Gellert, M. Molecular analysis of V(D)J recombination. Annu. Rev. Genet. 26, 425–446 (1992)
Corneo, B. et al. Rag mutations reveal robust alternative end joining. Nature 449, 483–486 (2007)
Chaudhuri, J. et al. Evolution of the immunoglobulin heavy chain class switch recombination mechanism. Adv. Immunol. 94, 157–214 (2007)
Franco, S. et al. H2AX prevents DNA breaks from progressing to chromosome breaks and translocations. Mol. Cell 21, 201–214 (2006)
Ramiro, A. R. et al. Role of genomic instability and p53 in AID-induced c-myc-Igh translocations. Nature 440, 105–109 (2006)
Lumsden, J. M. et al. Immunoglobulin class switch recombination is impaired in Atm-deficient mice. J. Exp. Med. 200, 1111–1121 (2004)
Reina-San-Martin, B., Chen, H. T., Nussenzweig, A. & Nussenzweig, M. C. ATM is required for efficient recombination between immunoglobulin switch regions. J. Exp. Med. 200, 1103–1110 (2004)
Bassing, C. H. et al. Increased ionizing radiation sensitivity and genomic instability in the absence of histone H2AX. Proc. Natl Acad. Sci. USA 99, 8173–8178 (2002)
Bassing, C. H. et al. Histone H2AX: a dosage-dependent suppressor of oncogenic translocations and tumors. Cell 114, 359–370 (2003)
Sekiguchi, J. et al. Genetic interactions between ATM and the nonhomologous end-joining factors in genomic stability and development. Proc. Natl Acad. Sci. USA 98, 3243–3248 (2001)
Helmink, B. A. et al. H2AX prevents CtIP-mediated DNA end resection and aberrant repair in G1-phase lymphocytes. Nature doi:10.1038/nature09585 (this issue).
Callén, E. et al. Essential role for DNA-PKcs in DNA double-strand break repair and apoptosis in ATM-deficient lymphocytes. Mol. Cell 34, 285–297 (2009)
Yano, K. et al. Ku recruits XLF to DNA double-strand breaks. EMBO Rep. 9, 91–96 (2008)
Celeste, A. et al. Genomic instability in mice lacking histone H2AX. Science 296, 922–927 (2002)
Acknowledgements
We thank Y. Fujiwara and P. Huang for technical support. We thank B. Sleckman for advice, reagents and for critical review of this manuscript. This work is supported by NIH grant AI076210 and AI020047 to F.W.A. F.W.A. is an investigator of the Howard Hughes Medical Institute. S.Z. was a fellow, then senior fellow of Leukemia and Lymphomas Society of America and a St Baldrick Scholar. C.G. and Y.Z. are fellows of Cancer Research Institute. C.B. receives support from the pre-doctoral training program of Cancer Research Institute. D.R.W. is supported by a career development award from AAAI/GlaxoSmithKline and by NIH training grant AI007376.
Author information
Authors and Affiliations
Contributions
S.Z., C.G. and F.W.A. designed experiments and wrote the paper. S.Z., C.G., C.B., V.O., H.-L.C., Y.Z., D.R.W., G.Y., H.P., P.H.G. and R.L.D. performed experiments.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1- 8 with legends and Supplementary Tables 1-2. (PDF 2318 kb)
Rights and permissions
About this article
Cite this article
Zha, S., Guo, C., Boboila, C. et al. ATM damage response and XLF repair factor are functionally redundant in joining DNA breaks. Nature 469, 250–254 (2011). https://doi.org/10.1038/nature09604
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature09604
This article is cited by
-
Autopolyploidization affects transcript patterns and gene targeting frequencies in Physcomitrella
Plant Cell Reports (2022)
-
ATM, ATR and DNA-PKcs kinases—the lessons from the mouse models: inhibition ≠ deletion
Cell & Bioscience (2020)
-
ERCC6L2 promotes DNA orientation-specific recombination in mammalian cells
Cell Research (2020)
-
Endogenous topoisomerase II-mediated DNA breaks drive thymic cancer predisposition linked to ATM deficiency
Nature Communications (2020)
-
ATM, DNA-PKcs and ATR: shaping development through the regulation of the DNA damage responses
Genome Instability & Disease (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.