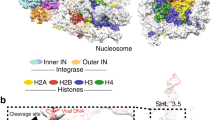
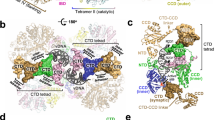
Abstract
To establish productive infection, a retrovirus must insert a DNA replica of its genome into host cell chromosomal DNA1,2. This process is operated by the intasome, a nucleoprotein complex composed of an integrase tetramer (IN) assembled on the viral DNA ends3,4. The intasome engages chromosomal DNA within a target capture complex to carry out strand transfer, irreversibly joining the viral and cellular DNA molecules. Although several intasome/transpososome structures from the DDE(D) recombinase superfamily have been reported4,5,6, the mechanics of target DNA capture and strand transfer by these enzymes remained unclear. Here we report crystal structures of the intasome from prototype foamy virus in complex with target DNA, elucidating the pre-integration target DNA capture and post-catalytic strand transfer intermediates of the retroviral integration process. The cleft between IN dimers within the intasome accommodates chromosomal DNA in a severely bent conformation, allowing widely spaced IN active sites to access the scissile phosphodiester bonds. Our results resolve the structural basis for retroviral DNA integration and provide a framework for the design of INs with altered target sequences.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
References
Craigie, R. in Mobile DNA II (eds Craig, N. L., Craigie, R., Gellert, M. & Lambowitz, A. M) 613–630 (ASM Press, 2002)
Lewinski, M. K. & Bushman, F. D. Retroviral DNA integration--mechanism and consequences. Adv. Genet. 55, 147–181 (2005)
Li, M., Mizuuchi, M., Burke, T. R., Jr & Craigie, R. Retroviral DNA integration: reaction pathway and critical intermediates. EMBO J. 25, 1295–1304 (2006)
Hare, S., Gupta, S. S., Valkov, E., Engelman, A. & Cherepanov, P. Retroviral intasome assembly and inhibition of DNA strand transfer. Nature 464, 232–236 (2010)
Davies, D. R., Goryshin, I. Y., Reznikoff, W. S. & Rayment, I. Three-dimensional structure of the Tn5 synaptic complex transposition intermediate. Science 289, 77–85 (2000)
Richardson, J. M., Colloms, S. D., Finnegan, D. J. & Walkinshaw, M. D. Molecular architecture of the Mos1 paired-end complex: the structural basis of DNA transposition in a eukaryote. Cell 138, 1096–1108 (2009)
Trobridge, G. D. et al. Foamy virus vector integration sites in normal human cells. Proc. Natl Acad. Sci. USA 103, 1498–1503 (2006)
Valkov, E. et al. Functional and structural characterization of the integrase from the prototype foamy virus. Nucleic Acids Res. 37, 243–255 (2009)
Berry, C., Hannenhalli, S., Leipzig, J. & Bushman, F. D. Selection of target sites for mobile DNA integration in the human genome. PLOS Comput. Biol. 2, e157 (2006)
Holman, A. G. & Coffin, J. M. Symmetrical base preferences surrounding HIV-1, avian sarcoma/leukosis virus, and murine leukemia virus integration sites. Proc. Natl Acad. Sci. USA 102, 6103–6107 (2005)
Wu, X., Li, Y., Crise, B., Burgess, S. M. & Munroe, D. J. Weak palindromic consensus sequences are a common feature found at the integration target sites of many retroviruses. J. Virol. 79, 5211–5214 (2005)
Johnson, R. C., Stella, S. & Heiss, J. K. in Protein-nucleic Acid Interactions (eds Rice, P. A. & Correll, C. C.) Ch. 8, 176–220 (RSC Publishing, 2008)
Luscombe, N. M., Laskowski, R. A. & Thornton, J. M. Amino acid-base interactions: a three-dimensional analysis of protein-DNA interactions at an atomic level. Nucleic Acids Res. 29, 2860–2874 (2001)
Konsavage, W. M., Jr, Burkholder, S., Sudol, M., Harper, A. L. & Katzman, M. A substitution in Rous sarcoma virus integrase that separates its two biologically relevant enzymatic activities. J. Virol. 79, 4691–4699 (2005)
Harper, A. L., Sudol, M. & Katzman, M. An amino acid in the central catalytic domain of three retroviral integrases that affects target site selection in nonviral DNA. J. Virol. 77, 3838–3845 (2003)
Müller, H. P. & Varmus, H. E. DNA bending creates favored sites for retroviral integration: an explanation for preferred insertion sites in nucleosomes. EMBO J. 13, 4704–4714 (1994)
Pruss, D., Bushman, F. D. & Wolffe, A. P. Human immunodeficiency virus integrase directs integration to sites of severe DNA distortion within the nucleosome core. Proc. Natl Acad. Sci. USA 91, 5913–5917 (1994)
Lim, K. I., Klimczak, R., Yu, J. H. & Schaffer, D. V. Specific insertions of zinc finger domains into Gag-Pol yield engineered retroviral vectors with selective integration properties. Proc. Natl Acad. Sci. USA 107, 12475–12480 (2010)
Lombardo, A. et al. Gene editing in human stem cells using zinc finger nucleases and integrase-defective lentiviral vector delivery. Nature Biotechnol. 25, 1298–1306 (2007)
Krishnan, L. et al. Structure-based modeling of the functional HIV-1 intasome and its inhibition. Proc. Natl Acad. Sci. USA 107, 15910–15915 (2010)
Kabsch, W. XDS. Acta Crystallogr. D 66, 125–132 (2010)
CCP4. The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994)
Murshudov, G. N., Vagin, A. A. & Dodson, E. J. Refinement of macromolecular structures by the maximum-likelihood method. Acta Crystallogr. D 53, 240–255 (1997)
Davis, I. W. et al. MolProbity: all-atom contacts and structure validation for proteins and nucleic acids. Nucleic Acids Res. 35, W375–W383 (2007)
Cherepanov, P. P. & Wackernagel, W. Gene disruption in Escherichia coli: TcR and KmR cassettes with the option of Flp-catalyzed excision of the antibiotic-resistance determinant. Gene 158, 9–14 (1995)
Crooks, G. E., Hon, G., Chandonia, J. M. & Brenner, S. E. WebLogo: a sequence logo generator. Genome Res. 14, 1188–1190 (2004)
Acknowledgements
We thank A. Engelman and F. Dyda for critical reading of the manuscript, J. Sanchez-Weatherby for assistance with X-ray data collection at the I02 beamline of the Diamond Light Source and J. Moore for help with crystallization screening and the X-ray generator. This work was funded by the UK Medical Research Council.
Author information
Authors and Affiliations
Contributions
G.N.M., S.H. and P.C. performed the experiments and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
The file contains Supplementary Table 1 and Supplementary Figures 1-8 with legends. (PDF 1224 kb)
Supplementary Movie 1
This movie shows the overall architecture of the PFV STC. Protein and DNA chains are coloured as in Fig. 1b and c. (MOV 8200 kb)
Supplementary Movie 2
This movie illustrates tDNA bending that transpires when the intasome engages hypothetical initial B-form tDNA to form the TCC and the subsequent DNA strand transfer. For clarity, only DNA strands are shown. (MOV 3119 kb)
Rights and permissions
About this article
Cite this article
Maertens, G., Hare, S. & Cherepanov, P. The mechanism of retroviral integration from X-ray structures of its key intermediates. Nature 468, 326–329 (2010). https://doi.org/10.1038/nature09517
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature09517
This article is cited by
-
Structure reveals why genome folding is necessary for site-specific integration of foreign DNA into CRISPR arrays
Nature Structural & Molecular Biology (2023)
-
DNA strand breaks and gaps target retroviral intasome binding and integration
Nature Communications (2023)
-
Structure and function of retroviral integrase
Nature Reviews Microbiology (2022)
-
The KT Jeang Retrovirology prize 2021: Peter Cherepanov
Retrovirology (2021)
-
Influence of the amino-terminal sequence on the structure and function of HIV integrase
Retrovirology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.