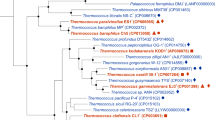
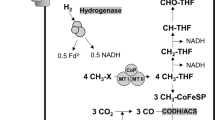
Abstract
Although a common reaction in anaerobic environments, the conversion of formate and water to bicarbonate and H2 (with a change in Gibbs free energy of ΔG° = +1.3 kJ mol−1) has not been considered energetic enough to support growth of microorganisms. Recently, experimental evidence for growth on formate was reported for syntrophic communities of Moorella sp. strain AMP and a hydrogen-consuming Methanothermobacter species and of Desulfovibrio sp. strain G11 and Methanobrevibacter arboriphilus strain AZ1. The basis of the sustainable growth of the formate-users is explained by H2 consumption by the methanogens, which lowers the H2 partial pressure, thus making the pathway exergonic2. However, it has not been shown that a single strain can grow on formate by catalysing its conversion to bicarbonate and H2. Here we report that several hyperthermophilic archaea belonging to the Thermococcus genus are capable of formate-oxidizing, H2-producing growth. The actual ΔG values for the formate metabolism are calculated to range between −8 and −20 kJ mol−1 under the physiological conditions where Thermococcus onnurineus strain NA1 are grown. Furthermore, we detected ATP synthesis in the presence of formate as a sole energy source. Gene expression profiling and disruption identified the gene cluster encoding formate hydrogen lyase, cation/proton antiporter and formate transporter, which were responsible for the growth of T. onnurineus NA1 on formate. This work shows formate-driven growth by a single microorganism with protons as the electron acceptor, and reports the biochemical basis of this ability.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Dolfing, J., Jiang, B., Henstra, A. M., Stams, A. J. M. & Plugge, C. M. Syntrophic growth on formate: a new microbial niche in anoxic environments. Appl. Environ. Microbiol. 74, 6126–6131 (2008)
Stams, A. J. M. & Plugge, C. M. Electron transfer in syntrophic communities of anaerobic bacteria and archaea. Nature Rev. Microbiol. 7, 568–577 (2009)
Andrews, S. C. et al. A 12-cistron Escherichia coli operon (hyf) encoding a putative proton-translocating formate hydrogenlyase system. Microbiology 143, 3633–3647 (1997)
Böhm, R., Sauter, M. & Böck, A. Nucleotide sequence and expression of an operon in Escherichia coli coding for formate hydrogenlyase components. Mol. Microbiol. 4, 231–243 (1990)
Sauter, M., Bohm, R. & Böck, A. Mutational analysis of the operon (hyc) determining hydrogenase 3 formation in Escherichia coli. Mol. Microbiol. 6, 1523–1532 (1992)
Bagramyan, K., Mnatsakanyan, N., Poladian, A., Vassilian, A. & Trchounian, A. The roles of hydrogenases 3 and 4, and the F0F1-ATPase, in H2 production by Escherichia coli at alkaline and acidic pH. FEBS Lett. 516, 172–178 (2002)
Takács, M. et al. Formate hydrogenlyase in the hyperthermophilic archaeon, Thermococcus litoralis. BMC Microbiol. 8, 88 (2008)
Schauer, N. L. & Ferry, J. G. Metabolism of formate in Methanobacterium formicicum. J. Bacteriol. 142, 800–807 (1980)
Baron, S. F. & Ferry, J. G. Reconstitution and properties of a coenzyme F420-mediated formate hydrogenlyase system in Methanobacterium formicicum. J. Bacteriol. 171, 3854–3859 (1989)
Wood, G. E., Haydock, A. K. & Leigh, J. A. Function and regulation of the formate dehydrogenase genes of the methanogenic archaeon Methanococcus maripaludis. J. Bacteriol. 185, 2548–2554 (2003)
Lupa, B., Hendrickson, E. L., Leigh, J. A. & Whitman, W. B. Formate-dependent H2 production by the mesophilic methanogen Methanococcus maripaludis. Appl. Environ. Microbiol. 74, 6584–6590 (2008)
Lee, H. S. et al. The complete genome sequence of Thermococcus onnurineus NA1 reveals a mixed heterotrophic and carboxydotrophic metabolism. J. Bacteriol. 190, 7491–7499 (2008)
Thauer, R. K. & Morris, J. G. in The Microbes 1984 Part II, Prokaryotes and Eukaryotes (eds Kelly, D. P. & Carr, N. G.) 123–168 (Cambridge Univ. Press, 1984)
Schink, B. Energetics of syntrophic cooperation in methanogenic degradation. Microbiol. Mol. Biol. Rev. 61, 262–280 (1997)
Zivanovic, Y. et al. Genome analysis and genome-wide proteomics of Thermococcus gammatolerans, the most radioresistant organism known amongst the Archaea. Genome Biol. 10, R70 (2009)
Möller, S., Croning, M. D. R. & Apweiler, R. Evaluation of methods for the prediction of membrane spanning regions. Bioinformatics 17, 646–653 (2001)
Gardy, J. L. et al. PSORTb v.2.0: expanded prediction of bacterial protein subcellular localization and insights gained from comparative proteome analysis. Bioinformatics 21, 617–623 (2005)
Sapra, R., Verhagen, M. F. & Adams, M. W. Purification and characterization of a membrane-bound hydrogenase from the hyperthermophilic archaeon Pyrococcus furiosus. J. Bacteriol. 182, 3423–3428 (2000)
Hedderich, R. & Forzi, L. Energy-converting [NiFe] hydrogenases: more than just H2 activation. J. Mol. Microbiol. Biotechnol. 10, 92–104 (2005)
Jenney, F. E., Jr & Adams, M. W. Hydrogenases of the model hyperthermophiles. Ann. NY Acad. Sci. 1125, 252–266 (2008)
Sokolova, T. G. et al. The first evidence of anaerobic CO oxidation coupled with H2 production by a hyperthermophilic archaeon isolated from a deep-sea hydrothermal vent. Extremophiles 8, 317–323 (2004)
Amend, J. P. & Shock, E. L. Energetics of overall metabolic reactions of thermophilic and hyperthermophilic Archaea and Bacteria. FEMS Microbiol. Rev. 25, 175–243 (2001)
Matsumi, R., Manabe, K., Fukui, T., Atomi, H. & Imanaka, T. Disruption of a sugar transporter gene cluster in a hyperthermophilic archaeon using a host-marker system based on antibiotic resistance. J. Bacteriol. 189, 2683–2691 (2007)
Bae, S. S. et al. Thermoccoccus onnurineus sp. nov., a hyperthermophilic archaeon isolated from a deep-sea hydrothermal vent area at the PACMANUS field. J. Microbiol. Biotechnol. 16, 1826–1831 (2006)
Holden, J. F. et al. Diversity among three novel groups of hyperthermophilic deep-sea Thermococcus species from three sites in the Northeastern Pacific Ocean. FEMS Microbiol. Ecol. 36, 51–60 (2001)
Amaratunga, D. & Cabrera, J. Analysis of data from viral DNA microchips. J. Am. Stat. Assoc. 96, 1161–1170 (2001)
Irizarry, R. A. et al. Exploration, normalization, and summaries of high density oligonucleotide array probe level data. Biostatistics 4, 249–264 (2003)
Acknowledgements
This work was supported by the KORDI in-house programme (PE98513), the Marine and Extreme Genome Research Center programme and the Development of Biohydrogen Production Technology using Hyperthermophilic Archaea programme of the Ministry of Land, Transport, and Maritime Affairs, Korea, as well as by the Molecular and Cell Biology programme of RAS and the Russian Foundation of Basic Research (grant no. 10-04-01180). We thank J. Querellou and the crew of the French scientific vessel Pourquoi pas?, and A.-L. Reysenbach and the crew of the American scientific vessel Thomas G. Thompson for opportunities to obtain deep-sea samples. We thank W. B. Whitman and R. K. Thauer for comments on the manuscript, and S. G. Jeon, K.-B. Yi and J.-G. Na for discussions.
Author information
Authors and Affiliations
Contributions
H.S.L., S.G.K. and J.-H.L. conceptualized and designed the experiments; Y.J.K., E.S.K. and S.S.B. performed most of the experiments with T. onurineus NA1 and analysed the data; J.K.L. contributed to the microarray and quantitative RT–PCR measurements; and K.K.K. performed gas analysis and analysed the data. T.G.S and D.A.K. performed growth experiments with Thermococcus strains other than T. onurineus NA1, and A.V.L. planned and analysed them. R.M., T.I. and H.A. contributed to developing a gene knockout system for T. onnurineus NA1. S.-S.C., E.A.B.-O. and S.-J.K. contributed critical comments on the manuscript. Y.J.K., H.S.L. and S.G.K. wrote the paper with input from the co-authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
The file contains Supplementary Tables 1-3 and Supplementary Figures 1-2 with legends. (PDF 304 kb)
Rights and permissions
About this article
Cite this article
Kim, Y., Lee, H., Kim, E. et al. Formate-driven growth coupled with H2 production. Nature 467, 352–355 (2010). https://doi.org/10.1038/nature09375
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature09375
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.