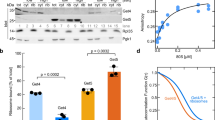
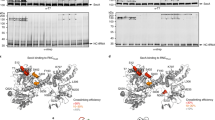
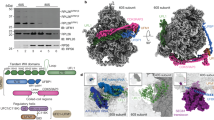
Abstract
Hundreds of proteins are inserted post-translationally into the endoplasmic reticulum (ER) membrane by a single carboxy-terminal transmembrane domain (TMD)1. During targeting through the cytosol, the hydrophobic TMD of these tail-anchored (TA) proteins requires constant chaperoning to prevent aggregation or inappropriate interactions. A central component of this targeting system is TRC40, a conserved cytosolic factor that recognizes the TMD of TA proteins and delivers them to the ER for insertion2,3,4. The mechanism that permits TRC40 to find and capture its TA protein cargos effectively in a highly crowded cytosol is unknown. Here we identify a conserved three-protein complex composed of Bat3, TRC35 and Ubl4A that facilitates TA protein capture by TRC40. This Bat3 complex is recruited to ribosomes synthesizing membrane proteins, interacts with the TMDs of newly released TA proteins, and transfers them to TRC40 for targeting. Depletion of the Bat3 complex allows non-TRC40 factors to compete for TA proteins, explaining their mislocalization in the analogous yeast deletion strains5,6,7. Thus, the Bat3 complex acts as a TMD-selective chaperone that effectively channels TA proteins to the TRC40 insertion pathway.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Rabu, C., Schmid, V., Schwappach, B. & High, S. Biogenesis of tail-anchored proteins: the beginning for the end? J. Cell Sci. 122, 3605–3612 (2009)
Stefanovic, S. & Hegde, R. S. Identification of a targeting factor for posttranslational membrane protein insertion into the ER. Cell 128, 1147–1159 (2007)
Schuldiner, M. et al. The GET complex mediates insertion of tail-anchored proteins into the ER membrane. Cell 134, 634–645 (2008)
Favaloro, V., Spasic, M., Schwappach, B. & Dobberstein, B. Distinct targeting pathways for the membrane insertion of tail-anchored (TA) proteins. J. Cell Sci. 121, 1832–1840 (2008)
Jonikas, M. C. et al. Comprehensive characterization of genes required for protein folding in the endoplasmic reticulum. Science 323, 1693–1697 (2009)
Copic, A. et al. Genomewide analysis reveals novel pathways affecting endoplasmic reticulum homeostasis, protein modification and quality control. Genetics 182, 757–769 (2009)
Costanzo, M. et al. The genetic landscape of a cell. Science 327, 425–431 (2010)
Chang, Y. W. et al. Crystal structure of Get4–Get5 complex and its interactions with Sgt2, Get3, and Ydj1. J. Biol. Chem. 285, 9962–9970 (2010)
Bozkurt, G. et al. The structure of Get4 reveals an α-solenoid fold adapted for multiple interactions in tail-anchored protein biogenesis. FEBS Lett. 584, 1509–1514 (2010)
Chartron, J. W., Suloway, C. J. M., Zaslaver, M. & Clemons, W. M., Jr Structural characterization of the Get4/5 complex and its interaction with Get3. Proc. Natl Acad. Sci. USA 107, 12127–12132 (2010)
Leznicki, P., Clancy, A., Schwappach, B. & High, S. Bat3 promotes the membrane integration of tail-anchored proteins. J. Cell Sci. 123, 2170–2178 (2010)
Ito, T. et al. A comprehensive two-hybrid analysis to explore the yeast protein interactome. Proc. Natl Acad. Sci. USA 98, 4569–4574 (2001)
Krogan, N. J. et al. Global landscape of protein complexes in the yeast Saccharomyces cerevisiae. Nature 440, 637–643 (2006)
Berndt, U., Oellerer, S., Zhang, Y., Johnson, A. E. & Rospert, S. A signal-anchor sequence stimulates signal recognition particle binding to ribosomes from inside the exit tunnel. Proc. Natl Acad. Sci. USA 106, 1398–1403 (2009)
Lodish, H. F. & Jacobsen, M. Regulation of hemoglobin synthesis: equal rates of translation and termination of α- and β-globin chains. J. Biol. Chem. 247, 3622–3629 (1972)
Wolin, S. L. & Walter, P. Signal recognition particle mediates a transient elongation arrest of preprolactin in reticulocyte lysate. J. Cell Biol. 109, 2617–2622 (1989)
Cao, J. & Geballe, A. P. Coding sequence-dependent ribosomal arrest at termination of translation. Mol. Cell. Biol. 16, 603–608 (1996)
Mateja, A. et al. The structural basis of tail-anchored membrane protein recognition by Get3. Nature 461, 361–366 (2009)
Bozkurt, G. et al. Structural insights into tail-anchored protein binding and membrane insertion by Get3. Proc. Natl Acad. Sci. USA 106, 21131–21136 (2009)
Suloway, C. J., Chartron, J. W., Zaslaver, M. & Clemons, W. M., Jr Model for eukaryotic tail-anchored protein binding based on the structure of Get3. Proc. Natl Acad. Sci. USA 106, 14849–14854 (2009)
Yamagata, A. et al. Structural insight into the membrane insertion of tail-anchored proteins by Get3. Genes Cells 15, 29–41 (2010)
Hu, J., Li, J., Qian, X., Denic, V. & Sha, B. The crystal structures of yeast Get3 suggest a mechanism for tail-anchored protein membrane insertion. PLoS ONE 4, e8061 (2009)
Desmots, F., Russell, H. R., Lee, Y., Boyd, K. & McKinnon, P. J. The Reaper-binding protein Scythe modulates apoptosis and proliferation during mammalian development. Mol. Cell. Biol. 25, 10329–10337 (2005)
Halic, M. et al. Structure of the signal recognition particle interacting with the elongation-arrested ribosome. Nature 427, 808–814 (2004)
Wang, S., Sakai, H. & Wiedmann, M. NAC covers ribosome-associated nascent chains thereby forming a protective environment for regions of nascent chains just emerging from the peptidyl transferase center. J. Cell Biol. 130, 519–528 (1995)
Gautschi, M. et al. RAC, a stable ribosome-associated complex in yeast formed by the DnaK-DnaJ homologs Ssz1p and zuotin. Proc. Natl Acad. Sci. USA 98, 3762–3767 (2001)
Kramer, G., Boehringer, D., Ban, N. & Bukau, B. The ribosome as a platform for co-translational processing, folding and targeting of newly synthesized proteins. Nature Struct. Mol. Biol. 16, 589–597 (2009)
Hobden, A. N. & Cundliffe, E. The mode of action of alpha sarcin and a novel assay of the puromycin reaction. Biochem. J. 170, 57–61 (1978)
High, S., Gorlich, D., Wiedmann, M., Rapoport, T. A. & Dobberstein, B. The identification of proteins in the proximity of signal-anchor sequences during their targeting to and insertion into the membrane of the ER. J. Cell Biol. 113, 35–44 (1991)
Fons, R. D., Bogert, B. A. & Hegde, R. S. Substrate-specific function of the translocon-associated protein complex during translocation across the ER membrane. J. Cell Biol. 160, 529–539 (2003)
Acknowledgements
We thank S. Appathurai and M. Downing for technical assistance, Hegde lab members for advice, and J. Weissman and W. Clemons for useful discussions and sharing results before publication. This work was supported by the Intramural Research Program of the National Institutes of Health (R.S.H.) and Edward Mallinckrodt Jr Foundation (R.J.K.).
Author information
Authors and Affiliations
Contributions
M.M. performed most of the functional analyses of the Bat3 complex, with significant contributions from X.L. during the initial phase of this study. S.S. and R.S.H. developed and characterized the RNC release assay, S.S. initially identified Bat3, and A.S. performed the tRNA-association experiments. A.M. and R.J.K. produced and functionally characterized recombinant proteins, and provided experimental ideas. M.M., X.L., S.S. and R.S.H. analysed data. R.S.H. conceived the project, guided experiments and wrote the paper with input from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures S1-S15 with legends (PDF 6141 kb)
Rights and permissions
About this article
Cite this article
Mariappan, M., Li, X., Stefanovic, S. et al. A ribosome-associating factor chaperones tail-anchored membrane proteins. Nature 466, 1120–1124 (2010). https://doi.org/10.1038/nature09296
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature09296
This article is cited by
-
UBXN1 maintains ER proteostasis and represses UPR activation by modulating translation
EMBO Reports (2024)
-
The role of RNF149 in the pre-emptive quality control substrate ubiquitination
Communications Biology (2023)
-
Ribosome-bound Get4/5 facilitates the capture of tail-anchored proteins by Sgt2 in yeast
Nature Communications (2021)
-
UBL4A inhibits autophagy-mediated proliferation and metastasis of pancreatic ductal adenocarcinoma via targeting LAMP1
Journal of Experimental & Clinical Cancer Research (2019)
-
The Ways of Tails: the GET Pathway and more
The Protein Journal (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.