Abstract
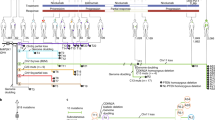
The question of whether tumorigenic cancer stem cells exist in human melanomas has arisen in the last few years1. Here we show that in melanomas, tumour stem cells (MTSCs, for melanoma tumour stem cells) can be isolated prospectively as a highly enriched CD271+ MTSC population using a process that maximizes viable cell transplantation1,2. The tumours sampled in this study were taken from a broad spectrum of sites and stages. High-viability cells isolated by fluorescence-activated cell sorting and re-suspended in a matrigel vehicle were implanted into T-, B- and natural-killer-deficient Rag2−/−γc−/− mice. The CD271+ subset of cells was the tumour-initiating population in 90% (nine out of ten) of melanomas tested. Transplantation of isolated CD271+ melanoma cells into engrafted human skin or bone in Rag2−/−γc−/− mice resulted in melanoma; however, melanoma did not develop after transplantation of isolated CD271− cells. We also show that in mice, tumours derived from transplanted human CD271+ melanoma cells were capable of metastatsis in vivo. CD271+ melanoma cells lacked expression of TYR, MART1 and MAGE in 86%, 69% and 68% of melanoma patients, respectively, which helps to explain why T-cell therapies directed at these antigens usually result in only temporary tumour shrinkage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
16 February 2011
Nature 466, 133–137 (2010) In this Letter, Fig. 1b and Table 2 (summarizing experiments presented in Supplementary Figs 2–4 and 7–9) contained an error reflecting the total number of tumours formed, and the total number of injections with CD271+ cells isolated directly from patients (26 out of 37). The correct number is 25 out of 38.
References
Quintana, E. et al. Efficient tumour formation by single human melanoma cells. Nature 456, 593–598 (2008)
Chan, K. S. et al. Identification, molecular characterization, clinical prognosis, and therapeutic targeting of human bladder tumor-initiating cells. Proc. Natl Acad. Sci. USA 106, 14016–14021 (2009)
Weissman, I. Stem cell research: paths to cancer therapies and regenerative medicine. J. Am. Med. Assoc. 294, 1359–1366 (2005)
Al-Hajj, M., Wicha, M. S., Benito-Hernandez, A., Morrison, S. J. & Clarke, M. F. Prospective identification of tumorigenic breast cancer cells. Proc. Natl Acad. Sci. USA 100, 3983–3988 (2003)
Jamieson, C. H. et al. Granulocyte-macrophage progenitors as candidate leukemic stem cells in blast-crisis CML. N. Engl. J. Med. 351, 657–667 (2004)
Singh, S. K. et al. Identification of a cancer stem cell in human brain tumors. Cancer Res. 63, 5821–5828 (2003)
Dalerba, P. et al. Phenotypic characterization of human colorectal cancer stem cells. Proc. Natl Acad. Sci. USA 104, 10158–10163 (2007)
O’Brien, C. A., Pollett, A., Gallinger, S. & Dick, J. E. A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature 445, 106–110 (2007)
Prince, M. E. et al. Identification of a subpopulation of cells with cancer stem cell properties in head and neck squamous cell carcinoma. Proc. Natl Acad. Sci. USA 104, 973–978 (2007)
Reya, T., Morrison, S. J., Clarke, M. F. & Weissman, I. L. Stem cells, cancer, and cancer stem cells. Nature 414, 105–111 (2001)
Baroffio, A., Dupin, E. & Le Douarin, N. M. Clone-forming ability and differentiation potential of migratory neural crest cells. Proc. Natl Acad. Sci. USA 85, 5325–5329 (1988)
Morrison, S. J., White, P. M., Zock, C. & Anderson, D. J. Prospective identification, isolation by flow cytometry, and in vivo self-renewal of multipotent mammalian neural crest stem cells. Cell 96, 737–749 (1999)
Chesa, P. G., Rettig, W. J., Thomson, T. M., Old, L. J. & Melamed, M. R. Immunohistochemical analysis of nerve growth factor receptor expression in normal and malignant human tissues. J. Histochem. Cytochem. 36, 383–389 (1988)
Pietra, G. et al. Natural killer cells kill human melanoma cells with characteristics of cancer stem cells. Int. Immunol. 21, 793–801 (2009)
Iwamoto, S., Odland, P. B., Piepkorn, M. & Bothwell, M. Evidence that the p75 neurotrophin receptor mediates perineural spread of desmoplastic melanoma. J. Am. Acad. Dermatol. 35, 725–731 (1996)
Marchetti, D., Menter, D., Jin, L., Nakajima, M. & Nicolson, G. L. Nerve growth factor effects on human and mouse melanoma cell invasion and heparanase production. Int. J. Cancer 55, 692–699 (1993)
Truzzi, F. et al. Neurotrophins and their receptors stimulate melanoma cell proliferation and migration. J. Invest. Dermatol. 128, 2031–2040 (2008)
Hérin, M. et al. Production of stable cytolytic T-cell clones directed against autologous human melanoma. Int. J. Cancer 39, 390–396 (1987)
van der Bruggen, P. et al. A gene encoding an antigen recognized by cytolytic T lymphocytes on a human melanoma. Science 254, 1643–1647 (1991)
Rosenberg, S. A. Development of cancer immunotherapies based on identification of the genes encoding cancer regression antigens. J. Natl Cancer Inst. 88, 1635–1644 (1996)
Cormier, J. N. et al. Enhancement of cellular immunity in melanoma patients immunized with a peptide from MART-1/Melan A. Cancer J. Sci. Am. 3, 37–44 (1997)
Scheibenbogen, C. et al. Phase 2 trial of vaccination with tyrosinase peptides and granulocyte-macrophage colony-stimulating factor in patients with metastatic melanoma. J. Immunother. 23, 275–281 (2000)
Schatton, T. et al. Identification of cells initiating human melanomas. Nature 451, 345–349 (2008)
La Porta, C. Cancer stem cells: lessons from melanoma. Stem Cell Rev. 5, 61–65 (2009)
Eaves, C. J. Cancer stem cells: Here, there, everywhere? Nature 456, 581–582 (2008)
Passegué, E., Rafii, S. & Herlyn, M. Cancer stem cells are everywhere. Nature Med. 15, 23 (2009)
Shackleton, M., Quintana, E., Fearon, E. R. & Morrison, S. J. Heterogeneity in cancer: cancer stem cells versus clonal evolution. Cell 138, 822–829 (2009)
Akashi, K., Traver, D., Miyamoto, T. & Weissman, I. L. A clonogenic common myeloid progenitor that gives rise to all myeloid lineages. Nature 404, 193–197 (2000)
Miyamoto, T., Weissman, I. L. & Akashi, K. AML1/ETO-expressing nonleukemic stem cells in acute myelogenous leukemia with 8;21 chromosomal translocation. Proc. Natl Acad. Sci. USA 97, 7521–7526 (2000)
Akala, O. O. et al. Long-term haematopoietic reconstitution by Trp53−/−p16Ink4a−/−p19Arf−/− multipotent progenitors. Nature 453, 228–232 (2008)
Acknowledgements
We thank J. Bueno and S. Hicks for obtaining patient consent and specimens; K. Montgomery for immunohistochemistry analyses; D. Kim for NSG bone-grafted mice; K. Chan, L. Ailles and C. Park for advice and help in FACS and tumour transplantation assays; L. Cohen for MAGE antibodies; L. Jerabek, T. Storm and A. Mosley for laboratory and mouse management; P. Lovelace for FACS management; A. Olson for help with microscopy; all members of the Weissman Laboratory for experimental suggestions; and M. Clarke and members of his laboratory for critical discussions and reading. This research was initially supported in part by the American Cancer Society fellowship and in part by NIH F32 CA126252 NRSA fellowship to A.D.B.; the Virginia & D.K. Ludwig Fund for Cancer Research to I.L.W.; NIH/NCRR CTSA grant UL1 RR025744 to Stanford Spectrum M.v.d.R.; The Oak Foundation and NIH 1RC2 DE02077-01 to M.T.L.; the Oak Foundation and Ellenburg Faculty Scholar Endowment to G.P.Y.
Author information
Authors and Affiliations
Contributions
A.D.B., M.T.L. and I.L.W. designed research; A.D.B. and O.V.R. performed research; M.v.d.R. and A.D.B. performed immunohistochemical and pathological analysis; S.M.S. provided clinical information; D.L.J., B.J. and M.J.K. provided human surgical melanoma tissues; D.P.L., P.D.B., G.P.Y. and M.T.L. provided humanized skin mouse model; A.D.B. and O.V.R. analysed the data; A.D.B. and I.L.W. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
I.L.W. was a member of the scientific advisory board of Amgen and owns significant Amgen stock; he co-founded and consulted for Systemix; he is a co-founder and director of Stem Cells, Inc.; and co-founded Cellerant, Inc. None of these companies is in the cancer stem cell field, at least while I.L.W. was an advisor or held the stock.
Supplementary information
Supplementary Information
This file contains Supplementary Tables 1-3, Supplementary Figures 1-28 and a list of the antibodies that were used in FACS analysis and cell sorting. The Supplementary Information file was corrected on 17 February 2011 (PDF 16088 kb)
Rights and permissions
About this article
Cite this article
Boiko, A., Razorenova, O., van de Rijn, M. et al. Human melanoma-initiating cells express neural crest nerve growth factor receptor CD271. Nature 466, 133–137 (2010). https://doi.org/10.1038/nature09161
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature09161
This article is cited by
-
Active targeting schemes for nano-drug delivery systems in osteosarcoma therapeutics
Journal of Nanobiotechnology (2023)
-
The journey from melanocytes to melanoma
Nature Reviews Cancer (2023)
-
RELA is required for CD271 expression and stem-like characteristics in hypopharyngeal cancer
Scientific Reports (2022)
-
Epigenetic control of melanoma cell invasiveness by the stem cell factor SALL4
Nature Communications (2021)
-
Rapid reprogramming of tumour cells into cancer stem cells on double-network hydrogels
Nature Biomedical Engineering (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.