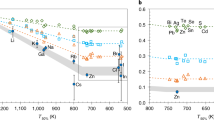
Abstract
The meaning of the age of the Earth defined by lead isotopes has long been unclear. Recently it has been proposed1 that the age of the Earth deduced from lead isotopes reflects volatile loss to space at the time of the Moon-forming giant impact rather than partitioning into metallic liquids during protracted core formation. Here we show that lead partitioning into liquid iron depends strongly on carbon content and that, given a content of ∼0.2% carbon2,3, experimental and isotopic data both provide evidence of strong partitioning of lead into the core throughout the Earth’s accretion. Earlier conclusions that lead is weakly partitioned into iron arose from the use of carbon-saturated (about 5% C) iron alloys. The lead isotopic age of the Earth is therefore consistent with partitioning into the core and with no significant late losses of moderately volatile elements to space during the giant impact.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lagos, M. et al. The Earth’s missing lead may not be in the core. Nature 456, 89–92 (2008)
Dasgupta, R. & Walker, D. Carbon solubility in core melts in a shallow magma ocean environment and distribution of carbon between the Earth’s core and the mantle. Geochim. Cosmochim. Acta 72, 4627–4641 (2008)
McDonough, W. F. in The Mantle and Core (ed. Carlson, R. W.) Vol. 2, 547–568 (Elsevier-Pergamon, Oxford, 2003)
Allègre, C. J., Manhes, G. & Gopel, C. The age of the Earth. Geochim. Cosmochim. Acta 59, 1445–1456 (1995)
Pepin, R. O. & Porcelli, D. R. Xenon isotope systematics, giant impacts, and mantle degassing on the early Earth. Earth Planet. Sci. Lett. 250, 470–485 (2006)
Pepin, R. O. Evolution of Earth’s noble gases: consequences of assuming hydrodynamic loss driven by giant impact. Icarus 126, 148–156 (1997)
Galer, S. J. G. & Goldstein, S. L. in Isotopic Studies of Crust-mantle Evolution (eds Basu, A. R. & Hart, S. R.) 75–98 (American Geophysical Union, 1996)
Touboul, M., Kleine, T., Bourdon, B., Palme, H. & Wieler, R. Late formation and prolonged differentiation of the Moon inferred from W isotopes in lunar metals. Nature 450, 1206–1209 (2007)
Halliday, A. N. A young Moon-forming giant impact at 70 to 110 million years accompanied by late-stage mixing, core formation and degassing of the Earth. Phil. Trans. R. Soc. Lond. A 366, 4163–4181 (2008)
Norman, M. D., Borg, L. E., Nyquist, L. E. & Bogard, D. D. Chronology, geochemistry, and petrology of a ferroan noritic anorthosite clast from Descartes breccia 67215: clues to the age, origin, structure, and impact history of the lunar crust. Meteorit. Planet. Sci. 38, 645–661 (2003)
Wasserburg, G. J., Papanastassiou, D. A., Tera, F. & Huneke, J. C. Outline of a lunar chronology. Phil. Trans. R. Soc. Lond., A 285, 7–22 (1977)
Malavergne, V. et al. New high-pressure and high-temperature metal/silicate partitioning of U and Pb: implications for the cores of the Earth and Mars. Geochim. Cosmochim. Acta 71, 2637–2655 (2007)
Allègre, C. J., Poirier, J.-P., Humler, E. & Hofmann, A. W. The chemical composition of the Earth. Earth Planet. Sci. Lett. 134, 515–526 (1995)
The Japan Society for the Promotion of Science and The 19th Committee on Steelmaking Steelmaking Data Sourcebook Part II, 273–312 (Gordon and Breach, 1988)
Malhotra, S., Chang, L. & Schlesinger, M. E. Liquid solution thermodynamics in the alumina-saturated Fe-C-Pb system. ISIJ Int. 38, 1–8 (1998)
Wood, B. J., Nielsen, S. G., Rehkämper, M. & Halliday, A. N. The effects of core formation on the Pb- and Tl-isotopic composition of the silicate Earth. Earth Planet. Sci. Lett. 269, 326–336 (2008)
Wade, J. & Wood, B. J. Core formation and the oxidation state of the Earth. Earth Planet. Sci. Lett. 236, 78–95 (2005)
Wood, B. J., Wade, J. & Kilburn, M. R. Core formation and the oxidation state of the Earth: additional constraints from Nb, V and Cr partitioning. Geochim. Cosmochim. Acta 72, 1415–1426 (2008)
Chabot, N. L., Saslow, S. A., McDonough, W. F. & Jones, J. H. An investigation of the behavior of Cu and Cr during iron meteorite crystallisation. Meteorit. Planet. Sci. 44, 505–520 (2009)
Dreibus, G. & Palme, H. Cosmochemical constraints on the sulfur content in the Earth’s core. Geochim. Cosmochim. Acta 60, 1125–1130 (1996)
Halliday, A. N. Mixing, volatile loss and compositional change during impact-driven accretion of the Earth. Nature 427, 505–509 (2004)
Yin, Q. Z. et al. A short timescale for terrestrial planet formation from Hf–W chronometry of meteorites. Nature 418, 949–952 (2002)
Canup, R. M. & Asphaug, E. Origin of the Moon in a giant impact near the end of the Earth’s formation. Nature 412, 708–712 (2001)
Wiechert, U. et al. Oxygen isotopes and the Moon-forming giant impact. Science 294, 345–348 (2001)
Georg, R. B., Halliday, A. N., Schauble, E. & Reynolds, B. C. Silicon in the Earth's core. Nature 447, 1102–1106 (2007)
Pahlevan, K. & Stevenson, D. J. Equilibration in the aftermath of the lunar-forming giant impact. Earth Planet. Sci. Lett. 262, 438–449 (2007)
Kleine, T. et al. Hf-W chronometry and the accretion and early evolution of asteroids and terrestrial planets. Geochim. Cosmochim. Acta (in the press)
Lodders, K. Solar system abundances and condensation temperatures of the elements. Astrophys. J. 591, 1220–1247 (2003)
McDonough, W. F. et al. Potassium, rubidium and cesium in the Earth and Moon and the evolution of the mantle of the Earth. Geochim. Cosmochim. Acta 53, 1001–1012 (1992)
Zahnle, K. et al. Emergence of a habitable planet. Space Sci. Rev. 129, 35–78 (2007)
Palme, H. & O'Neill, H. S. C. in The Mantle and Core (ed. Carlson, R. W.) Vol. 2, 1–38 (Elsevier, Amsterdam, 2003)
Barin, I., Sauert, F., Schultze-Rhonhof, E. & Sheng, W. S. Thermochemical Data of Pure Substances Parts I and II (CH Verlagsgesellschaft, 1989)
Nyquist, L. E., Shih, C. Y., Wiesmann, H. & Mikouchi, T. Fossil 26Al and 53Mn in d’Orbigny and Sahara 99555 and the timescale for angrite magmatism. Lunar Planet. Sci. Conf. XXXIV, 1388 (2004)
Norman, M. D., Pearson, N. J., Sharma, A. & Griffin, W. L. Quantitative analysis of trace elements in geological materials by laser ablation ICPMS: instrumental operating conditions and calibration values of NIST glasses. Geostand. Newsl. 20, 247–261 (1996)
Van Achterbergh, E., Ryan, C. G., Jackson, S. E. & Griffin, W. L. in Laser Ablation ICPMS in the Earth Sciences (ed. Sylvester, P.) Vol. 29, 239–243 (Mineralogical Association of Canada, 2001)
Acknowledgements
The assistance of N. Pearson (Macquarie), N. Charnley (Oxford) and J. Day (Cambridge) with microprobe and laser ICP-MS analysis is acknowledged with thanks. B.J.W. acknowledges the support of the Australian Research Council through Federation Fellowship FF 0456999 and the NERC (UK) through grant NE/F018266/1. A.N.H. acknowledges support from STFC. Experiments at the Bayerisches Geoinstitut were performed under the EU ‘Research Infrastructures: Transnational Access’ Programme (contract number 505320; RITA—High Pressure).
Author information
Authors and Affiliations
Contributions
B.J.W. performed all the experiments and all the electron microprobe and laser ICP-MS analyses, and the Pb-isotopic modelling of Fig. 3. A.N.H. performed the Sr-isotope modelling depicted in Fig. 4. Both authors contributed to the writing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Tables 1-2, Supplementary Data and Supplementary Figure S1 with legend. (PDF 623 kb)
Rights and permissions
About this article
Cite this article
Wood, B., Halliday, A. The lead isotopic age of the Earth can be explained by core formation alone. Nature 465, 767–770 (2010). https://doi.org/10.1038/nature09072
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature09072
This article is cited by
-
The accretion of planet Earth
Nature Reviews Earth & Environment (2022)
-
Ruthenium isotopic evidence for an inner Solar System origin of the late veneer
Nature (2017)
-
The great sulfur depletion of Earth’s mantle is not a signature of mantle–core equilibration
Contributions to Mineralogy and Petrology (2017)
-
Water in the Earth’s Interior: Distribution and Origin
Space Science Reviews (2017)
-
Missing Lead and High 3He/4He in Ancient Sulfides Associated with Continental Crust Formation
Scientific Reports (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.