Abstract
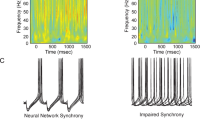
Abnormalities in functional connectivity between brain areas have been postulated as an important pathophysiological mechanism underlying schizophrenia1,2. In particular, macroscopic measurements of brain activity in patients suggest that functional connectivity between the frontal and temporal lobes may be altered3,4. However, it remains unclear whether such dysconnectivity relates to the aetiology of the illness, and how it is manifested in the activity of neural circuits. Because schizophrenia has a strong genetic component5, animal models of genetic risk factors are likely to aid our understanding of the pathogenesis and pathophysiology of the disease. Here we study Df(16)A+/– mice, which model a microdeletion on human chromosome 22 (22q11.2) that constitutes one of the largest known genetic risk factors for schizophrenia6. To examine functional connectivity in these mice, we measured the synchronization of neural activity between the hippocampus and the prefrontal cortex during the performance of a task requiring working memory, which is one of the cognitive functions disrupted in the disease. In wild-type mice, hippocampal–prefrontal synchrony increased during working memory performance, consistent with previous reports in rats7. Df(16)A+/– mice, which are impaired in the acquisition of the task, showed drastically reduced synchrony, measured both by phase-locking of prefrontal cells to hippocampal theta oscillations and by coherence of prefrontal and hippocampal local field potentials. Furthermore, the magnitude of hippocampal–prefrontal coherence at the onset of training could be used to predict the time it took the Df(16)A+/– mice to learn the task and increased more slowly during task acquisition. These data suggest how the deficits in functional connectivity observed in patients with schizophrenia may be realized at the single-neuron level. Our findings further suggest that impaired long-range synchrony of neural activity is one consequence of the 22q11.2 deletion and may be a fundamental component of the pathophysiology underlying schizophrenia.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Stephan, K. E., Friston, K. J. & Frith, C. D. Dysconnection in schizophrenia: from abnormal synaptic plasticity to failures of self-monitoring. Schizophr. Bull. 35, 509–527 (2009)
Wernicke, C. Grundrisse der Psychiatrie (Thieme, 1906)
Meyer-Lindenberg, A. S. et al. Regionally specific disturbance of dorsolateral prefrontal-hippocampal functional connectivity in schizophrenia. Arch. Gen. Psychiatry 62, 379–386 (2005)
Lawrie, S. M. et al. Reduced frontotemporal functional connectivity in schizophrenia associated with auditory hallucinations. Biol. Psychiatry 51, 1008–1011 (2002)
Gottesman, I. I. & Shields, J. A polygenic theory of schizophrenia. Proc. Natl Acad. Sci. USA 58, 199–205 (1967)
Karayiorgou, M. & Gogos, J. A. The molecular genetics of the 22q11-associated schizophrenia. Brain Res. Mol. Brain Res. 132, 95–104 (2004)
Jones, M. W. & Wilson, M. A. Theta rhythms coordinate hippocampal-prefrontal interactions in a spatial memory task. PLoS Biol. 3, e402 (2005)
Ford, J. M., Mathalon, D. H., Whitfield, S., Faustman, W. O. & Roth, W. T. Reduced communication between frontal and temporal lobes during talking in schizophrenia. Biol. Psychiatry 51, 485–492 (2002)
Karayiorgou, M. et al. Schizophrenia susceptibility associated with interstitial deletions of chromosome 22q11. Proc. Natl Acad. Sci. USA 92, 7612–7616 (1995)
Xu, B. et al. Strong association of de novo copy number mutations with sporadic schizophrenia. Nature Genet. 40, 880–885 (2008)
The International Schizophrenia Consortium. Rare chromosomal deletions and duplications increase risk of schizophrenia. Nature 455, 237–241 (2008)
Stefansson, H. et al. Large recurrent microdeletions associated with schizophrenia. Nature 455, 232–236 (2008)
Mukai, J. et al. Palmitoylation-dependent neurodevelopmental deficits in a mouse model of 22q11 microdeletion. Nature Neurosci. 11, 1302–1310 (2008)
Stark, K. L. et al. Altered brain microRNA biogenesis contributes to phenotypic deficits in a 22q11-deletion mouse model. Nature Genet. 40, 751–760 (2008)
Forbes, N. F., Carrick, L. A., McIntosh, A. M. & Lawrie, S. M. Working memory in schizophrenia: a meta-analysis. Psychol. Med. 39, 889–905 (2009)
Floresco, S. B., Seamans, J. K. & Phillips, A. G. Selective roles for hippocampal, prefrontal cortical, and ventral striatal circuits in radial-arm maze tasks with or without a delay. J. Neurosci. 17, 1880–1890 (1997)
Siapas, A. G., Lubenov, E. V. & Wilson, M. A. Prefrontal phase locking to hippocampal theta oscillations. Neuron 46, 141–151 (2005)
Buzsáki, G. et al. Hippocampal network patterns of activity in the mouse. Neuroscience 116, 201–211 (2003)
Kates, W. R. et al. The neural correlates of non-spatial working memory in velocardiofacial syndrome (22q11.2 deletion syndrome). Neuropsychologia 45, 2863–2873 (2007)
Sobin, C. et al. Neuropsychological characteristics of children with the 22q11 deletion syndrome: a descriptive analysis. Child Neuropsychol. 11, 39–53 (2005)
Lewandowski, K. E., Shashi, V., Berry, P. M. & Kwapil, T. R. Schizophrenic-like neurocognitive deficits in children and adolescents with 22q11 deletion syndrome. Am. J. Med. Genet. B 144B, 27–36 (2007)
Bertolino, A. et al. Prefrontal-hippocampal coupling during memory processing is modulated by COMT Val158Met genotype. Biol. Psychiatry 60, 1250–1258 (2006)
Esslinger, C. et al. Neural mechanisms of a genome-wide supported psychosis variant. Science 324, 605 (2009)
Liu, H. et al. Genetic variation in the 22q11 locus and susceptibility to schizophrenia. Proc. Natl Acad. Sci. USA 99, 16859–16864 (2002)
Mukai, J. et al. Evidence that the gene encoding ZDHHC8 contributes to the risk of schizophrenia. Nature Genet. 36, 725–731 (2004)
Paterlini, M. et al. Transcriptional and behavioral interaction between 22q11.2 orthologs modulates schizophrenia-related phenotypes in mice. Nature Neurosci. 8, 1586–1594 (2005)
Raux, G. et al. Involvement of hyperprolinemia in cognitive and psychiatric features of the 22q11 deletion syndrome. Hum. Mol. Genet. 16, 83–91 (2007)
Yavich, L., Forsberg, M. M., Karayiorgou, M., Gogos, J. A. & Mannisto, P. T. Site-specific role of catechol-O-methyltransferase in dopamine overflow within prefrontal cortex and dorsal striatum. J. Neurosci. 27, 10196–10209 (2007)
Schmitzer-Torbert, N., Jackson, J., Henze, D., Harris, K. & Redish, A. D. Quantitative measures of cluster quality for use in extracellular recordings. Neuroscience 131, 1–11 (2005)
Kelly, R. C. et al. Comparison of recordings from microelectrode arrays and single electrodes in the visual cortex. J. Neurosci. 27, 261–264 (2007)
Acknowledgements
We would like to thank M. Topiwala, A. Adhikari and Y. Sun for technical assistance. We also thank A. Adhikari, L. Drew and A. Arguello for comments on the manuscript. This work was supported by the Simons Foundation (J. A. Gogos), US National Institute of Mental Health grants MH67068 (M.K. and J. A. Gogos) and MH081968 (J. A. Gordon), and the Lieber Center for Schizophrenia Research and Treatment.
Author Contributions T.S., M.K., J. A. Gogos and J. A. Gordon designed the experiments. T.S. carried out the behavioural and electrophysiology experiments. K.L.S. engineered and supplied the mutant and control mice and contributed to the experimental design. T.S. and J. A. Gordon analysed the data. T.S., M.K., J. A. Gogos and J. A. Gordon interpreted the results and wrote the paper.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Table 1 and Supplementary Figures 1-7 with legends. (PDF 1968 kb)
Rights and permissions
About this article
Cite this article
Sigurdsson, T., Stark, K., Karayiorgou, M. et al. Impaired hippocampal–prefrontal synchrony in a genetic mouse model of schizophrenia. Nature 464, 763–767 (2010). https://doi.org/10.1038/nature08855
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature08855
This article is cited by
-
A developmental increase of inhibition promotes the emergence of hippocampal ripples
Nature Communications (2024)
-
Atypical cortical networks in children at high-genetic risk of psychiatric and neurodevelopmental disorders
Neuropsychopharmacology (2024)
-
Inversed Effects of Nav1.2 Deficiency at Medial Prefrontal Cortex and Ventral Tegmental Area for Prepulse Inhibition in Acoustic Startle Response
Molecular Neurobiology (2024)
-
Striatum-projecting prefrontal cortex neurons support working memory maintenance
Nature Communications (2023)
-
Osteocalcin ameliorates cognitive dysfunctions in a mouse model of Alzheimer’s Disease by reducing amyloid β burden and upregulating glycolysis in neuroglia
Cell Death Discovery (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.