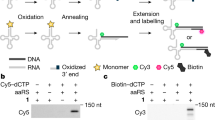
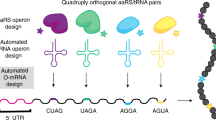
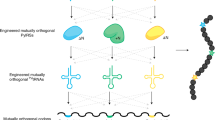
Abstract
The in vivo, genetically programmed incorporation of designer amino acids allows the properties of proteins to be tailored with molecular precision1. The Methanococcus jannaschii tyrosyl-transfer-RNA synthetase–tRNACUA (MjTyrRS–tRNACUA)2,3 and the Methanosarcina barkeri pyrrolysyl-tRNA synthetase–tRNACUA (MbPylRS–tRNACUA)4,5,6 orthogonal pairs have been evolved to incorporate a range of unnatural amino acids in response to the amber codon in Escherichia coli1,6,7. However, the potential of synthetic genetic code expansion is generally limited to the low efficiency incorporation of a single type of unnatural amino acid at a time, because every triplet codon in the universal genetic code is used in encoding the synthesis of the proteome. To encode efficiently many distinct unnatural amino acids into proteins we require blank codons and mutually orthogonal aminoacyl-tRNA synthetase–tRNA pairs that recognize unnatural amino acids and decode the new codons. Here we synthetically evolve an orthogonal ribosome8,9 (ribo-Q1) that efficiently decodes a series of quadruplet codons and the amber codon, providing several blank codons on an orthogonal messenger RNA, which it specifically translates8. By creating mutually orthogonal aminoacyl-tRNA synthetase–tRNA pairs and combining them with ribo-Q1 we direct the incorporation of distinct unnatural amino acids in response to two of the new blank codons on the orthogonal mRNA. Using this code, we genetically direct the formation of a specific, redox-insensitive, nanoscale protein cross-link by the bio-orthogonal cycloaddition of encoded azide- and alkyne-containing amino acids10. Because the synthetase–tRNA pairs used have been evolved to incorporate numerous unnatural amino acids1,6,7, it will be possible to encode more than 200 unnatural amino acid combinations using this approach. As ribo-Q1 independently decodes a series of quadruplet codons, this work provides foundational technologies for the encoded synthesis and synthetic evolution of unnatural polymers in cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Xie, J. & Schultz, P. G. A chemical toolkit for proteins–an expanded genetic code. Nature Rev. Mol. Cell Biol. 7, 775–782 (2006)
Steer, B. A. & Schimmel, P. Major anticodon-binding region missing from an archaebacterial tRNA synthetase. J. Biol. Chem. 274, 35601–35606 (1999)
Chin, J. W., Martin, A. B., King, D. S., Wang, L. & Schultz, P. G. Addition of a photocrosslinking amino acid to the genetic code of Escherichia coli. Proc. Natl Acad. Sci. USA 99, 11020–11024 (2002)
Srinivasan, G., James, C. M. & Krzycki, J. A. Pyrrolysine encoded by UAG in Archaea: charging of a UAG-decoding specialized tRNA. Science 296, 1459–1462 (2002)
Polycarpo, C. et al. An aminoacyl-tRNA synthetase that specifically activates pyrrolysine. Proc. Natl Acad. Sci. USA 101, 12450–12454 (2004)
Neumann, H., Peak-Chew, S. Y. & Chin, J. W. Genetically encoding Nε-acetyllysine in recombinant proteins. Nature Chem. Biol. 4, 232–234 (2008)
Nguyen, D. P. et al. Genetic encoding and labeling of aliphatic azides and alkynes in recombinant proteins via a pyrrolysyl-tRNA synthetase/tRNA(CUA) pair and click chemistry. J. Am. Chem. Soc. 131, 8720–8721 (2009)
Rackham, O. & Chin, J. W. A network of orthogonal ribosome·mRNA pairs. Nature Chem. Biol. 1, 159–166 (2005)
Wang, K., Neumann, H., Peak-Chew, S. Y. & Chin, J. W. Evolved orthogonal ribosomes enhance the efficiency of synthetic genetic code expansion. Nature Biotechnol. 25, 770–777 (2007)
Rostovtsev, V. V., Green, L. G., Fokin, V. V. & Sharpless, K. B. A stepwise huisgen cycloaddition process: copper(i)-catalyzed regioselective “ligation” of azides and terminal alkynes. Angew. Chem. Int. Edn Engl. 41, 2596–2599 (2002)
Hohsaka, T. & Sisido, M. Incorporation of non-natural amino acids into proteins. Curr. Opin. Chem. Biol. 6, 809–815 (2002)
Ohtsuki, T., Manabe, T. & Sisido, M. Multiple incorporation of non-natural amino acids into a single protein using tRNAs with non-standard structures. FEBS Lett. 579, 6769–6774 (2005)
Murakami, H., Hohsaka, T., Ashizuka, Y. & Sisido, M. Site-directed incorporation of p-nitrophenylalanine into streptavidin and site-to-site photoinduced electron transfer from a pyrenyl group to a nitrophenyl group on the protein framework. J. Am. Chem. Soc. 120, 7520–7529 (1998)
Rodriguez, E. A., Lester, H. A. & Dougherty, D. A. In vivo incorporation of multiple unnatural amino acids through nonsense and frameshift suppression. Proc. Natl Acad. Sci. USA 103, 8650–8655 (2006)
Monahan, S. L., Lester, H. A. & Dougherty, D. A. Site-specific incorporation of unnatural amino acids into receptors expressed in mammalian cells. Chem. Biol. 10, 573–580 (2003)
Anderson, J. C. et al. An expanded genetic code with a functional quadruplet codon. Proc. Natl Acad. Sci. USA 101, 7566–7571 (2004)
Atkins, J. F. & Bjork, G. R. A gripping tale of ribosomal frameshifting: extragenic suppressors of frameshift mutations spotlight P-site realignment. Microbiol. Mol. Biol. Rev. 73, 178–210 (2009)
Stahl, G., McCarty, G. P. & Farabaugh, P. J. Ribosome structure: revisiting the connection between translational accuracy and unconventional decoding. Trends Biochem. Sci. 27, 178–183 (2002)
Selmer, M. et al. Structure of the 70S ribosome complexed with mRNA and tRNA. Science 313, 1935–1942 (2006)
Magliery, T. J., Anderson, J. C. & Schultz, P. G. Expanding the genetic code: selection of efficient suppressors of four-base codons and identification of “shifty” four-base codons with a library approach in Escherichia coli. J. Mol. Biol. 307, 755–769 (2001)
Khazaie, K., Buchanan, J. H. & Rosenberger, R. F. The accuracy of Qβ RNA translation. 1. Errors during the synthesis of Qβ proteins by intact Escherichia coli cells. Eur. J. Biochem. 144, 485–489 (1984)
Laughrea, M., Latulippe, J., Filion, A. M. & Boulet, L. Mistranslation in twelve Escherichia coli ribosomal proteins. Cysteine misincorporation at neutral amino acid residues other than tryptophan. Eur. J. Biochem. 169, 59–64 (1987)
Kramer, E. B. & Farabaugh, P. J. The frequency of translational misreading errors in E. coli is largely determined by tRNA competition. RNA 13, 87–96 (2007)
Chin, J. W. et al. Addition of p-azido-l-phenylalanine to the genetic code of Escherichia coli. J. Am. Chem. Soc. 124, 9026–9027 (2002)
Mukai, T. et al. Adding l-lysine derivatives to the genetic code of mammalian cells with engineered pyrrolysyl-tRNA synthetases. Biochem. Biophys. Res. Commun. 371, 818–822 (2008)
Camarero, J. A., Pavel, J. & Muir, T. W. Chemical synthesis of a circular protein domain: evidence for folding-assisted cyclization. Angew. Chem. Int. Ed. 37, 347–349 (1998)
Scott, C. P., Abel-Santos, E., Wall, M., Wahnon, D. C. & Benkovic, S. J. Production of cyclic peptides and proteins in vivo. Proc. Natl Acad. Sci. USA 96, 13638–13643 (1999)
Li, P. & Roller, P. P. Cyclization strategies in peptide derived drug design. Curr. Top. Med. Chem. 2, 325–341 (2002)
Walensky, L. D. et al. Activation of apoptosis in vivo by a hydrocarbon-stapled BH3 helix. Science 305, 1466–1470 (2004)
Trauger, J. W., Kohli, R. M., Mootz, H. D., Marahiel, M. A. & Walsh, C. T. Peptide cyclization catalysed by the thioesterase domain of tyrocidine synthetase. Nature 407, 215–218 (2000)
Acknowledgements
We are grateful to P. B. Kapadnis for synthesizing CAK and to W. An for assistance with some experiments. J.W.C. is grateful to the ERC and the MRC for funding. K.W. is grateful to Trinity College, Cambridge for a fellowship.
Author Contributions K.W., H.N., L.D. and J.W.C. planned the experiments. K.W. selected and characterized ribo-Q, with help from L.D. K.W. and L.D. characterized amber and quadruplet incorporation by protein expression and mass spectrometry. L.D. and M.G.-A. performed protein expression experiments. H.N. demonstrated the mutual orthogonality of synthetase tRNA pairs, evolved synthetases, and characterized the double incorporation and protein cyclization, with help from M.G.-A. H.N., K.W., L.D. and J.W.C. analysed the data and wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Methods, Supplementary Figures 1-13 with Legends, Supplementary Table 1 and Supplementary References. (PDF 5555 kb)
Rights and permissions
About this article
Cite this article
Neumann, H., Wang, K., Davis, L. et al. Encoding multiple unnatural amino acids via evolution of a quadruplet-decoding ribosome. Nature 464, 441–444 (2010). https://doi.org/10.1038/nature08817
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08817
This article is cited by
-
Adding α,α-disubstituted and β-linked monomers to the genetic code of an organism
Nature (2024)
-
Anticodon table of the chloroplast genome and identification of putative quadruplet anticodons in chloroplast tRNAs
Scientific Reports (2023)
-
Extensive breaking of genetic code degeneracy with non-canonical amino acids
Nature Communications (2023)
-
Quintuply orthogonal pyrrolysyl-tRNA synthetase/tRNAPyl pairs
Nature Chemistry (2023)
-
Nature-inspired protein ligation and its applications
Nature Reviews Chemistry (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.