Abstract
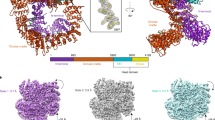
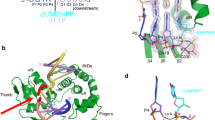
Broken chromosomes arising from DNA double-strand breaks result from endogenous events such as the production of reactive oxygen species during cellular metabolism, as well as from exogenous sources such as ionizing radiation1,2,3. Left unrepaired or incorrectly repaired they can lead to genomic changes that may result in cell death or cancer. DNA-dependent protein kinase (DNA-PK), a holoenzyme that comprises the DNA-PK catalytic subunit (DNA-PKcs)4,5 and the heterodimer Ku70/Ku80, has a major role in non-homologous end joining—the main pathway in mammals used to repair double-strand breaks6,7,8. DNA-PKcs is a serine/threonine protein kinase comprising a single polypeptide chain of 4,128 amino acids and belonging to the phosphatidylinositol-3-OH kinase (PI(3)K)-related protein family9. DNA-PKcs is involved in the sensing and transmission of DNA damage signals to proteins such as p53, setting off events that lead to cell cycle arrest10,11. It phosphorylates a wide range of substrates in vitro, including Ku70/Ku80, which is translocated along DNA12. Here we present the crystal structure of human DNA-PKcs at 6.6 Å resolution, in which the overall fold is clearly visible, to our knowledge, for the first time. The many α-helical HEAT repeats (helix–turn–helix motifs) facilitate bending and allow the polypeptide chain to fold into a hollow circular structure. The carboxy-terminal kinase domain is located on top of this structure, and a small HEAT repeat domain that probably binds DNA is inside. The structure provides a flexible cradle to promote DNA double-strand-break repair.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Kemp, L. M., Sedgwick, S. G. & Jeggo, P. A. X-ray sensitive mutants of Chinese hamster ovary cells defective in double-strand break rejoining. Mutat. Res. 132, 189–196 (1984)
Zdzienicka, M. Z., Tran, Q., van der Schans, G. P. & Simons, J. W. I. Characterization of an X-ray-hypersensitive mutant of V79 Chinese hamster cells. Mutat. Res. 194, 239–249 (1988)
Biedermann, K. A., Sun, J., Giaccia, A. J., Tosto, L. M. & Brown, J. M. Scid mutation in mice confers hypersensitivity to ionizing radiation and a deficiency in DNA double-strand break repair. Proc. Natl Acad. Sci. USA 88, 1394–1397 (1991)
Dvir, A., Stein, L. Y., Calore, B. L. & Dynan, W. S. Purification and characterization of a template associated protein kinase that phosphorylates RNA polymerase II. J. Biol. Chem. 268, 10440–10447 (1993)
Carter, T., Vancurová, I., Sun, I., Lou, W. & DeLeon, S. A DNA-activated protein kinase from HeLa cell nuclei. Mol. Cell. Biol. 10, 6460–6471 (1990)
Critchlow, S. E. & Jackson, S. P. DNA end-joining: from yeast to man. Trends Biochem. Sci. 23, 394–398 (1998)
Gottlieb, T. M. & Jackson, S. P. The DNA-dependent protein kinase requirement for DNA ends and association with Ku antigen. Cell 72, 131–142 (1993)
Ma, Y., Pannicke, U., Schwarz, K. & Lieber, M. R. Hairpin opening and overhang processing by an Artemis/DNA-dependent protein kinase complex in nonhomologous end joining and V(D)J recombination. Cell 108, 781–794 (2002)
Hartley, K. O. et al. DNA-dependent protein kinase catalytic subunit: a relative of phosphatidylinositol 3-kinase and the ataxia telangiectasia gene product. Cell 82, 849–856 (1995)
Hoekstra, M. F. Responses to DNA damage and regulation of cell cycle checkpoints by the ATM protein kinase family. Curr. Opin. Genet. Dev. 7, 170–175 (1997)
Anderson, C. W. DNA damage and the DNA-activated protein kinase. Trends Biochem. Sci. 18, 433–437 (1993)
Yoo, S. & Dynan, W. S. Geometry of a complex formed by double strand break repair proteins at a single DNA end: recruitment of DNA-PKcs induces inward translocation of Ku protein. Nucleic Acids Res. 27, 4679–4686 (1999)
Harris, R. et al. The 3D solution structure of the C-terminal region of Ku86 (Ku86CTR). J. Mol. Biol. 335, 573–582 (2004)
Zhang, Z. et al. Solution structure of the C-terminal domain of Ku80 suggests important sites for protein–protein interactions. Structure 12, 495–502 (2004)
Chiu, C. Y., Cary, R. B., Chen, D. J., Peterson, S. R. & Stewart, P. L. Cryo-EM imaging of the catalytic subunit of the DNA-dependent protein kinase. J. Mol. Biol. 284, 1075–1081 (1998)
Boskovic, J. et al. Visualization of DNA-induced conformational changes in the DNA repair kinase DNA-PKcs. EMBO J. 22, 5875–5882 (2003)
Rivera-Calzada, A., Maman, J. P., Spagnolo, L., Pearl, L. H. & Llorca, O. Three-dimensional structure and regulation of the DNA-dependent protein kinase catalytic subunit (DNA-PKcs). Structure 13, 243–255 (2005)
Williams, D. R., Lee, K.-J., Shi, J., Chen, D. J. & Stewart, P. L. Cryo-EM structure of the DNA-dependent protein kinase catalytic subunit at subnanometer resolution reveals α-helices and insight into DNA binding. Structure 16, 468–477 (2008)
Groves, M. R., Hanlon, N., Turowski, P., Hemmings, B. A. & Barford, D. The structure of the protein phosphatase 2A PR65/A subunit reveals the conformation of its 15 tandemly repeated HEAT motifs. Cell 96, 99–110 (1999)
Cingolani, G., Petosa, C., Weis, K. & Müller, C. W. Structure of importin-β bound to the IBB domain of importin-α. Nature 399, 221–229 (1999)
Goldenberg, S. J. et al. Structure of the Cand1–Cul1–Roc1 complex reveals regulatory mechanisms for the assembly of the multisubunit cullin-dependent ubiquitin ligases. Cell 119, 517–528 (2004)
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004)
Murshudov, G. N., Vagin, A. A. & Dodson, E. J. Refinement of macromolecular structures by the maximum-likelihood method. Acta Crystallogr. D 53, 240–255 (1997)
Spagnolo, L., Rivera-Calzada, A., Pearl, L. H. & Llorca, O. Three-dimensional structure of the human DNA-PKcs/Ku70/Ku80 complex assembled on DNA and its implications for DNA DSB repair. Mol. Cell 22, 511–519 (2006)
Meek, K., Douglas, P., Cui, X., Ding, Q. & Lees-Miller, S. P. trans autophosphorylation at DNA-dependent protein kinase’s two major autophosphorylation site clusters facilitates end processing but not end joining. Mol. Cell. Biol. 27, 3881–3890 (2007)
Yaneva, M., Kowalewski, T. & Lieber, M. R. Interaction of DNA-dependent protein kinase with DNA and with Ku: biochemical and atomic force microscopy studies. EMBO J. 16, 5098–5112 (1997)
Walker, E. H., Perisic, O., Ried, C., Stephens, L. & Williams, R. L. Structural insights into phosphoinositide 3-kinase catalysis and signaling. Nature 402, 313–320 (1999)
Bosotti, R., Isacchi, A. & Sonnhammer, E. L. L. FAT: a novel domain in PIK-related kinases. Trends Biochem. Sci. 25, 225–227 (2000)
Dames, S. A., Mulet, J. M., Rathgeb-Szabo, K., Hall, M. N. & Grzesiek, S. The solution structure of the FATC Domain of the protein kinase TOR suggests a role for redox-dependent structural and cellular stability. J. Biol. Chem. 280, 20558–20564 (2005)
Leuther, K. K., Hammarsten, O., Kornberg, R. D. & Chu, G. Structure of DNA-dependent protein kinase: implications for its regulation by DNA. EMBO J. 18, 1114–1123 (1999)
Gell, D. & Jackson, S. P. Mapping of protein-protein interactions within the DNA-dependent protein kinase complex. Nucleic Acids Res. 27, 3494–3502 (1999)
Ausubel, F. M. et al. in Short Protocols in Molecular Biology: A Compendium of Methods from Current Protocols in Molecular Biology 5th edn 12.3–12.6 (Wiley, 2002)
Schneider, G. & Lindqvist, Y. Ta6Brl4 is a useful cluster compound for isomorphous replacement in protein crystallography. Acta Crystallogr. D 50, 186–191 (1994)
Leonard, G. A. et al. Online collection and analysis of X-ray fluorescence spectra on the macromolecular crystallography beamlines of the ESRF. J. Appl. Crystallogr. 42, 333–335 (2009)
Leslie, A. G. W. Recent changes to the MOSFLM package for processing film and image plate data. Joint CCP4 and ESF-EAMCB Newsletter on Protein Crystallography no. 26, (1992)
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997)
Collaborative Computational Project, Number 4. The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994)
Adams, P. D. et al. PHENIX: building new software for automated crystallographic structure determination. Acta Crystallogr. D 58, 1948–1954 (2002)
Bricogne, G., Vonrhein, C., Flensburg, C., Schiltz, M. & Paciorek, W. Generation, representation and flow of phase information in structure determination: recent developments in and around SHARP 2.0. Acta Crystallogr. D 59, 2023–2030 (2003)
Kleywegt, G. J. Use of non-crystallographic symmetry in protein structure refinement. Acta Crystallogr. D 52, 842–857 (1996)
Acknowledgements
We wish to thank G. Smith for providing pure DNA-PKcs that was used in the initial experiments as a marker and also for providing the DNA-PKcs and Ku70 antibodies, L. Pellegrini for help and advice at the beginning of the project, and S. Jackson for discussions. We are also grateful to R. Peat for preparing HeLa cells, L. Packman for help in identifying DNA-PKcs in polyacrylamide gels, N. Ban for providing Ta6Br122+ and C. Müller-Dieckmann at the ESRF for support during the diffraction data collection experiments. This work was funded by the Wellcome Trust and CR-UK.
Author Contributions T.L.B. and B.L.S. conceived the project. B.L.S. characterized, purified, crystallized and analysed the electron density for the DNA-PKcs–Ku80ct complex. D.Y.C. and B.L.S. carried out data collection and structure modelling. D.Y.C. carried out data processing, electron density calculations and refinement, with significant input from T.L.B. in the interpretation of the data. B.L.S. wrote the paper and all authors contributed and edited the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Information
This file contains Supplementary Table 1, Supplementary Figures 1-3 with Legends and a Legend for Supplementary Movie 1. (PDF 1711 kb)
Supplementary Information
This movie shows the 360 degrees view of a single DNA-PKcs molecule displayed as molecular surface (see Supplementary Information file for full Legend). (MOV 7455 kb)
Rights and permissions
About this article
Cite this article
Sibanda, B., Chirgadze, D. & Blundell, T. Crystal structure of DNA-PKcs reveals a large open-ring cradle comprised of HEAT repeats. Nature 463, 118–121 (2010). https://doi.org/10.1038/nature08648
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08648
This article is cited by
-
Human DNA-dependent protein kinase activation mechanism
Nature Structural & Molecular Biology (2023)
-
Role of PRKDC in cancer initiation, progression, and treatment
Cancer Cell International (2021)
-
Pak2 reduction induces a failure of early embryonic development in mice
Reproductive Biology and Endocrinology (2021)
-
Dimers of DNA-PK create a stage for DNA double-strand break repair
Nature Structural & Molecular Biology (2021)
-
The activation mechanisms of master kinases in the DNA damage response
Genome Instability & Disease (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.