Abstract
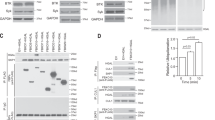
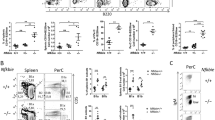
A role for B-cell-receptor (BCR) signalling in lymphomagenesis has been inferred by studying immunoglobulin genes in human lymphomas1,2 and by engineering mouse models3, but genetic and functional evidence for its oncogenic role in human lymphomas is needed. Here we describe a form of ‘chronic active’ BCR signalling that is required for cell survival in the activated B-cell-like (ABC) subtype of diffuse large B-cell lymphoma (DLBCL). The signalling adaptor CARD11 is required for constitutive NF-κB pathway activity and survival in ABC DLBCL4. Roughly 10% of ABC DLBCLs have mutant CARD11 isoforms that activate NF-κB5, but the mechanism that engages wild-type CARD11 in other ABC DLBCLs was unknown. An RNA interference genetic screen revealed that a BCR signalling component, Bruton’s tyrosine kinase, is essential for the survival of ABC DLBCLs with wild-type CARD11. In addition, knockdown of proximal BCR subunits (IgM, Ig-κ, CD79A and CD79B) killed ABC DLBCLs with wild-type CARD11 but not other lymphomas. The BCRs in these ABC DLBCLs formed prominent clusters in the plasma membrane with low diffusion, similarly to BCRs in antigen-stimulated normal B cells. Somatic mutations affecting the immunoreceptor tyrosine-based activation motif (ITAM) signalling modules6 of CD79B and CD79A were detected frequently in ABC DLBCL biopsy samples but rarely in other DLBCLs and never in Burkitt’s lymphoma or mucosa-associated lymphoid tissue lymphoma. In 18% of ABC DLBCLs, one functionally critical residue of CD79B, the first ITAM tyrosine, was mutated. These mutations increased surface BCR expression and attenuated Lyn kinase, a feedback inhibitor of BCR signalling. These findings establish chronic active BCR signalling as a new pathogenetic mechanism in ABC DLBCL, suggesting several therapeutic strategies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Klein, G. & Klein, E. Conditioned tumorigenicity of activated oncogenes. Cancer Res. 46, 3211–3224 (1986)
Bahler, D. W. & Levy, R. Clonal evolution of a follicular lymphoma: evidence for antigen selection. Proc. Natl Acad. Sci. USA 89, 6770–6774 (1992)
Refaeli, Y. et al. The B cell antigen receptor and overexpression of MYC can cooperate in the genesis of B cell lymphomas. PLoS Biol. 6, e152 (2008)
Ngo, V. N. et al. A loss-of-function RNA interference screen for molecular targets in cancer. Nature 441, 106–110 (2006)
Lenz, G. et al. Oncogenic CARD11 mutations in human diffuse large B cell lymphoma. Science 319, 1676–1679 (2008)
Reth, M. Antigen receptor tail clue. Nature 338, 383–384 (1989)
Alizadeh, A. A. et al. Distinct types of diffuse large B-cell lymphoma identified by gene expression profiling. Nature 403, 503–511 (2000)
Staudt, L. M. & Dave, S. The biology of human lymphoid malignancies revealed by gene expression profiling. Adv. Immunol. 87, 163–208 (2005)
Davis, R. E., Brown, K. D., Siebenlist, U. & Staudt, L. M. Constitutive nuclear factor κB activity is required for survival of activated B cell-like diffuse large B cell lymphoma cells. J. Exp. Med. 194, 1861–1874 (2001)
Dal Porto, J. M. et al. B cell antigen receptor signaling 101. Mol. Immunol. 41, 599–613 (2004)
Chen, L. et al. SYK-dependent tonic B-cell receptor signaling is a rational treatment target in diffuse large B-cell lymphoma. Blood 111, 2230–2237 (2008)
Tolar, P., Hanna, J., Krueger, P. D. & Pierce, S. K. The constant region of the membrane immunoglobulin mediates B cell-receptor clustering and signaling in response to membrane antigens. Immunity 30, 44–55 (2009)
Lam, K. P., Kuhn, R. & Rajewsky, K. In vivo ablation of surface immunoglobulin on mature B cells by inducible gene targeting results in rapid cell death. Cell 90, 1073–1083 (1997)
Kraus, M., Alimzhanov, M. B., Rajewsky, N. & Rajewsky, K. Survival of resting mature B lymphocytes depends on BCR signaling via the Igα/β heterodimer. Cell 117, 787–800 (2004)
Thome, M. CARMA1, BCL-10 and MALT1 in lymphocyte development and activation. Nature Rev. Immunol. 4, 348–359 (2004)
Gordon, M. S., Kanegai, C. M., Doerr, J. R. & Wall, R. Somatic hypermutation of the B cell receptor genes B29 (Igβ, CD79b) and mb1 (Igα, CD79a). Proc. Natl Acad. Sci. USA 100, 4126–4131 (2003)
Gazumyan, A., Reichlin, A. & Nussenzweig, M. C. Igβ tyrosine residues contribute to the control of B cell receptor signaling by regulating receptor internalization. J. Exp. Med. 203, 1785–1794 (2006)
Gauld, S. B. & Cambier, J. C. Src-family kinases in B-cell development and signaling. Oncogene 23, 8001–8006 (2004)
Chan, V. W., Meng, F., Soriano, P., DeFranco, A. L. & Lowell, C. A. Characterization of the B lymphocyte populations in Lyn-deficient mice and the role of Lyn in signal initiation and down-regulation. Immunity 7, 69–81 (1997)
Niiro, H. et al. The B lymphocyte adaptor molecule of 32 kilodaltons (Bam32) regulates B cell antigen receptor internalization. J. Immunol. 173, 5601–5609 (2004)
Ma, H. et al. Visualization of Syk-antigen receptor interactions using green fluorescent protein: differential roles for Syk and Lyn in the regulation of receptor capping and internalization. J. Immunol. 166, 1507–1516 (2001)
Kraus, M., Saijo, K., Torres, R. M. & Rajewsky, K. Ig-α cytoplasmic truncation renders immature B cells more sensitive to antigen contact. Immunity 11, 537–545 (1999)
Torres, R. M. & Hafen, K. A negative regulatory role for Ig-α during B cell development. Immunity 11, 527–536 (1999)
Hantschel, O. et al. The Btk tyrosine kinase is a major target of the Bcr-Abl inhibitor dasatinib. Proc. Natl Acad. Sci. USA 104, 13283–13288 (2007)
Pan, Z. et al. Discovery of selective irreversible inhibitors for Bruton’s tyrosine kinase. ChemMedChem 2, 58–61 (2007)
Jin, S., DiPaola, R. S., Mathew, R. & White, E. Metabolic catastrophe as a means to cancer cell death. J. Cell Sci. 120, 379–383 (2007)
Lam, L. T. et al. Small molecule inhibitors of IκB-kinase are selectively toxic for subgroups of diffuse large B cell lymphoma defined by gene expression profiling. Clin. Cancer Res. 11, 28–40 (2005)
Lenz, G. et al. Stromal gene signatures in large-B-cell lymphomas. N. Engl. J. Med. 359, 2313–2323 (2008)
Blethrow, J., Zhang, C., Shokat, K. M. & Weiss, E. L. Design and use of analog-sensitive protein kinases. Curr. Protocols Mol. Biol. Unit 18.11 10.1002/0471142727.mb1811s66 (2004)
Tolar, P., Sohn, H. W. & Pierce, S. K. The initiation of antigen-induced B cell antigen receptor signaling viewed in living cells by fluorescence resonance energy transfer. Nature Immunol. 6, 1168–1176 (2005)
Jiang, S. et al. Chemical genetic transcriptional fingerprinting for selectivity profiling of kinase inhibitors. Assay Drug Dev. Technol. 5, 49–64 (2007)
Stewart, D. M., Kurman, C. C. & Nelson, D. L. Production of monoclonal antibodies to Bruton’s tyrosine kinase. Hybridoma 14, 243–246 (1995)
Acknowledgements
We thank L. Honigberg for PCI-32765, L. Dang for IKK-β inhibitors, S. Ansher for dasatinib, and S. Tohda for the TMD8 cell line. This research was supported by the Intramural Research Program of the National Institutes of Health, the National Cancer Institute, the Center for Cancer Research, the National Institute of Allergy and Infectious Disease, and the National Human Genome Research Institute. P.B.R. was a Howard Hughes Medical Institute-National Institutes of Health Research Scholar.
Author Contributions R.E.D., V.N.N., G.L., P.T., R.M.Y., P.B.R., H.K., L.L. and A.L.S. designed and performed experiments. H.Z., Y.Y. and W.X. performed experiments. G.W., W.X. and J.P. analysed data. J.J. and C.J.T. synthesized reagents. A.R., G.O., H.K.M.-H., R.D.G., J.M.C., N.A.J., L.M.R., E.C., E.S.J., W.H.W., J.D., E.B.S., R.I.F., R.M.B., R.R.T., J.R.C., D.D.W. and W.C.C. supplied samples from patients and reviewed pathological and clinical data. S.K.P. supervised research. L.M.S. designed and supervised research and wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1-15 with Legends. (PDF 545 kb)
Supplementary Tables
This file contains Supplementary Tables 1-3. (PDF 649 kb)
Rights and permissions
About this article
Cite this article
Davis, R., Ngo, V., Lenz, G. et al. Chronic active B-cell-receptor signalling in diffuse large B-cell lymphoma. Nature 463, 88–92 (2010). https://doi.org/10.1038/nature08638
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature08638
This article is cited by
-
The dual HCK/BTK inhibitor KIN-8194 impairs growth and integrin-mediated adhesion of BTKi-resistant mantle cell lymphoma
Leukemia (2024)
-
Nomogram for predicting survival of patients with diffuse large B-cell lymphoma
Annals of Hematology (2024)
-
Neue Therapiestrategien beim primären Lymphom des Zentralnervensystems
Der Nervenarzt (2024)
-
Molecular complexity of diffuse large B-cell lymphoma: a molecular perspective and therapeutic implications
Journal of Applied Genetics (2024)
-
The progress of novel strategies on immune-based therapy in relapsed or refractory diffuse large B-cell lymphoma
Experimental Hematology & Oncology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.