Abstract
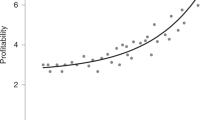
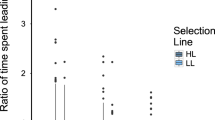
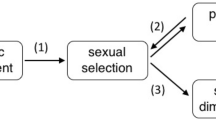
Male animals are typically more elaborately ornamented than females1. Classic sexual selection theory notes that because sperm are cheaper to produce than eggs2, and because males generally compete more intensely for reproductive opportunities and invest less in parental care than females3, males can obtain greater fitness benefits from mating multiply2,4. Therefore, sexual selection typically results in male-biased sex differences in secondary sexual characters1,4. This generality has recently been questioned, because in cooperatively breeding vertebrates, the strength of selection on traits used in intrasexual competition for access to mates (sexual selection1,4) or other resources linked to reproduction (social selection5,6) is similar in males and females7,8. Because selection is acting with comparable intensity in both sexes in cooperatively breeding species, the degree of sexual dimorphism in traits used in intrasexual competition should be reduced in cooperative breeders6. Here we use the socially diverse African starlings (Sturnidae) to demonstrate that the degree of sexual dimorphism in plumage and body size is reduced in cooperatively breeding species as a result of increased selection on females for traits that increase access to reproductive opportunities, other resources, or higher social status. In cooperative breeders such as these, where there is unequal sharing of reproduction (reproductive skew) among females, and where female dominance rank influences access to mates and other resources, intrasexual competition among females may be intense7 and ultimately select for female trait elaboration9. Selection is thereby acting with different intensities on males and females in cooperatively versus non-cooperatively breeding species, and female–female interactions in group-living vertebrates will have important consequences for the evolution of female morphological, physiological and behavioural traits.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Darwin, C. The Descent of Man, and Selection in Relation to Sex (Murray, London, 1871)
Bateman, A. J. Intra-sexual selection in Drosophila . Heredity 2, 349–368 (1948)
Trivers, R. L. in Sexual Selection and the Descent of Man 1871–1971 (ed. Campbell, B.) 136–179 (Aldine, 1972)
Andersson, M. Sexual Selection (Princeton Univ. Press, 1994)
West-Eberhard, M. J. Sexual selection, social competition, and evolution. Proc. Am. Phil. Soc. 123, 222–234 (1979)
West-Eberhard, M. J. Sexual selection, social competition, and speciation. Q. Rev. Biol. 58, 155–183 (1983)
Clutton-Brock, T. H. et al. Intrasexual competition and sexual selection in cooperative mammals. Nature 444, 1065–1068 (2006)
Hauber, M. E. & Lacey, E. A. Bateman's principle in cooperatively breeding vertebrates: the effects of non-breeding alloparents on variability in female and male reproductive success. Integr. Comp. Biol. 45, 903–914 (2005)
Clutton-Brock, T. H. Sexual selection in males and females. Science 318, 1882–1885 (2007)
Roughgarden, J., Oishi, M. & Akcay, E. Reproductive social behavior: cooperative games to replace sexual selection. Science 311, 965–969 (2006)
Amundsen, T. Why are female birds ornamented? Trends Ecol. Evol. 15, 149–155 (2000)
Clutton-Brock, T. H. Sexual selection in females. Anim. Behav. 77, 3–11 (2009)
Clutton-Brock, T. H. in Evolution from Molecules to Men 457–481 (Cambridge Univ. Press, 1983)
Arnold, S. J. Bateman's principle and the measurement of sexual selection in plants and animals. Am. Nat. 144, S1–S22 (1994)
Clutton-Brock, T. H. Reproductive Success: Studies of Individual Variation in Contrasting Breeding Systems (Univ. Chicago Press, 1988)
Rubenstein, D. R. Temporal but not spatial environmental variation drives adaptive offspring sex allocation in a plural cooperative breeder. Am. Nat. 170, 155–165 (2007)
Rubenstein, D. R. & Lovette, I. J. Temporal environmental variability drives the evolution of cooperative breeding in birds. Curr. Biol. 17, 1414–1419 (2007)
Rubenstein, D. R. Female extrapair mate choice in a cooperative breeder: trading sex for help and increasing offspring heterozygosity. Proc. R. Soc. Lond. B 274, 1895–1903 (2007)
Wilkinson, R. Social organization and communal breeding in the chestnut-bellied starling (Spreo pulcher). Anim. Behav. 30, 1118–1128 (1982)
Lovette, I. J. & Rubenstein, D. R. A comprehensive molecular phylogeny of the starlings (Aves: Sturnidae) and mockingbirds (Aves: Mimidae): congruent mtDNA and nuclear trees for a cosmopolitan avian radiation. Mol. Phylogenet. Evol. 44, 1031–1056 (2007)
Craig, A. J. F. K. & Hartley, A. H. The arrangement and structure of feather melanin granules as a taxonomic character in African starlings (Sturnidae). Auk 102, 629–632 (1985)
Badyaev, A. V. & Hill, G. E. Avian sexual dichromatism in relation to phylogeny and ecology. Annu. Rev. Ecol. Syst. 34, 27–49 (2003)
Irwin, R. E. The evolution of plumage dichromatism in the New World blackbirds: social selection of female brightness? Am. Nat. 144, 890–907 (1994)
Dloniak, S. M., French, J. A. & Holekamp, K. E. Rank-related maternal effects of androgens on behaviour in wild spotted hyenas. Nature 440, 1190–1193 (2006)
Bulmer, M. G. The effect of selection on genetic variability. Am. Nat. 105, 201–211 (1971)
Lande, R. Quantitative genetic analysis of multivariate evolution, applied to brain:body size allometry. Evolution 33, 402–416 (1979)
Rowe, L. & Houle, D. The lek paradox and the capture of genetic variance by condition dependent traits. Proc. R. Soc. Lond. B 263, 1415–1421 (1996)
Delhey, K. & Peters, A. Quantifying variability of avian colours: are signaling traits more variable. PLoS One 3, e1689 (2008)
Van Homrigh, A., Higgie, M., McGuigan, K. & Blows, M. W. The depletion of genetic variance by sexual selection. Curr. Biol. 17, 528–532 (2007)
Kotiaho, J. S., Simmons, L. W. & Tomkins, J. L. Towards a resolution of the lek paradox. Nature 410, 684–686 (2001)
Acknowledgements
This research was supported by grants from NSF to I.J.L. (DEB-0515981) and a postdoctoral fellowship from the Miller Institute for Basic Research in Science at the University of California, Berkeley to D.R.R. We thank E. Lacey, B. Lyon, B. McCleery, D. I. Rubenstein, R. Safran, A. L. Talaba and the Lacey laboratory for advice and assistance. P. Hohohnle helped conduct and interpret the MIPoD analyses. We acknowledge S. Edwards, J. Trimble (Harvard Museum of Comparative Zoology), R. Prum, K. Zyskowski (Yale Peabody Museum), J. Cracraft, P. Sweet (American Museum of Natural History), N. Rice (Academy of Natural Sciences, Philadelphia), M. Braun, D. James (National Museum of Natural History) and R. Prys-Jones (British Museum) for access to their collections.
Author Contributions D.R.R. conceived the project and analysed the data. D.R.R. and I.J.L. jointly collected the data and prepared the manuscript.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains a Supplementary Discussion, Supplementary Data, Supplementary Methods, Supplementary References and Supplementary Figure 1 with Legend. (PDF 479 kb)
Rights and permissions
About this article
Cite this article
Rubenstein, D., Lovette, I. Reproductive skew and selection on female ornamentation in social species. Nature 462, 786–789 (2009). https://doi.org/10.1038/nature08614
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature08614
This article is cited by
-
Puppies in the problem-solving paradigm: quick males and social females
Animal Cognition (2023)
-
Maladaptive evolution or how a beneficial mutation may get lost due to nepotism
Communications Biology (2022)
-
Is this love? Sex differences in dog-owner attachment behavior suggest similarities with adult human bonds
Animal Cognition (2022)
-
Female ornamentation does not predict aggression in a tropical songbird
Behavioral Ecology and Sociobiology (2022)
-
Sex differences in the behavioral responses of dogs exposed to human chemosignals of fear and happiness
Animal Cognition (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.