Abstract
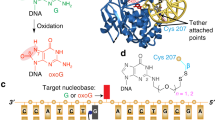
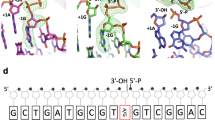
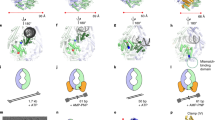
How living systems detect the presence of genotoxic damage embedded in a million-fold excess of undamaged DNA is an unresolved question in biology. Here we have captured and structurally elucidated a base-excision DNA repair enzyme, MutM, at the stage of initial encounter with a damaged nucleobase, 8-oxoguanine (oxoG), nested within a DNA duplex. Three structures of intrahelical oxoG-encounter complexes are compared with sequence-matched structures containing a normal G base in place of an oxoG lesion. Although the protein–DNA interfaces in the matched complexes differ by only two atoms—those that distinguish oxoG from G—their pronounced structural differences indicate that MutM can detect a lesion in DNA even at the earliest stages of encounter. All-atom computer simulations show the pathway by which encounter of the enzyme with the lesion causes extrusion from the DNA duplex, and they elucidate the critical free energy difference between oxoG and G along the extrusion pathway.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
Primary accessions
Protein Data Bank
Data deposits
Atomic coordinates and structure factors for the reported crystal structures have been deposited with the Protein Data Bank under accession codes 3GPY (LRC3), 3GO8 (EC3), 3GP1 (EC3V222P), 3GPP (EC3T224P), 3GPU (EC4), 3GPX (IC4), 3GQ4 (LRC5), 3GQ3(EC5) and 3GQ5 (IC5).
References
Lindahl, T. Instability and decay of the primary structure of DNA. Nature 362, 709–715 (1993)
Loeb, L. A. A mutator phenotype in cancer. Cancer Res. 61, 3230–3239 (2001)
Barnes, D. E. & Lindahl, T. Repair and genetic consequences of endogenous DNA base damage in mammalian cells. Annu. Rev. Genet. 38, 445–476 (2004)
Fromme, J. C. & Verdine, G. L. Base excision repair. Adv. Protein Chem. 69, 1–41 (2004)
Grollman, A. P. & Moriya, M. Mutagenesis by 8-oxoguanine: an enemy within. Trends Genet. 9, 246–249 (1993)
Shibutani, S., Takeshita, M. & Grollman, A. P. Insertion of specific bases during DNA synthesis past the oxidation-damaged base 8-oxodG. Nature 349, 431–434 (1991)
Lipscomb, L. A. et al. X-ray structure of a DNA decamer containing 7,8-dihydro-8-oxoguanine. Proc. Natl Acad. Sci. USA 92, 719–723 (1995)
Oda, Y. et al. NMR studies of a DNA containing 8-hydroxydeoxyguanosine. Nucleic Acids Res. 19, 1407–1412 (1991)
Plum, G. E., Grollman, A. P., Johnson, F. & Breslauer, K. J. Influence of the oxidatively damaged adduct 8-oxodeoxyguanosine on the conformation, energetics, and thermodynamic stability of a DNA duplex. Biochemistry 34, 16148–16160 (1995)
Bowman, B. R., Lee, S., Wang, S. & Verdine, G. L. Structure of the E. coli DNA glycosylase AlkA bound to the ends of duplex DNA: a system for the structure determination of lesion-containing DNA. Structure 16, 1166–1174 (2008)
Fromme, J. C. & Verdine, G. L. DNA lesion recognition by the bacterial repair enzyme MutM. J. Biol. Chem. 278, 51543–51548 (2003)
Bruner, S. D., Norman, D. P. G. & Verdine, G. L. Structural basis for recognition and repair of the endogenous mutagen 8-oxoguanine in DNA. Nature 403, 859–866 (2000)
Zharkov, D. O. Base excision DNA repair. Cell. Mol. Life Sci. 65, 1544–1565 (2008)
Banerjee, A., Santos, W. L. & Verdine, G. L. Structure of a DNA glycosylase searching for lesions. Science 311, 1153–1157 (2006)
Hsu, G. W., Ober, M., Carell, T. & Beese, L. S. Error-prone replication of oxidatively damaged DNA by a high-fidelity DNA polymerase. Nature 431, 217–221 (2004)
Hu, J., Ma, A. & Dinner, A. R. A two-step nucleotide-flipping mechanism enables kinetic discrimination of DNA lesions by AGT. Proc. Natl Acad. Sci. USA 105, 4615–4620 (2008)
Priyakumar, U. D. & Mackerell, A. D. NMR imino proton exchange experiments on duplex DNA primarily monitor the opening of purine bases. J. Am. Chem. Soc. 128, 678–679 (2006)
Banavali, N. K. & MacKerell, A. D. Free energy and structural pathways of base flipping in a DNA GCGC containing sequence. J. Mol. Biol. 319, 141–160 (2002)
Cheng, X. et al. Dynamic behavior of DNA base pairs containing 8-oxoguanine. J. Am. Chem. Soc. 127, 13906–13918 (2005)
Yang, W. Poor base stacking at DNA lesions may initiate recognition by many repair proteins. DNA Repair (Amst.) 5, 654–666 (2006)
Blainey, P. C., van Oijen, A. M., Banerjee, A., Verdine, G. L. & Xie, X. S. A base-excision DNA-repair protein finds intrahelical lesion bases by fast sliding in contact with DNA. Proc. Natl Acad. Sci. USA 103, 5752–5757 (2006)
Minetti, C. A. et al. Energetics of lesion recognition by a DNA repair protein: thermodynamic characterization of formamidopyrimidine-glycosylase (Fpg) interactions with damaged DNA duplexes. J. Mol. Biol. 328, 1047–1060 (2003)
Fedorova, O. S. et al. Stopped-flow kinetic studies of the interaction between Escherichia coli Fpg protein and DNA substrates. Biochemistry 41, 1520–1528 (2002)
Ishchenko, A. A. et al. Thermodynamic, kinetic, and structural basis for recognition and repair of 8-oxoguanine in DNA by Fpg protein from Escherichia coli . Biochemistry 41, 7540–7548 (2002)
Parker, J. B. et al. Enzymatic capture of an extrahelical thymine in the search for uracil in DNA. Nature 449, 433–437 (2007)
MacKerell, A. D. et al. All-atom empirical potential for molecular modeling and dynamics studies of proteins. J. Phys. Chem. B 102, 3586–3616 (1998)
Mackerell, A. D. & Banavali, N. K. All-atom empirical force field for nucleic acids: II. Application to molecular dynamics simulations of DNA and RNA in solution. J. Comput. Chem. 21, 105–120 (2000)
Foloppe, N. & Mackerell, A. D. All-atom empirical force field for nucleic acids: I. Parameter optimization based on small molecule and condensed phase macromolecular target data. J. Comput. Chem. 21, 86–104 (2000)
Jorgensen, W. L., Chandrasekhar, J., Madura, J. D., Impey, R. W. & Klein, M. L. Comparison of simple potential functions for simulating liquid water. J. Chem. Phys. 79, 926–935 (1983)
Darden, T., York, D. & Pedersen, L. Particle mesh Ewald: an N·log(N) method for Ewald sums in large systems. J. Chem. Phys. 98, 10089 (1993)
van der Vaart, A. & Karplus, M. Simulation of conformational transitions by the restricted perturbation-targeted molecular dynamics method. J. Chem. Phys. 122, 114903 (2005)
Paci, E. & Karplus, M. Forced unfolding of fibronectin type 3 modules: an analysis by biased molecular dynamics simulations. J. Mol. Biol. 288, 441–459 (1999)
Hu, J., Ma, A. & Dinner, A. R. Bias annealing: A method for obtaining transition paths de novo . J. Chem. Phys. 125, 114101 (2006)
Torrie, G. M. & Valleau, J. P. Nonphysical sampling distributions in Monte Carlo free-energy estimation: umbrella sampling. J. Comput. Phys. 23, 187 (1977)
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997)
Brünger, A. T. et al. Crystallography and NMR system (CNS): a new software system for macromolecular structure determination. Acta Crystallogr. D 54, 905–921 (1998)
Emsley, P. & Cowan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004)
Winn, M. D., Isupov, M. N. & Murshudov, G. N. Use of TLS parameters to model anisotropic displacements in macromolecular refinement. Acta Crystallogr. D 57, 122–133 (2001)
Painter, J. & Merritt, E. A. Optimal description of a protein structure in terms of multiple groups undergoing TLS motion. Acta Crystallogr. D Biol. Crystallogr. 62, 439–450 (2006)
Brooks, B. R. et al. CHARMM: a program for macromolecular energy, minimization, and dynamics calculations. J. Comput. Chem. 4, 187 (1983)
Macke, T. & Case, D. A. in Molecular modeling of nucleic acids (eds Leontes, N. B. & SantaLucia, J. Jr) 379–393 (American Chemical Society, 1998)
Allen, M. P. & Tildesley, D. J. Computer simulation of liquids (Oxford Univ. Press, 1989)
Ryckaert, J. P., Ciccotti, G. & Berendsen, H. J. C. Numerical integration of the Cartesian equations of motion of a system with constraints: molecular dynamics of n-alkanes. J. Comput. Phys. 23, 327–341 (1977)
Berendsen, H. J. C., Postma, J. P. M., Van Gunsteren, W. F., DiNola, A. & Haak, J. R. Molecular dynamics with coupling to an external bath. J. Chem. Phys. 81, 3684 (1984)
Schlitter, J., Engels, M., Krueger, P., Jacoby, E. & Wollmer, A. Targeted molecular dynamics simulation of conformational change-application to the T↔R transition in insulin. Mol. Simul. 10, 291–308 (1993)
Banavali, N. K. & MacKerell, A. D. Free energy and structural pathways of base flipping in a DNA GCGC containing sequence. J. Mol. Biol. 319, 141–160 (2002)
Kumar, S., Bouzida, D., Swendsen, R. H., Kollman, P. A. & Rosenberg, J. M. The weighted histogram analysis method for free-energy calculations on biomolecules. I. The method. J. Comput. Chem. 13, 1011–1021 (1992)
Rajamani, R., Naidoo, K. J. & Gao, J. Implementation of an adaptive umbrella sampling method for the calculation of multidimensional potential of mean force of chemical reactions in solution. J. Comput. Chem. 24, 1775–1781 (2003)
Acknowledgements
This work was supported by grants from the NIH: GM044853, GM047467, CA100742 (G.L.V.) and GM030804 (M.K.). Y.Q. is supported by a predoctoral fellowship from the National Science Foundation; M.C.S. by a predoctoral fellowship from the Howard Hughes Medical Institute; K.N. by a postdoctoral fellowship from National Cancer Center. We thank the staff of the NSLS, APS and CHESS synchrotron facilities. These experiments made use of the computing facilities at NERSC and Harvard FAS. We are grateful to J. Jin for experimental assistance and J. Pu, V. Ovchinnikov and other members of the Karplus and Verdine groups for helpful advice.
Author Contributions Y.Q., M.C.S., K.N. and A.B. contributed equally to the study. A.B., S.J. and M.C.S. cloned the constructs. A.B., M.C.S. and S.J. performed the biochemical and FP assays. A.B., S.J., M.C.S. and Y.Q. purified, crystallized and collected X-ray diffraction data and solved structures (A.B., S.J.: EC4, EC5; M.C.S.: EC3, EC3V222P, EC3T224P, IC4, LRC5; Y.Q.: LRC3, EC3T224P, EC5, IC5, LRC5). A.B. and G.L.V. designed the trapping strategy and crystallographic studies. K.N. and M.K. designed the computational studies, which K.N. then performed. A.B., M.C.S., Y.Q., K.N., G.L.V. and M.K. analysed data and wrote the paper. G.L.V. and M.K. directed the research. All authors discussed the results and commented on the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Information
This file contains Supplementary Notes, Supplementary References, Supplementary Figures 1-21 with Legends and Supplementary Tables S1- S3. (PDF 9032 kb)
Supplementary Movie 1
This movie file shows the MutM-assisted base extrusion pathway calculated using the EC4 crystal structure. The target base is oxoG. (MOV 6863 kb)
Supplementary Movie 2
This movie file shows the MutM-assisted base extrusion pathway calculated using the IC4 crystal structure. The target base is G. (MOV 6548 kb)
Supplementary Movie 3
This movie file shows the MutM-assisted base extrusion pathway calculated using the EC5 crystal structure. The target base is oxoG. (MOV 6869 kb)
Supplementary Movie 4
This movie file shows the MutM-assisted base extrusion pathway calculated using the IC5 crystal structure. The target base is G. (MOV 6616 kb)
Rights and permissions
About this article
Cite this article
Qi, Y., Spong, M., Nam, K. et al. Encounter and extrusion of an intrahelical lesion by a DNA repair enzyme. Nature 462, 762–766 (2009). https://doi.org/10.1038/nature08561
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature08561
This article is cited by
-
The trajectory of intrahelical lesion recognition and extrusion by the human 8-oxoguanine DNA glycosylase
Nature Communications (2020)
-
Molecular dynamics simulation of the opposite-base preference and interactions in the active site of formamidopyrimidine-DNA glycosylase
BMC Structural Biology (2018)
-
Both DNA global deformation and repair enzyme contacts mediate flipping of thymine dimer damage
Scientific Reports (2017)
-
The DNA glycosylase AlkD uses a non-base-flipping mechanism to excise bulky lesions
Nature (2015)
-
Duplex interrogation by a direct DNA repair protein in search of base damage
Nature Structural & Molecular Biology (2012)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.