Abstract
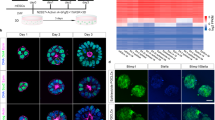
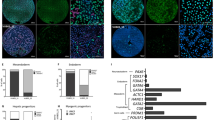
The pluripotent state, which is first established in the primitive ectoderm cells of blastocysts, is lost progressively and irreversibly during subsequent development1. For example, development of post-implantation epiblast cells from primitive ectoderm involves significant transcriptional and epigenetic changes, including DNA methylation and X chromosome inactivation2, which create a robust epigenetic barrier and prevent their reversion to a primitive-ectoderm-like state. Epiblast cells are refractory to leukaemia inhibitory factor (LIF)–STAT3 signalling, but they respond to activin/basic fibroblast growth factor to form self-renewing epiblast stem cells (EpiSCs), which exhibit essential properties of epiblast cells3,4 and that differ from embryonic stem (ES) cells derived from primitive ectoderm5. Here we show reprogramming of advanced epiblast cells from embryonic day 5.5–7.5 mouse embryos with uniform expression of N-cadherin and inactive X chromosome to ES-cell-like cells (rESCs) in response to LIF–STAT3 signalling. Cultured epiblast cells overcome the epigenetic barrier progressively as they proceed with the erasure of key properties of epiblast cells, resulting in DNA demethylation, X reactivation and expression of E-cadherin. The accompanying changes in the transcriptome result in a loss of phenotypic and epigenetic memory of epiblast cells. Using this approach, we report reversion of established EpiSCs to rESCs. Moreover, unlike epiblast and EpiSCs, rESCs contribute to somatic tissues and germ cells in chimaeras. Further studies may reveal how signalling-induced epigenetic reprogramming may promote reacquisition of pluripotency.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Gardner, R. L. & Rossant, J. Investigation of the fate of 4–5 day post-coitum mouse inner cell mass cells by blastocyst injection. J. Embryol. Exp. Morphol. 52, 141–152 (1979)
Surani, M. A., Hayashi, K. & Hajkova, P. Genetic and epigenetic regulators of pluripotency. Cell 128, 747–762 (2007)
Tesar, P. J. et al. New cell lines from mouse epiblast share defining features with human embryonic stem cells. Nature 448, 196–199 (2007)
Brons, I. G. et al. Derivation of pluripotent epiblast stem cells from mammalian embryos. Nature 448, 191–195 (2007)
Evans, M. J. & Kaufman, M. H. Establishment in culture of pluripotential cells from mouse embryos. Nature 292, 154–156 (1981)
Yeom, Y. I. et al. Germline regulatory element of Oct-4 specific for the totipotent cycle of embryonal cells. Development 122, 881–894 (1996)
Chen, X. et al. Integration of external signaling pathways with the core transcriptional network in embryonic stem cells. Cell 133, 1106–1117 (2008)
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006)
Niwa, H. How is pluripotency determined and maintained? Development 134, 635–646 (2007)
Rathjen, J., Washington, J. M., Bettess, M. D. & Rathjen, P. D. Identification of a biological activity that supports maintenance and proliferation of pluripotent cells from the primitive ectoderm of the mouse. Biol. Reprod. 69, 1863–1871 (2003)
Chou, Y. F. et al. The growth factor environment defines distinct pluripotent ground states in novel blastocyst-derived stem cells. Cell 135, 449–461 (2008)
Takagi, N., Sugawara, O. & Sasaki, M. Regional and temporal changes in the pattern of X-chromosome replication during the early post-implantation development of the female mouse. Chromosoma 85, 275–286 (1982)
Silva, J. et al. Establishment of histone h3 methylation on the inactive X chromosome requires transient recruitment of Eed-Enx1 polycomb group complexes. Dev. Cell 4, 481–495 (2003)
Chuva de Sousa Lopes, S. M. et al. X chromosome activity in mouse XX primordial germ cells. PLoS Genet. 4, e30 (2008)
Plath, K. et al. Role of histone H3 lysine 27 methylation in X inactivation. Science 300, 131–135 (2003)
Mikkelsen, T. S. et al. Dissecting direct reprogramming through integrative genomic analysis. Nature 454, 49–55 (2008)
Maherali, N. et al. Directly reprogrammed fibroblasts show global epigenetic remodeling and widespread tissue contribution. Cell Stem Cell 1, 55–70 (2007)
Tada, M., Takahama, Y., Abe, K., Nakatsuji, N. & Tada, T. Nuclear reprogramming of somatic cells by in vitro hybridization with ES cells. Curr. Biol. 11, 1553–1558 (2001)
Bao, S. et al. Initiation of epigenetic reprogramming of the X chromosome in somatic nuclei transplanted to a mouse oocyte. EMBO Rep. 6, 748–754 (2005)
Hayashi, K., Lopes, S. M., Tang, F. & Surani, M. A. Dynamic equilibrium and heterogeneity of mouse pluripotent stem cells with distinct functional and epigenetic states. Cell Stem Cell 3, 391–401 (2008)
Ohinata, Y. et al. A signaling principle for the specification of the germ cell lineage in mice. Cell 137, 571–584 (2009)
Shovlin, T. C., Durcova-Hills, G., Surani, A. & McLaren, A. Heterogeneity in imprinted methylation patterns of pluripotent embryonic germ cells derived from pre-migratory mouse germ cells. Dev. Biol. 313, 674–681 (2008)
Meissner, A. et al. Genome-scale DNA methylation maps of pluripotent and differentiated cells. Nature 454, 766–770 (2008)
Guo, G. et al. Klf4 reverts developmentally programmed restriction of ground state pluripotency. Development 136, 1063–1069 (2009)
Hanna, J. et al. Metastable pluripotent states in NOD-mouse-derived ESCs. Cell Stem Cell 4, 513–524 (2009)
Thomson, J. A. et al. Embryonic stem cell lines derived from human blastocysts. Science 282, 1145–1147 (1998)
Silva, J. et al. Promotion of reprogramming to ground state pluripotency by signal inhibition. PLoS Biol. 6, e253 (2008)
Wernig, M. et al. A drug-inducible transgenic system for direct reprogramming of multiple somatic cell types. Nature Biotechnol. 26, 916–924 (2008)
Hayashi, K. & Surani, M. A. Resetting the epigenome beyond pluripotency in the germline. Cell Stem Cell 4, 493–498 (2009)
Surani, M. A., Durcova-Hills, G., Hajkova, P., Hayashi, K. & Tee, W. W. Germ line, stem cells, and epigenetic reprogramming. Cold Spring Harb. Symp. Quant. Biol. 73, 9–15 (2008)
Hayashi, K. & Surani, M. A. Self-renewing epiblast stem cells exhibit continual delineation of germ cells with epigenetic reprogramming in vitro. Development 136, 3549–3556 (2009)
Acknowledgements
We thank C. Lee for assistance. This work was supported by grants from the Wellcome Trust to M.A.S.
Author Contributions S.B., F.T., X.L. and M.A.S. designed the research project; S.B. and F.T. performed most of the experiments, with contributions from X.L., K.H. and A.G.; microarray analysis was performed by K.L.; S.B., F.T., K.H., A.G. and M.A.S carried out critical assessment of the data; M.A.S. wrote the paper with input from all the authors.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Methods, Supplementary Figures 1-7 with legends, Supplementary Tables 1-6 with legends and Supplementary References. (PDF 3175 kb)
Rights and permissions
About this article
Cite this article
Bao, S., Tang, F., Li, X. et al. Epigenetic reversion of post-implantation epiblast to pluripotent embryonic stem cells . Nature 461, 1292–1295 (2009). https://doi.org/10.1038/nature08534
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08534
This article is cited by
-
c-Jun as a one-way valve at the naive to primed interface
Cell & Bioscience (2023)
-
Elevated retrotransposon activity and genomic instability in primed pluripotent stem cells
Genome Biology (2021)
-
Generation of mouse–human chimeric embryos
Nature Protocols (2021)
-
BMP4 resets mouse epiblast stem cells to naive pluripotency through ZBTB7A/B-mediated chromatin remodelling
Nature Cell Biology (2020)
-
Derivation of stable embryonic stem cell-like, but transcriptionally heterogenous, induced pluripotent stem cells from non-permissive mouse strains
Mammalian Genome (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.