Abstract
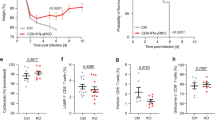
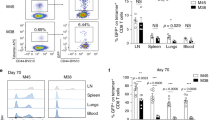
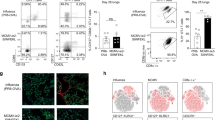
CD4+ T helper cells are well known for their role in providing critical signals during priming of cytotoxic CD8+ T lymphocyte (CTL) responses in vivo. T-cell help is required for the generation of primary CTL responses as well as in promoting protective CD8+ memory T-cell development1. However, the role of CD4 help in the control of CTL responses at the effector stage is unknown. Here we show that fully helped effector CTLs are themselves not self-sufficient for entry into the infected tissue, but rely on the CD4+ T cells to provide the necessary cue. CD4+ T helper cells control the migration of CTL indirectly through the secretion of IFN-γ and induction of local chemokine secretion in the infected tissue. Our results reveal a previously unappreciated role of CD4 help in mobilizing effector CTL to the peripheral sites of infection where they help to eliminate infected cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Williams, M. A. & Bevan, M. J. Effector and memory CTL differentiation. Annu. Rev. Immunol. 25, 171–192 (2007)
Bennett, S. R. et al. Help for cytotoxic-T-cell responses is mediated by CD40 signalling. Nature 393, 478–480 (1998)
Ridge, J. P., Di Rosa, F. & Matzinger, P. A conditioned dendritic cell can be a temporal bridge between a CD4+ T-helper and a T-killer cell. Nature 393, 474–478 (1998)
Schoenberger, S. P., Toes, R. E., van der Voort, E. I., Offringa, R. & Melief, C. J. T-cell help for cytotoxic T lymphocytes is mediated by CD40–CD40L interactions. Nature 393, 480–483 (1998)
Jennings, S. R., Bonneau, R. H., Smith, P. M., Wolcott, R. M. & Chervenak, R. CD4-positive T lymphocytes are required for the generation of the primary but not the secondary CD8-positive cytolytic T lymphocyte response to herpes simplex virus in C57BL/6 mice. Cell. Immunol. 133, 234–252 (1991)
Janssen, E. M. et al. CD4+ T cells are required for secondary expansion and memory in CD8+ T lymphocytes. Nature 421, 852–856 (2003)
Shedlock, D. J. & Shen, H. Requirement for CD4 T cell help in generating functional CD8 T cell memory. Science 300, 337–339 (2003)
Sun, J. C. & Bevan, M. J. Defective CD8 T cell memory following acute infection without CD4 T cell help. Science 300, 339–342 (2003)
Jones, C. A., Taylor, T. J. & Knipe, D. M. Biological properties of herpes simplex virus 2 replication-defective mutant strains in a murine nasal infection model. Virology 278, 137–150 (2000)
Zhao, X. et al. Vaginal submucosal dendritic cells, but not Langerhans cells, induce protective Th1 responses to herpes simplex virus-2. J. Exp. Med. 197, 153–162 (2003)
Smith, C. M. et al. Cognate CD4+ T cell licensing of dendritic cells in CD8+ T cell immunity. Nature Immunol. 5, 1143–1148 (2004)
Mueller, S. N., Heath, W., McLain, J. D., Carbone, F. R. & Jones, C. M. Characterization of two TCR transgenic mouse lines specific for herpes simplex virus. Immunol. Cell Biol. 80, 156–163 (2002)
Stock, A. T. et al. Optimization of TCR transgenic T cells for in vivo tracking of immune responses. Immunol. Cell Biol. 85, 394–396 (2007)
Marshall, D. R. et al. Measuring the diaspora for virus-specific CD8+ T cells. Proc. Natl Acad. Sci. USA 98, 6313–6318 (2001)
Masopust, D., Vezys, V., Marzo, A. L. & Lefrancois, L. Preferential localization of effector memory cells in nonlymphoid tissue. Science 291, 2413–2417 (2001)
Lund, J. M., Hsing, L., Pham, T. T. & Rudensky, A. Y. Coordination of early protective immunity to viral infection by regulatory T cells. Science 320, 1220–1224 (2008)
Reboldi, A. et al. C–C chemokine receptor 6-regulated entry of TH-17 cells into the CNS through the choroid plexus is required for the initiation of EAE. Nature Immunol. 10, 514–523 (2009)
Milligan, G. N. & Bernstein, D. I. Interferon-γ enhances resolution of herpes simplex virus type 2 infection of the murine genital tract. Virology 229, 259–268 (1997)
Iijima, N. et al. Dendritic cells and B cells maximize mucosal Th1 memory response to herpes simplex virus. J. Exp. Med. 205, 3041–3052 (2008)
Thapa, M., Welner, R. S., Pelayo, R. & Carr, D. J. CXCL9 and CXCL10 expression are critical for control of genital herpes simplex virus type 2 infection through mobilization of HSV-specific CTL and NK cells to the nervous system. J. Immunol. 180, 1098–1106 (2008)
Iijima, N., Linehan, M. M., Saeland, S. & Iwasaki, A. Vaginal epithelial dendritic cells renew from bone marrow precursors. Proc. Natl Acad. Sci. USA 104, 19061–19066 (2007)
Hancock, W. W. et al. Requirement of the chemokine receptor CXCR3 for acute allograft rejection. J. Exp. Med. 192, 1515–1520 (2000)
Sato, A. & Iwasaki, A. Induction of antiviral immunity requires Toll-like receptor signaling in both stromal and dendritic cell compartments. Proc. Natl Acad. Sci. USA 101, 16274–16279 (2004)
Fontenot, J. D. et al. Regulatory T cell lineage specification by the forkhead transcription factor Foxp3. Immunity 22, 329–341 (2005)
Iwasaki, A. & Kelsall, B. L. Localization of distinct Peyer’s patch dendritic cell subsets and their recruitment by chemokines macrophage inflammatory protein (MIP)-3α, MIP-3β, and secondary lymphoid organ chemokine. J. Exp. Med. 191, 1381–1394 (2000)
Acknowledgements
We thank F. R. Carbone and W. R. Heath for the gBT-I transgenic mouse, A. Rudensky for the foxp3 knock-in mouse, R. Medzhitov and J. M. Thompson for critical reading of the manuscript, and N. Iijima for technical assistance. This work is supported by NIH grants to A.I. (AI054359 and AI062428) and to C.G. (AI39759 and HL51366). Y.N. was a Japan Society for the Promotion of Science fellow. A.I. is a recipient of the Burroughs Wellcome Investigators in Pathogenesis of Infectious Disease.
Author Contributions Experiments were conceived and designed by Y.N. and A.I. Experiments were performed by Y.N. Data were analysed by Y.N. and A.I. The paper was written by Y.N. and A.I. C.G. and B.L. provided CXCR3-knockout mice and discussed the manuscript.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1-13 with Legends. (PDF 3307 kb)
Rights and permissions
About this article
Cite this article
Nakanishi, Y., Lu, B., Gerard, C. et al. CD8+ T lymphocyte mobilization to virus-infected tissue requires CD4+ T-cell help. Nature 462, 510–513 (2009). https://doi.org/10.1038/nature08511
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08511
This article is cited by
-
Nanotechnology of inhalable vaccines for enhancing mucosal immunity
Drug Delivery and Translational Research (2024)
-
Focus on T cell exhaustion: new advances in traditional Chinese medicine in infection and cancer
Chinese Medicine (2023)
-
Microbial peptides activate tumour-infiltrating lymphocytes in glioblastoma
Nature (2023)
-
Characterization of immunomodulatory factors and cells in bronchoalveolar lavage fluid for immune checkpoint inhibitor-related pneumonitis
Journal of Cancer Research and Clinical Oncology (2023)
-
Pan-cancer analysis combined with experiments explores the oncogenic role of spindle apparatus coiled-coil protein 1 (SPDL1)
Cancer Cell International (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.