Abstract
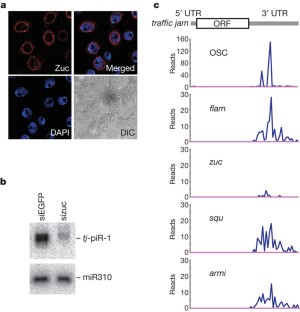
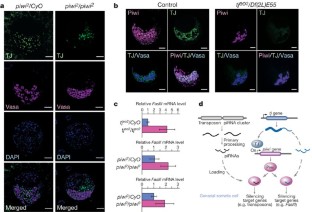
PIWI-interacting RNAs (piRNAs) silence retrotransposons in Drosophila germ lines by associating with the PIWI proteins Argonaute 3 (AGO3), Aubergine (Aub) and Piwi1,2. piRNAs in Drosophila are produced from intergenic repetitive genes and piRNA clusters by two systems: the primary processing pathway and the amplification loop1,2,3,4,5,6,7. The amplification loop occurs in a Dicer-independent, PIWI-Slicer-dependent manner3,4,8. However, primary piRNA processing remains elusive. Here we analysed piRNA processing in a Drosophila ovarian somatic cell line where Piwi, but not Aub or AGO3, is expressed; thus, only the primary piRNAs exist. In addition to flamenco, a Piwi-specific piRNA cluster3, traffic jam (tj)9, a large Maf gene, was determined as a new piRNA cluster. piRNAs arising from tj correspond to the untranslated regions of tj messenger RNA and are sense-oriented. piRNA loading on to Piwi may occur in the cytoplasm. zucchini10, a gene encoding a putative cytoplasmic nuclease, is required for tj-derived piRNA production. In tj and piwi mutant ovaries, somatic cells fail to intermingle with germ cells and Fasciclin III is overexpressed. Loss of tj abolishes Piwi expression in gonadal somatic cells. Thus, in gonadal somatic cells, tj gives rise simultaneously to two different molecules: the TJ protein, which activates Piwi expression, and piRNAs, which define the Piwi targets for silencing.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Aravin, A. A., Hannon, G. J. & Brennecke, J. The Piwi-piRNA pathway provides an adaptive defense in the transposon arms race. Science 318, 761–764 (2007)
Siomi, H. & Siomi, M. C. On the road to reading the RNA-interference code. Nature 457, 396–404 (2009)
Brennecke, J. et al. Discrete small RNA-generating loci as master regulators of transposon activity in Drosophila. Cell 128, 1089–1103 (2007)
Gunawardane, L. S. et al. A slicer-mediated mechanism for repeat-associated siRNA 5′ end formation in Drosophila. Science 315, 1587–1590 (2007)
Li, C. et al. Collapse of germline piRNAs in the absent of Argonaute3 reveals somatic piRNAs in flies. Cell 137, 509–521 (2009)
Malone, C. et al. Specialized piRNA pathways act in germline and somatic tissues of the Drosophila ovary. Cell 137, 522–535 (2009)
Lau, N. et al. Abundant primary piRNAs, endo-siRNAs and microRNAs in a Drosophila ovary cell line. Genome Res. 10.1101/gr.094896.109 (14 July 2009)
Vagin, V. V. et al. A distinct small RNA pathway silences selfish genetic elements in the germline. Science 313, 320–324 (2006)
Li, M. A., Alls, J. D., Avancini, R. M., Koo, K. & Godt, D. The large Maf factor Traffic Jam controls gonad morphogenesis in Drosophila.. Nature Cell Biol. 5, 994–1000 (2003)
Pane, A., Wehr, K. & Schupbach, T. zucchini and squash encode two putative nucleases required for rasiRNA production in the Drosophila germilne. Dev. Cell 12, 851–862 (2007)
Cox, D. N. et al. A novel class of evolutionarily conserved genes defined by piwi are essential for stem cell self-renewal. Genes Dev. 12, 3715–3727 (1998)
Harris, A. N. & Macdonald, P. M. aubergine encodes a Drosophila polar granule component required for pole cell formation and related to eIF2C. Development 128, 2823–2832 (2001)
Niki, Y., Yamaguchi, T. & Mahowald, A. P. Establishment of stable cell lines of Drosophila germ-line stem cells. Proc. Natl Acad. Sci. USA 103, 16325–16330 (2006)
Saito, K. et al. Specific association of Piwi with rasiRNAs derived from retrotransposon and heterochromatic regions in the Drosophila genome. Genes Dev. 20, 2214–2222 (2006)
Cox, D. N., Chao, A. & Lin, H. piwi encodes a nucleoplasmic factor whose activity modulates the number and division rate of germline stem cells. Genes Dev. 127, 503–514 (2000)
Nishida, M. K. et al. Gene silencing mechanisms mediated by Aubergine piRNA complexes in Drosophila male gonad. RNA 13, 1911–1922 (2007)
Saito, K. et al. Pimet, the Drosophila homolog of HEN1, mediates 2'-O-methylation of Piwi-interacting RNAs at their 3′ ends. Genes Dev. 21, 1603–1608 (2007)
Horwich, M. D. et al. The Drosophila RNA methyltransferase, DmHen1, modifies germline piRNAs and single-stranded siRNAs in RISC. Curr. Biol. 17, 1265–1272 (2007)
Yin, H. & Lin, H. An epigenetic activation role of Piwi and a Piwi-associated piRNA in Drosophila melanogaster. Nature 450, 304–308 (2007)
Czech, B. et al. An endogenous small interfering RNA pathway in Drosophila. Nature 453, 798–802 (2008)
Arama, E., Dickman, D., Kimchie, Z., Shearn, A. & Lev, Z. Mutations in the beta-propeller domain of the Drosophila brain tumor (brat) protein induce neoplasm in the larval brain. Oncogene 19, 3706–3716 (2000)
Rogers, G. C. et al. Two mitotic kinesins cooperate to drive sister chromatid separation during anaphase. Nature 427, 364–370 (2004)
Lin, H. & Spradling, A. C. A novel group of pumilio mutations affects the asymmetric division of germline stem cells in the Drosophila ovary. Development 124, 2463–2476 (1997)
Kataoka, K. Multiple mechanisms and functions of Maf transcriptional factors in the regulation of tissue-specific genes. J. Biochem. 141, 775–781 (2007)
Kawamura, Y. et al. Drosophila endogenous small RNAs bind to Argonaute 2 in somatic cells. Nature 453, 793–797 (2008)
Kitadate, Y., Shigenobu, S., Arita, K. & Kobayashi, S. Boss/Sev signaling from germline to soma restricts germline-stem-cell-niche formation in the anterior region of Drosophila male gonads. Dev. Cell 13, 151–159 (2007)
Miyoshi, K., Tsukumo, H., Nagami, T., Siomi, H. & Siomi, M. C. Slicer function of Drosophila Argonautes and its involvement in RISC formation. Genes Dev. 19, 2837–2848 (2005)
Wilson, R. J., Goodman, J. L., Strelets, V. B. & FlyBase Consortium FlyBase: integration and improvements to query tools. Nucleic Acids Res. 36, D588–D593 (2008)
Mituyama, T. et al. The Functional RNA Database 3.0: databases to support mining and annotation of functional RNAs. Nucleic Acids Res. 37, D89–D92 (2009)
Crooks, G. E., Hon, G., Chandonia, J. M. & Brenner, S. E. WebLogo, a sequence logo generator. Genome Res. 14, 1188–1190 (2004)
Acknowledgements
We thank Y. Niki, D. Godt, H. Lin, E. Matunis, S. Kobayashi, Y. Kitadate, Y. Kageyama and H. Sano for providing reagents. We also thank Bloomington and Kyoto Drosophila Stock Center for the supply of Drosophila strains. We thank K. Yamada, E. Hattori, K. M. Nishida and T. N. Okada for technical assistance; S. Takahashi and K. Kataoka for discussions and suggestions; and other members of the Siomi laboratory for discussions and comments on the manuscript. We also thank K. Greer and D. McGowan for encouragement. This work was supported by MEXT grants to H.S. and NEDO (New Energy and Industrial Technology Development Organization) grants to M.C.S., T.M. and K.A. M.C.S. is supported by CREST from JST. M.C.S. is Associate Professor of Global COE for Human Metabolomics Systems Biology by MEXT.
Author Contributions K.S., S.I., Y.K. and H.K. conducted biochemical experiments. T.M., Y.O., E.S. and K.A. performed bioinformatics. K.S., T.M., H.S. and M.C.S designed experiments, interpreted data and prepared the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Information
This file contains Supplementary Figures S1-S10 with Legends and Supplementary Tables 1-2. (PDF 9481 kb)
Rights and permissions
About this article
Cite this article
Saito, K., Inagaki, S., Mituyama, T. et al. A regulatory circuit for piwi by the large Maf gene traffic jam in Drosophila. Nature 461, 1296–1299 (2009). https://doi.org/10.1038/nature08501
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08501
This article is cited by
-
HRDE-2 drives small RNA specificity for the nuclear Argonaute protein HRDE-1
Nature Communications (2024)
-
Regulation of insect behavior by non-coding RNAs
Science China Life Sciences (2024)
-
Themes and variations on piRNA-guided transposon control
Mobile DNA (2023)
-
Emerging roles and functional mechanisms of PIWI-interacting RNAs
Nature Reviews Molecular Cell Biology (2023)
-
Transposable element expansion and low-level piRNA silencing in grasshoppers may cause genome gigantism
BMC Biology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.