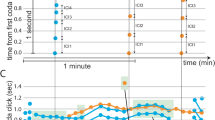
Abstract
Social interactions depend on individuals recognizing each other, and in this context many organisms use chemical signals to indicate species and sex1. Cuticular hydrocarbon signals are used by insects, including Drosophila melanogaster, to distinguish conspecific individuals from others1,2,3. These chemicals also contribute to intraspecific courtship and mating interactions1,2,3. However, the possibility that sex and species identification are linked by common chemical signalling mechanisms has not been formally tested. Here we provide direct evidence that a single compound is used to communicate female identity among D. melanogaster, and to define a reproductive isolation barrier between D. melanogaster and sibling species. A transgenic manipulation eliminated cuticular hydrocarbons by ablating the oenocytes, specialized cells required for the expression of these chemical signals. The resulting oenocyte-less (oe-) females elicited the normal repertoire of courtship behaviours from males, but were actually preferred over wild-type females by courting males. In addition, wild-type males attempted to copulate with oe- males. Thus, flies lacking hydrocarbons are a sexual hyperstimulus. Treatment of virgin females with the aversive male pheromone cis-vaccenyl acetate (cVA) significantly delayed mating of oe- females compared to wild-type females. This difference was eliminated when oe- females were treated with a blend of cVA and the female aphrodisiac (7Z,11Z)-heptacosadiene (7,11-HD), showing that female aphrodisiac compounds can attenuate the effects of male aversive pheromones. 7,11-HD also was shown to have a crucial role in heterospecific encounters. Specifically, the species barrier was lost because males of other Drosophila species courted oe- D. melanogaster females, and D. simulans males consistently mated with them. Treatment of oe- females with 7,11-HD restored the species barrier, showing that a single compound can confer species identity. These results identify a common mechanism for sexual and species recognition regulated by cuticular hydrocarbons.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Wyatt, T. D. Pheromones and Animal Behaviour: Communication by Smell and Taste (Cambridge Univ. Press, 2003)
Jallon, J. M. A few chemical words exchanged by Drosophila during courtship and mating. Behav. Genet. 14, 441–478 (1984)
Ferveur, J. F. Cuticular hydrocarbons: their evolution and roles in Drosophila pheromonal communication. Behav. Genet. 35, 279–295 (2005)
Coyne, J. A., Crittenden, A. P. & Mah, K. Genetics of a pheromonal difference contributing to reproductive isolation in Drosophila . Science 265, 1461–1464 (1994)
Savarit, F., Sureau, G., Cobb, M. & Ferveur, J. F. Genetic elimination of known pheromones reveals the fundamental chemical bases of mating and isolation in Drosophila . Proc. Natl Acad. Sci. USA 96, 9015–9020 (1999)
Grillet, M., Dartevelle, L. & Ferveur, J. F. A Drosophila male pheromone affects female sexual receptivity. Proc. Biol. Sci. 273, 315–323 (2006)
Lacaille, F. et al. An inhibitory sex pheromone tastes bitter for Drosophila males. PLoS One 2, e661 (2007)
Antony, C., Davis, T. L., Carlson, D. A., Pechine, J. M. & Jallon, J. M. Compared behavioral responses of male Drosophila melanogaster (Canton S) to natural and synthetic aphrodisiacs. J. Chem. Ecol. 11, 1617–1629 (1985)
Ferveur, J. F. et al. Genetic feminization of pheromones and its behavioral consequences in Drosophila males. Science 276, 1555–1558 (1997)
Fan, Y., Zurek, L., Dykstra, M. J. & Schal, C. Hydrocarbon synthesis by enzymatically dissociated oenocytes of the abdominal integument of the German Cockroach, Blattella germanica . Naturwissenschaften 90, 121–126 (2003)
Marcillac, F., Bousquet, F., Alabouvette, J., Savarit, F. & Ferveur, J. F. A mutation with major effects on Drosophila melanogaster sex pheromones. Genetics 171, 1617–1628 (2005)
Krupp, J. J. et al. Social experience modifies pheromone expression and mating behavior in male Drosophila melanogaster . Curr. Biol. 18, 1373–1383 (2008)
Venken, K. J. & Bellen, H. Emerging technologies for gene manipulation in Drosophila melanogaster . Nature Rev. Genet. 6, 167–178 (2005)
Zhou, L. et al. Cooperative functions of the reaper and head involution defective genes in the programmed cell death of Drosophila central nervous system midline cells. Proc. Natl Acad. Sci. USA 94, 5131–5136 (1997)
Gutierrez, E., Wiggins, D., Fielding, B. & Gould, A. P. Specialized hepatocyte-like cells regulate Drosophila lipid metabolism. Nature 445, 275–280 (2007)
McGuire, S. E., Le, P. T., Osborn, A. J., Matsumoto, K. & Davis, R. L. Spatiotemporal rescue of memory dysfunction in Drosophila . Science 302, 1765–1768 (2003)
Butterworth, F. M. Lipids of Drosophila: a newly detected lipid in the male. Science 163, 1356–1357 (1969)
Mane, S. D., Tompkins, L. & Richmond, R. C. Male Esterase 6 catalyzes the synthesis of a sex pheromone in Drosophila melanogaster females. Science 222, 419–421 (1983)
Kurtovic, A., Widmer, A. & Dickson, B. J. A single class of olfactory neurons mediates behavioural responses to a Drosophila sex pheromone. Nature 446, 542–546 (2007)
Imhof, M., Harr, B., Brem, G. & Schlötterer, C. Multiple mating in wild Drosophila melanogaster revisited by microsatellite analysis. Mol. Ecol. 7, 915–917 (1998)
Hosken, D. J., Stockley, P., Tregenza, T. & Wedell, N. Monogamy and the battle of the sexes. Annu. Rev. Entomol. 54, 361–378 (2009)
Jallon, J. M. & David, J. R. Variations in cuticular hydrocarbons among the eight species of the Drosophila melanogaster subgroup. Evolution 41, 294–302 (1987)
Cobb, M. & Jallon, J. Pheromones, mate recognition and courtship stimulation in the Drosophila melanogaster species subgroup. Anim. Behav. 39, 1058–1067 (1990)
Cowling, D. E. & Burnet, B. Courtship songs and genetic control of their acoustic characteristics in sibling species of the Drosophila melanogaster subgroup. Anim. Behav. 29, 924–935 (1981)
Kyriacou, C. P. & Hall, J. C. Interspecific genetic control of courtship song production and reception in Drosophila . Science 232, 494–497 (1986)
Wheeler, D. A. et al. Molecular transfer of a species-specific behavior from Drosophila simulans to Drosophila melanogaster . Science 251, 1082–1085 (1991)
Kent, C., Azanchi, R., Smith, B., Formosa, A. & Levine, J. D. Social context influences chemical communication in D. melanogaster males. Curr. Biol. 18, 1384–1389 (2008)
Barolo, S., Carver, L. A. & Posakony, J. W. GFP and β-galactosidase transformation vectors for promoter/enhancer analysis in Drosophila . Biotechniques 29, 726–732 (2000)
Russo, C. A., Takezaki, N. & Nei, M. Molecular phylogeny and divergence times of drosophilid species. Mol. Biol. Evol. 12, 391–404 (1995)
Cobb, M., Connolly, K. & Burnet, B. Courtship behaviour in the melanogaster species sub-group of Drosophila Behaviour 95, 203–230 (1985)
Acknowledgements
We thank H. Pavlou for help with RNA collection; N. Gompel for sharing fly strains; and A. Dornan, S. Douglas, J. C. Hall, I. Dover, C. Kent, C. Kyriacou, K. Luoto, S. Goodwin, M. Ritchie, J. Schneider and M. Sokolowski for critical comments on the manuscript. J.-C.B. was supported by a fellowship for advanced researcher from the Swiss National Science Foundation. This work was supported by the Canadian Institutes of Health Research and Canada Research Chair grants awarded to J.D.L.
Author Contributions J.-C.B. and J.D.L designed and interpreted the study. J.-C.B., J.G.M. and J.D.L. wrote the paper. J.-C. B. generated the Gal4 transgenic line. J.-C.B. and J.A. performed the behavioural experiments and GC-FID analyses, and analysed the behavioural and analytical chemical data. J.J.K. performed the anatomical analyses of the oenocyte-less flies. J.G.M. synthesized and characterized 7-T and 7,11-HD. All authors read and commented on the paper.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
The file contains Supplementary Figures 1-3, Supplementary Tables 1-5, Supplementary Methods and additional references. (PDF 1229 kb)
Supplementary Movie
Supplementary Movie 1 shows two Oenocyte-less males engaged in head-to-head interaction. (MOV 10057 kb)
Supplementary Movie
Supplementary Movie 2 shows two Oenocyte-less males with one male attempting copulation towards the head of the other. (MOV 19817 kb)
Rights and permissions
About this article
Cite this article
Billeter, JC., Atallah, J., Krupp, J. et al. Specialized cells tag sexual and species identity in Drosophila melanogaster. Nature 461, 987–991 (2009). https://doi.org/10.1038/nature08495
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature08495
This article is cited by
-
Intrasexual cuticular hydrocarbon dimorphism in a wasp sheds light on hydrocarbon biosynthesis genes in Hymenoptera
Communications Biology (2023)
-
Aggregation pheromones have a non-linear effect on oviposition behavior in Drosophila melanogaster
Nature Communications (2023)
-
The human neuropsychiatric risk gene Drd2 is necessary for social functioning across evolutionary distant species
Molecular Psychiatry (2023)
-
Volatiles as Attractants of Mahogany Shoot Borer, Hypsipyla grandella Zeller (Lepidoptera: Pyralidae)
Journal of Chemical Ecology (2023)
-
Heterologous expression and functional characterization of Drosophila suzukii OR69a transcript variants unveiled response to kairomones and to a candidate pheromone
Journal of Pest Science (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.