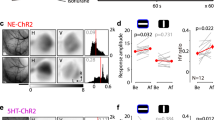
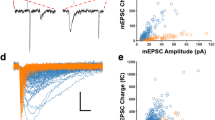
Abstract
Experience-dependent plasticity in the brain requires balanced excitation–inhibition1. How individual circuit elements contribute to plasticity outcome in complex neocortical networks remains unknown. Here we report an intracellular analysis of ocular dominance plasticity—the loss of acuity and cortical responsiveness for an eye deprived of vision in early life2,3. Unlike the typical progressive loss of pyramidal-cell bias, direct recording from fast-spiking cells in vivo reveals a counterintuitive initial shift towards the occluded eye followed by a late preference for the open eye, consistent with a spike-timing-dependent plasticity rule for these inhibitory neurons. Intracellular pharmacology confirms a dynamic switch of GABA (γ-aminobutyric acid) impact to pyramidal cells following deprivation in juvenile mice only. Together these results suggest that the bidirectional recruitment of an initially binocular GABA circuit may contribute to experience-dependent plasticity in the developing visual cortex.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hensch, T. K. Critical period plasticity in local cortical circuits. Nature Rev. Neurosci. 6, 877–888 (2005)
Wiesel, T. N. & Hubel, D. H. Single-cell responses in striate cortex of kittens deprived of vision in one eye. J. Neurophysiol. 26, 1003–1017 (1963)
Prusky, G. T. & Douglas, R. M. Developmental plasticity of mouse visual acuity. Eur. J. Neurosci. 17, 167–173 (2003)
Hensch, T. K. et al. Local GABA circuit control of experience-dependent plasticity in developing visual cortex. Science 282, 1504–1508 (1998)
Gandhi, S. P., Yanagawa, Y. & Stryker, M. P. Delayed plasticity of inhibitory neurons in developing visual cortex. Proc. Natl Acad. Sci. USA 105, 16797–16802 (2008)
Mainardi, M., Landi, S., Berardi, N., Maffei, L. & Pizzorusso, T. Reduced responsiveness to long-term monocular deprivation of parvalbumin neurons assessed by c-Fos staining in rat visual cortex. PLoS ONE 4, e4342 (2009)
Fagiolini, M. et al. Specific GABA-A circuits for visual cortical plasticity. Science 303, 1681–1683 (2004)
Katagiri, H., Fagiolini, M. & Hensch, T. K. Optimization of somatic inhibition at critical period onset. Neuron 53, 805–812 (2007)
Di Cristo, G. et al. Activity-dependent PSA expression regulates inhibitory maturation and onset of critical period plasticity. Nature Neurosci. 10, 1569–1577 (2007)
Sugiyama, S. et al. Experience-dependent transfer of Otx2 homeoprotein into the visual cortex activates postnatal plasticity. Cell 134, 508–520 (2008)
McBain, C. J., Freund, T. F. & Mody, I. Glutamatergic synapses onto hippocampal interneurons: precision timing without lasting plasticity. Trends Neurosci. 22, 228–235 (1999)
Lu, J. T., Li, C. Y., Zhao, J. P., Poo, M. M. & Zhang, X. H. Spike-timing-dependent plasticity of neocortical excitatory synapses on inhibitory interneurons depends on target cell type. J. Neurosci. 27, 9711–9720 (2007)
Inomata, N., Tokutomi, N., Oyama, Y. & Akaike, N. Intracellular picrotoxin blocks pentobarbital-gated Cl- conductance. Neurosci. Res. 6, 72–75 (1988)
Metherate, R. & Ashe, J. H. Ionic flux contributions to neocortical slow waves and nucleus basalis-mediated activation: whole-cell recordings in vivo. J. Neurosci. 13, 5312–5323 (1993)
Mancilla, J. G. & Ulinski, P. S. Role of GABA(A)-mediated inhibition in controlling the responses of regular spiking cells in turtle visual cortex. Vis. Neurosci. 18, 9–24 (2001)
Nelson, S., Toth, L., Sheth, B. & Sur, M. Orientation selectivity of cortical neurons during intracellular blockade of inhibition. Science 265, 774–777 (1994)
Soltesz, I., Smetters, D. K. & Mody, I. Tonic inhibition originates from synapses close to the soma. Neuron 14, 1273–1283 (1995)
Gordon, J. A. & Stryker, M. P. Experience-dependent plasticity of binocular responses in the primary visual cortex of the mouse. J. Neurosci. 16, 3274–3286 (1996)
Dudek, S. M. & Friedlander, M. J. Intracellular blockade of inhibitory synaptic responses in visual cortical layer IV neurons. J. Neurophysiol. 75, 2167–2173 (1996)
Duffy, F. H., Burchfiel, J. L. & Conway, J. L. Bicuculline reversal of deprivation amblyopia in the cat. Nature 260, 256–257 (1976)
Maffei, A., Nataraj, K., Nelson, S. B. & Turrigiano, G. G. Potentiation of cortical inhibition by visual deprivation. Nature 443, 81–84 (2006)
Pizzorusso, T. et al. Reactivation of ocular dominance plasticity in the adult visual cortex. Science 298, 1248–1251 (2002)
Cruikshank, S. J., Lewis, T. J. & Connors, B. W. Synaptic basis for intense thalamocortical activation of feedforward inhibitory cells in neocortex. Nature Neurosci. 10, 462–468 (2007)
Erisir, A. & Dreusicke, M. Quantitative morphology and postsynaptic targets of thalamocortical axons in critical period and adult ferret visual cortex. J. Comp. Neurol. 485, 11–31 (2005)
Kaneko, M., Stellwagen, D., Malenka, R. C. & Stryker, M. P. Tumor necrosis factor-alpha mediates one component of competitive, experience-dependent plasticity in developing visual cortex. Neuron 58, 673–680 (2008)
Frenkel, M. Y. & Bear, M. F. How monocular deprivation shifts ocular dominance in visual cortex of young mice. Neuron 44, 917–923 (2004)
Zheng, W. & Knudsen, E. I. Gabaergic inhibition antagonizes adaptive adjustment of the owl’s auditory space map during the initial phase of plasticity. J. Neurosci. 21, 4356–4365 (2001)
Lamsa, K. P., Heeroma, J. H., Somogyi, P., Rusakov, D. A. & Kullmann, D. M. Anti-Hebbian long-term potentiation in the hippocampal feedback inhibitory circuit. Science 315, 1262–1266 (2007)
Mitchell, J. F., Sundberg, K. A. & Reynolds, J. H. Differential attention-dependent response modulation across cell classes in macaque visual area V4. Neuron 55, 131–141 (2007)
Levi, D. M. & Li, R. W. Improving the performance of the amblyopic visual system. Phil. Trans. R. Soc. Lond. B 364, 399–407 (2009)
Coleman, M. J. & Mooney, R. Synaptic transformations underlying highly selective auditory representations of learned birdsong. J. Neurosci. 24, 7251–7265 (2004)
Volgushev, M., Pernberg, J. & Eysel, U. T. Comparison of the selectivity of postsynaptic potentials and spike responses in cat visual cortex. Eur. J. Neurosci. 12, 257–263 (2000)
Izhikevich, E. M. Simple model of spiking neurons. IEEE Trans. Neural Netw. 14, 1569–1572 (2003)
Stevens, C. F. & Zador, A. M. Input synchrony and the irregular firing of cortical neurons. Nature Neurosci. 1, 210–217 (1998)
Durstewitz, D., Seamans, J. K. & Sejnowski, T. J. Dopamine-mediated stabilization of delay-period activity in a network model of prefrontal cortex. J. Neurophysiol. 83, 1733–1750 (2000)
Erisir, A., Lau, D., Rudy, B. & Leonard, C. S. Function of specific K+ channels in sustained high-frequency firing of fast-spiking neocortical interneurons. J. Neurophysiol. 82, 2476–2489 (1999)
Nomura, M., Fukai, T. & Aoyagi, T. Synchrony of fast-spiking interneurons interconnected by GABAergic and electrical synapses. Neural Comput. 15, 2179–2198 (2003)
Acknowledgements
We thank A. Scheuber for slice experiments, and T. Hosoya and S. Yanagihara for MATLAB programming advice. This work was supported in part by a Grant-in-aid for Scientific Research on Priority Areas (‘Integrative Brain Research’) from the Japanese Ministry of Education, Culture, Sports, Science & Technology (T.F., S.K. and T.K.H.).
Author Contributions T.K.H. and Y.Y.-S. designed the experiments; S.K., H.C. and T.F. formulated the computational models; Y.Y.-S. performed the intracellular recording in vivo; S.K. ran the simulations; and T.K.H., Y.Y.-S., S.K. and T.F. wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-5 with Legends and a Supplementary Reference for Supplementary Figure 2. (PDF 597 kb)
Rights and permissions
About this article
Cite this article
Yazaki-Sugiyama, Y., Kang, S., Câteau, H. et al. Bidirectional plasticity in fast-spiking GABA circuits by visual experience. Nature 462, 218–221 (2009). https://doi.org/10.1038/nature08485
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature08485
This article is cited by
-
Enriched binocular experience followed by sleep optimally restores binocular visual cortical responses in a mouse model of amblyopia
Communications Biology (2023)
-
The plasticitome of cortical interneurons
Nature Reviews Neuroscience (2023)
-
PV network plasticity mediated by neuregulin1-ErbB4 signalling controls fear extinction
Molecular Psychiatry (2022)
-
Parvalbumin interneuron vulnerability and brain disorders
Neuropsychopharmacology (2021)
-
Enriched Environment and Social Isolation Affect Cognition Ability via Altering Excitatory and Inhibitory Synaptic Density in Mice Hippocampus
Neurochemical Research (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.