Abstract
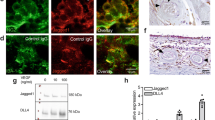
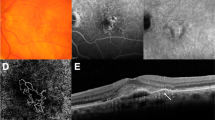
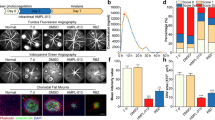
Age-related macular degeneration (AMD), a leading cause of blindness worldwide, is as prevalent as cancer in industrialized nations. Most blindness in AMD results from invasion of the retina by choroidal neovascularisation (CNV). Here we show that the eosinophil/mast cell chemokine receptor CCR3 is specifically expressed in choroidal neovascular endothelial cells in humans with AMD, and that despite the expression of its ligands eotaxin-1, -2 and -3, neither eosinophils nor mast cells are present in human CNV. Genetic or pharmacological targeting of CCR3 or eotaxins inhibited injury-induced CNV in mice. CNV suppression by CCR3 blockade was due to direct inhibition of endothelial cell proliferation, and was uncoupled from inflammation because it occurred in mice lacking eosinophils or mast cells, and was independent of macrophage and neutrophil recruitment. CCR3 blockade was more effective at reducing CNV than vascular endothelial growth factor A (VEGF-A) neutralization, which is in clinical use at present, and, unlike VEGF-A blockade, is not toxic to the mouse retina. In vivo imaging with CCR3-targeting quantum dots located spontaneous CNV invisible to standard fluorescein angiography in mice before retinal invasion. CCR3 targeting might reduce vision loss due to AMD through early detection and therapeutic angioinhibition.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ambati, J., Ambati, B. K., Yoo, S. H., Ianchulev, S. & Adamis, A. P. Age–related macular degeneration: etiology, pathogenesis, and therapeutic strategies. Surv. Ophthalmol. 48, 257–293 (2003)
Gragoudas, E. S., Adamis, A. P., Cunningham, E. T., Feinsod, M. & Guyer, D. R. Pegaptanib for neovascular age-related macular degeneration. N. Engl. J. Med. 351, 2805–2816 (2004)
Brown, D. M. et al. Ranibizumab versus verteporfin for neovascular age-related macular degeneration. N. Engl. J. Med. 355, 1432–1444 (2006)
Rosenfeld, P. J. et al. Ranibizumab for neovascular age-related macular degeneration. N. Engl. J. Med. 355, 1419–1431 (2006)
Famiglietti, E. V. et al. Immunocytochemical localization of vascular endothelial growth factor in neurons and glial cells of human retina. Brain Res. 969, 195–204 (2003)
Nishijima, K. et al. Vascular endothelial growth factor-A is a survival factor for retinal neurons and a critical neuroprotectant during the adaptive response to ischemic injury. Am. J. Pathol. 171, 53–67 (2007)
Saint-Geniez, M. et al. Endogenous VEGF is required for visual function: evidence for a survival role on muller cells and photoreceptors. PLoS One 3, e3554 (2008)
Rothenberg, M. E. & Hogan, S. P. The eosinophil. Annu. Rev. Immunol. 24, 147–174 (2006)
Submacular Surgery Trials Research Group Histopathologic and ultrastructural features of surgically excised subfoveal choroidal neovascular lesions: submacular surgery trials report no. 7. Arch. Ophthalmol. 123, 914–921 (2005)
Justice, J. P. et al. Ablation of eosinophils leads to a reduction of allergen-induced pulmonary pathology. Am. J. Physiol. Lung Cell. Mol. Physiol. 284, L169–L178 (2003)
Humbles, A. A. et al. A critical role for eosinophils in allergic airways remodeling. Science 305, 1776–1779 (2004)
Pope, S. M., Zimmermann, N., Stringer, K. F., Karow, M. L. & Rothenberg, M. E. The eotaxin chemokines and CCR3 are fundamental regulators of allergen-induced pulmonary eosinophilia. J. Immunol. 175, 5341–5350 (2005)
Jose, P. J. et al. Eotaxin: a potent eosinophil chemoattractant cytokine detected in a guinea pig model of allergic airways inflammation. J. Exp. Med. 179, 881–887 (1994)
Teixeira, M. M. et al. Chemokine-induced eosinophil recruitment. Evidence of a role for endogenous eotaxin in an in vivo allergy model in mouse skin. J. Clin. Invest. 100, 1657–1666 (1997)
Blanchard, C. et al. Eotaxin-3 and a uniquely conserved gene-expression profile in eosinophilic esophagitis. J. Clin. Invest. 116, 536–547 (2006)
Salcedo, R. et al. Eotaxin (CCL11) induces in vivo angiogenic responses by human CCR3+ endothelial cells. J. Immunol. 166, 7571–7578 (2001)
Puxeddu, I. et al. Human peripheral blood eosinophils induce angiogenesis. Int. J. Biochem. Cell Biol. 37, 628–636 (2005)
Heissig, B. et al. Low-dose irradiation promotes tissue revascularization through VEGF release from mast cells and MMP-9-mediated progenitor cell mobilization. J. Exp. Med. 202, 739–750 (2005)
Tobe, T. et al. Targeted disruption of the FGF2 gene does not prevent choroidal neovascularization in a murine model. Am. J. Pathol. 153, 1641–1646 (1998)
Nozaki, M. et al. Drusen complement components C3a and C5a promote choroidal neovascularization. Proc. Natl Acad. Sci. USA 103, 2328–2333 (2006)
Nozaki, M. et al. Loss of SPARC-mediated VEGFR-1 suppression after injury reveals a novel antiangiogenic activity of VEGF-A. J. Clin. Invest. 116, 422–429 (2006)
Kleinman, M. E. et al. Sequence- and target-independent angiogenesis suppression by siRNA via TLR3. Nature 452, 591–597 (2008)
Sakurai, E., Anand, A., Ambati, B. K., van Rooijen, N. & Ambati, J. Macrophage depletion inhibits experimental choroidal neovascularization. Invest. Ophthalmol. Vis. Sci. 44, 3578–3585 (2003)
Sakurai, E. et al. Targeted disruption of the CD18 or ICAM-1 gene inhibits choroidal neovascularization. Invest. Ophthalmol. Vis. Sci. 44, 2743–2749 (2003)
Humbles, A. A. et al. The murine CCR3 receptor regulates both the role of eosinophils and mast cells in allergen-induced airway inflammation and hyperresponsiveness. Proc. Natl Acad. Sci. USA 99, 1479–1484 (2002)
Rothenberg, M. E., MacLean, J. A., Pearlman, E., Luster, A. D. & Leder, P. Targeted disruption of the chemokine eotaxin partially reduces antigen-induced tissue eosinophilia. J. Exp. Med. 185, 785–790 (1997)
Kitamura, Y., Go, S. & Hatanaka, K. Decrease of mast cells in W/Wv mice and their increase by bone marrow transplantation. Blood 52, 447–452 (1978)
Zhou, J. et al. Neutrophils promote experimental choroidal neovascularization. Mol. Vis. 11, 414–424 (2005)
Fulkerson, P. C. et al. Negative regulation of eosinophil recruitment to the lung by the chemokine monokine induced by IFN-γ (Mig, CXCL9). Proc. Natl Acad. Sci. USA 101, 1987–1992 (2004)
Fulkerson, P. C., Zhu, H., Williams, D. A., Zimmermann, N. & Rothenberg, M. E. CXCL9 inhibits eosinophil responses by a CCR3- and Rac2-dependent mechanism. Blood 106, 436–443 (2005)
Green, W. R. & Key, S. N. Senile macular degeneration: a histopathologic study. Trans. Am. Ophthalmol. Soc. 75, 180–254 (1977)
Ambati, J. et al. An animal model of age-related macular degeneration in senescent Ccl-2- or Ccr-2-deficient mice. Nature Med. 9, 1390–1397 (2003)
Ip, M. S. et al. Anti-vascular endothelial growth factor pharmacotherapy for age-related macular degeneration: a report by the American Academy of Ophthalmology. Ophthalmology 115, 1837–1846 (2008)
Sayanagi, K., Sharma, S. & Kaiser, P. K. Photoreceptor status after anti-vascular endothelial growth factor therapy in exudative age-related macular degeneration. Br. J. Ophthalmol. 93, 622–626 (2009)
Yodoi, Y. et al. Central retinal sensitivity after intravitreal injection of bevacizumab for myopic choroidal neovascularization. Am. J. Ophthalmol. 147, 816–824 (2009)
Carmeliet, P. et al. Abnormal blood vessel development and lethality in embryos lacking a single VEGF allele. Nature 380, 435–439 (1996)
Ferrara, N. et al. Heterozygous embryonic lethality induced by targeted inactivation of the VEGF gene. Nature 380, 439–442 (1996)
Marneros, A. G. et al. Vascular endothelial growth factor expression in the retinal pigment epithelium is essential for choriocapillaris development and visual function. Am. J. Pathol. 167, 1451–1459 (2005)
Bressler, S. B., Maguire, M. G., Bressler, N. M. & Fine, S. L. (The Macular Photocoagulation Study Group). Relationship of drusen and abnormalities of the retinal pigment epithelium to the prognosis of neovascular macular degeneration. Arch. Ophthalmol. 108, 1442–1447 (1990)
Macular Photocoagulation Study Group Risk factors for choroidal neovascularization in the second eye of patients with juxtafoveal or subfoveal choroidal neovascularization secondary to age-related macular degeneration. Arch. Ophthalmol. 115, 741–747 (1997)
St Croix, B. et al. Genes expressed in human tumor endothelium. Science 289, 1197–1202 (2000)
Zhang, L. et al. Gene expression profiles in normal and cancer cells. Science 276, 1268–1272 (1997)
Wenzel, S. E. Eosinophils in asthma—closing the loop or opening the door? N. Engl. J. Med. 360, 1026–1028 (2009)
Geisen, P., McColm, J. R. & Hartnett, M. E. Choroidal endothelial cells transmigrate across the retinal pigment epithelium but do not proliferate in response to soluble vascular endothelial growth factor. Exp. Eye Res. 82, 608–619 (2006)
Peterson, L. J., Wittchen, E. S., Geisen, P., Burridge, K. & Hartnett, M. E. Heterotypic RPE-choroidal endothelial cell contact increases choroidal endothelial cell transmigration via PI 3-kinase and Rac1. Exp. Eye Res. 84, 737–744 (2007)
Smith, J. R. et al. Unique gene expression profiles of donor-matched human retinal and choroidal vascular endothelial cells. Invest. Ophthalmol. Vis. Sci. 48, 2676–2684 (2007)
Zamora, D. O. et al. Proteomic profiling of human retinal and choroidal endothelial cells reveals molecular heterogeneity related to tissue of origin. Mol. Vis. 13, 2058–2065 (2007)
Acknowledgements
We thank R. King, L. Xu, M. McConnell, K. Emerson, G. R. Pattison and M. Mingler for technical assistance, J. M. Farber for the gift of a reagent, R. J. Kryscio for statistical guidance, and B. Appukuttan, M. W. Fannon, R. Mohan, A. P. Pearson, A. M. Rao, G. S. Rao and K. Ambati for discussions. J.A. was supported by National Eye Institute/National Institutes of Health (NIH) grants EY015422, EY018350 and EY018836, the Doris Duke Distinguished Clinical Scientist Award, the Burroughs Wellcome Fund Clinical Scientist Award in Translational Research, the Macula Vision Research Foundation, the E. Matilda Ziegler Foundation for the Blind, the Dr. E. Vernon Smith and Eloise C. Smith Macular Degeneration Endowed Chair, the Lew R. Wassermann Merit & Physician Scientist Awards (Research to Prevent Blindness, RPB), the American Health Assistance Foundation, and a departmental unrestricted grant from the RPB. J.Z.B. was supported by the University of Kentucky Physician Scientist Award. M.E.K. was supported by the International Retinal Research Foundation Dr. Charles Kelman Postdoctoral Scholar Award. R.J.C.A. was supported by Fight for Sight. B.K.A. was supported by NIH grants EY017182 and EY017950, the VA Merit Award and the Department of Defense. M.E.R. was supported by NIH grants AI45898 and DK076893. C.J.G. was supported by NIH grant AI039759. M.E.H. was supported by NIH grants EY017011 and EY015130 and a RPB departmental unrestricted grant. J.R.S. was supported by NIH grant EY010572, and RPB Career Development Award and a departmental unrestricted grant.
Author Contributions A.T., J.Z.B., M.E.K., W.G.C., M.N., K.Y., H.K., R.J.C.A., S.D., K.S., B.J.R., M.G.G., S.J.B., P.G. and A.M. performed experiments. S.G., A.A.H., Y.P., J.D.W., J.R.S., Y.O. and T.I. provided reagents. J.A. conceived and directed the project, and, with assistance from B.K.A., M.E.H., M.E.R., R.J.C.A. and J.R.S., wrote the paper. All authors had the opportunity to discuss the results and comment on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
J.A. and M.E.K. are named as inventors in a patent application filed by the University of Kentucky on the intellectual property presented in this Article.
Supplementary information
Supplementary Information
This file contains Supplementary Figures S1-S13 with Legends, Supplementary Methods and Supplementary References. (PDF 1615 kb)
Rights and permissions
About this article
Cite this article
Takeda, A., Baffi, J., Kleinman, M. et al. CCR3 is a target for age-related macular degeneration diagnosis and therapy. Nature 460, 225–230 (2009). https://doi.org/10.1038/nature08151
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08151
This article is cited by
-
Fecal microbiota transfer between young and aged mice reverses hallmarks of the aging gut, eye, and brain
Microbiome (2022)
-
IL-4 induces reparative phenotype of RPE cells and protects against retinal neurodegeneration via Nrf2 activation
Cell Death & Disease (2022)
-
Secretogranin III stringently regulates pathological but not physiological angiogenesis in oxygen-induced retinopathy
Cellular and Molecular Life Sciences (2022)
-
Aqueous humor cytokine levels through microarray analysis and a sub-analysis based on optical coherence tomography in wet age-related macular degeneration patients
BMC Ophthalmology (2021)
-
Sensitization of ON-bipolar cells with ambient light activatable multi-characteristic opsin rescues vision in mice
Gene Therapy (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.