Abstract
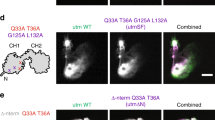
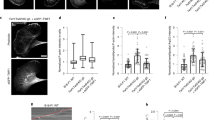
Understanding cell motility will require detailed knowledge not only of the localization of signalling networks regulating actin polymerization, but also of their dynamics. Unfortunately, many signalling networks are not amenable to such analysis, as they are frequently transient and dispersed. By contrast, the signalling pathways used by pathogens undergoing actin-based motility are highly localized and operate in a constitutive fashion1,2,3,4,5. Taking advantage of this, we have analysed the dynamics of neuronal Wiskott–Aldrich syndrome protein (N-WASP), WASP-interacting protein (WIP), GRB2 and NCK, which are required to stimulate actin-related protein (ARP)2/3-complex-dependent actin-based motility of vaccinia virus6,7,8,9, using fluorescence recovery after photobleaching. Here we show that all four proteins are rapidly exchanging, albeit at different rates, and that the turnover of N-WASP depends on its ability to stimulate ARP2/3-complex-mediated actin polymerization. Conversely, disruption of the interaction of N-WASP with GRB2 and/or the barbed ends of actin filaments increases its exchange rate and results in a faster rate of virus movement. We suggest that the exchange rate of N-WASP controls the rate of ARP2/3-complex-dependent actin-based motility by regulating the extent of actin polymerization by antagonizing filament capping.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Frischknecht, F. & Way, M. Surfing pathogens and the lessons learned for actin polymerization. Trends Cell Biol. 11, 30–38 (2001)
Munter, S., Way, M. & Frischknecht, F. Signaling during pathogen infection. Sci. STKE 2006 10.1126/stke.3352006re5 (2006)
Bhavsar, A. P., Guttman, J. A. & Finlay, B. B. Manipulation of host-cell pathways by bacterial pathogens. Nature 449, 827–834 (2007)
Pawson, T. & Warner, N. Oncogenic re-wiring of cellular signaling pathways. Oncogene 26, 1268–1275 (2007)
Backert, S., Feller, S. M. & Wessler, S. Emerging roles of Abl family tyrosine kinases in microbial pathogenesis. Trends Biochem. Sci. 33, 80–90 (2008)
Frischknecht, F. et al. Actin based motility of vaccinia virus mimics receptor tyrosine kinase signalling. Nature 401, 926–929 (1999)
Moreau, V. et al. A complex of N-WASP and WIP integrates signalling cascades that lead to actin polymerization. Nature Cell Biol. 2, 441–448 (2000)
Snapper, S. B. et al. N-WASP deficiency reveals distinct pathways for cell surface projections and microbial actin-based motility. Nature Cell Biol. 3, 897–904 (2001)
Scaplehorn, N. et al. Grb2 and Nck act cooperatively to promote actin-based motility of vaccinia virus. Curr. Biol. 12, 740–745 (2002)
Newsome, T. P., Scaplehorn, N. & Way, M. SRC mediates a switch from microtubule- to actin-based motility of vaccinia virus. Science 306, 124–129 (2004)
Reeves, P. M. et al. Disabling poxvirus pathogenesis by inhibition of Abl-family tyrosine kinases. Nature Med. 11, 731–739 (2005)
Newsome, T. P., Weisswange, I., Frischknecht, F. & Way, M. Abl collaborates with Src family kinases to stimulate actin-based motility of vaccinia virus. Cell. Microbiol. 8, 233–241 (2006)
Zettl, M. & Way, M. The WH1 and EVH1 domains of WASP and Ena/VASP family members bind distinct sequence motifs. Curr. Biol. 12, 1617–1622 (2002)
Co, C., Wong, D. T., Gierke, S., Chang, V. & Taunton, J. Mechanism of actin network attachment to moving membranes: barbed end capture by N-WASP WH2 domains. Cell 128, 901–913 (2007)
Kueh, H. Y., Charras, G. T., Mitchison, T. J. & Brieher, W. M. Actin disassembly by cofilin, coronin, and Aip1 occurs in bursts and is inhibited by barbed-end cappers. J. Cell Biol. 182, 341–353 (2008)
Delatour, V. et al. Arp2/3 controls the motile behavior of N-WASP-functionalized GUVs and modulates N-WASP surface distribution by mediating transient links with actin filaments. Biophys. J. 94, 4890–4905 (2008)
Pantaloni, D., Boujemaa, R., Didry, D., Gounon, P. & Carlier, M.-F. The Arp2/3 complex branches filament barbed ends: functional antagonism with capping proteins. Nature Cell Biol. 2, 385–391 (2000)
Akin, O. & Mullins, R. D. Capping protein increases the rate of actin-based motility by promoting filament nucleation by the Arp2/3 complex. Cell 133, 841–851 (2008)
Arakawa, Y., Cordeiro, J. V. & Way, M. F11L-mediated inhibition of RhoA-mDia signaling stimulates microtubule dynamics during vaccinia virus infection. Cell Host Microbe 1, 213–226 (2007)
Arakawa, Y., Cordeiro, J. V., Schleich, S., Newsome, T. & Way, M. The release of vaccinia virus from infected cells requires RhoA-mDia modulation of cortical actin. Cell Host Microbe 1, 227–240 (2007)
Shewan, A. M. et al. Myosin 2 is a key Rho kinase target necessary for the local concentration of E-cadherin at cell-cell contacts. Mol. Biol. Cell 16, 4531–4542 (2005)
Bladt, F. et al. The murine Nck SH2/SH3 adaptors are important for the development of mesoderm-derived embryonic structures and for regulating the cellular actin network. Mol. Cell. Biol. 23, 4586–4597 (2003)
Rubinson, D. A. et al. A lentivirus-based system to functionally silence genes in primary mammalian cells, stem cells and transgenic mice by RNA interference. Nature Genet. 33, 401–406 (2003)
Hiller, G. & Weber, K. Golgi-derived membranes that contain an acylated viral polypeptide are used for vaccinia virus envelopment. J. Virol. 55, 651–659 (1985)
Acknowledgements
We would like to thank T. Pawson and S. Snapper for NCK- and N-WASP-null cell lines and the FACS laboratory (Cancer Research UK, London) for help sorting stable cell lines. We are also very grateful to the members of the Way laboratory, J. Beaudouin, M.-F. Carlier, H. Gerhardt, B. Mayer, K. Rottner, M. Rosen and M. Welch for discussions and comments on the manuscript. T.P.N. was funded in part by a fellowship from the Human Frontier Science Program (HFSP). This work was funded by Cancer Research UK.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Figures S1-S9 with Legends (PDF 3953 kb)
Rights and permissions
About this article
Cite this article
Weisswange, I., Newsome, T., Schleich, S. et al. The rate of N-WASP exchange limits the extent of ARP2/3-complex-dependent actin-based motility. Nature 458, 87–91 (2009). https://doi.org/10.1038/nature07773
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature07773
This article is cited by
-
Forces generated by lamellipodial actin filament elongation regulate the WAVE complex during cell migration
Nature Cell Biology (2021)
-
The WASP-Arp2/3 complex signal cascade is involved in actin-dependent sperm nuclei migration during double fertilization in tobacco and maize
Scientific Reports (2017)
-
NPF motifs in the vaccinia virus protein A36 recruit intersectin-1 to promote Cdc42:N-WASP-mediated viral release from infected cells
Nature Microbiology (2016)
-
Isoform diversity in the Arp2/3 complex determines actin filament dynamics
Nature Cell Biology (2016)
-
Vaccinia virus dissemination requires p21-activated kinase 1
Archives of Virology (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.