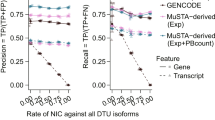
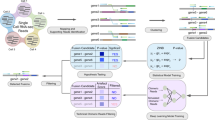
Abstract
Recurrent gene fusions, typically associated with haematological malignancies and rare bone and soft-tissue tumours1, have recently been described in common solid tumours2,3,4,5,6,7,8,9. Here we use an integrative analysis of high-throughput long- and short-read transcriptome sequencing of cancer cells to discover novel gene fusions. As a proof of concept, we successfully used integrative transcriptome sequencing to ‘re-discover’ the BCR–ABL1 (ref. 10) gene fusion in a chronic myelogenous leukaemia cell line and the TMPRSS2–ERG2,3 gene fusion in a prostate cancer cell line and tissues. Additionally, we nominated, and experimentally validated, novel gene fusions resulting in chimaeric transcripts in cancer cell lines and tumours. Taken together, this study establishes a robust pipeline for the discovery of novel gene chimaeras using high-throughput sequencing, opening up an important class of cancer-related mutations for comprehensive characterization.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Mitelman, F., Johansson, B. & Mertens, F. Fusion genes and rearranged genes as a linear function of chromosome aberrations in cancer. Nature Genet. 36, 331–334 (2004)
Tomlins, S. A. et al. Recurrent fusion of TMPRSS2 and ETS transcription factor genes in prostate cancer. Science 310, 644–648 (2005)
Tomlins, S. A. et al. Distinct classes of chromosomal rearrangements create oncogenic ETS gene fusions in prostate cancer. Nature 448, 595–599 (2007)
Kumar-Sinha, C., Tomlins, S. A. & Chinnaiyan, A. M. Recurrent gene fusions in prostate cancer. Nature Rev. Cancer 8, 497–511 (2008)
Choi, Y. L. et al. Identification of novel isoforms of the EML4-ALK transforming gene in non-small cell lung cancer. Cancer Res. 68, 4971–4976 (2008)
Koivunen, J. P. et al. EML4-ALK fusion gene and efficacy of an ALK kinase inhibitor in lung cancer. Clin. Cancer Res. 14, 4275–4283 (2008)
Perner, S. et al. EML4-ALK fusion lung cancer: a rare acquired event. Neoplasia 10, 298–302 (2008)
Rikova, K. et al. Global survey of phosphotyrosine signaling identifies oncogenic kinases in lung cancer. Cell 131, 1190–1203 (2007)
Soda, M. et al. Identification of the transforming EML4-ALK fusion gene in non-small-cell lung cancer. Nature 448, 561–566 (2007)
Rowley, J. D. Chromosome translocations: dangerous liaisons revisited. Nature Rev. Cancer 1, 245–250 (2001)
Lynch, T. J. et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N. Engl. J. Med. 350, 2129–239 (2004)
Slamon, D. J. et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N. Engl. J. Med. 344, 783–792 (2001)
Demetri, G. D. et al. Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. N. Engl. J. Med. 347, 472–480 (2002)
Druker, B. J. et al. Five-year follow-up of patients receiving imatinib for chronic myeloid leukemia. N. Engl. J. Med. 355, 2408–2417 (2006)
Futreal, P. A. et al. A census of human cancer genes. Nature Rev. Cancer 4, 177–183 (2004)
Shtivelman, E., Lifshitz, B., Gale, R. P. & Canaani, E. Fused transcript of abl and bcr genes in chronic myelogenous leukaemia. Nature 315, 550–554 (1985)
Takahara, T., Tasic, B., Maniatis, T., Akanuma, H. & Yanagisawa, S. Delay in synthesis of the 3′ splice site promotes trans-splicing of the preceding 5′ splice site. Mol. Cell 18, 245–251 (2005)
Communi, D., Suarez-Huerta, N., Dussossoy, D., Savi, P. & Boeynaems, J. M. Cotranscription and intergenic splicing of human P2Y11 and SSF1 genes. J. Biol. Chem. 276, 16561–16566 (2001)
Gleave, M. et al. The effects of the dual 5α-reductase inhibitor dutasteride on localized prostate cancer – results from a 4-month pre-radical prostatectomy study. Prostate 66, 1674–1685 (2006)
Han, B. et al. A fluorescence in situ hybridization screen for E26 transformation-specific aberrations: identification of DDX5–ETV4 fusion protein in prostate cancer. Cancer Res. 68, 7629–7637 (2008)
Barber, T. D., Vogelstein, B., Kinzler, K. W. & Velculescu, V. E. Somatic mutations of EGFR in colorectal cancers and glioblastomas. N. Engl. J. Med. 351, 2883 (2004)
Cheung, V. G. et al. Integration of cytogenetic landmarks into the draft sequence of the human genome. Nature 409, 953–958 (2001)
Greenman, C. et al. Patterns of somatic mutation in human cancer genomes. Nature 446, 153–158 (2007)
Stephens, P. et al. A screen of the complete protein kinase gene family identifies diverse patterns of somatic mutations in human breast cancer. Nature Genet. 37, 590–592 (2005)
Strausberg, R. L., Buetow, K. H., Emmert-Buck, M. R. & Klausner, R. D. The cancer genome anatomy project: building an annotated gene index. Trends Genet. 16, 103–106 (2000)
Weir, B. A. et al. Characterizing the cancer genome in lung adenocarcinoma. Nature 450, 893–898 (2007)
Wood, L. D. et al. The genomic landscapes of human breast and colorectal cancers. Science 318, 1108–1113 (2007)
Korenchuk, S. et al. VCaP, a cell-based model system of human prostate cancer. In Vivo 15, 163–168 (2001)
Rubin, M. A. et al. Rapid (‘warm’) autopsy study for procurement of metastatic prostate cancer. Clin. Cancer Res. 6, 1038–1045 (2000)
Karolchik, D. et al. The UCSC Table Browser data retrieval tool. Nucleic Acids Res. 32 (Database issue). D493–D496 (2004)
Abouelhoda, M. I., Kurtz, S. & Ohlebusch, E. Replacing suffix trees with enhanced suffix arrays. J. Discrete Algorithms 2, 53–86 (2004)
Kent, W. J. BLAT – the BLAST-like alignment tool. Genome Res. 12, 656–664 (2002)
Vandesompele, J. et al. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 3, 34–50 (2002)
Acknowledgements
We thank Illumina and 454 for technical support, R. Mehra and J. Siddiqui for providing tissue samples, Y. Gong, S. Shankar, X. Wang and A. Menon for technical assistance, J. Yu for help with the Illumina Genome Analyzer, and R. J. Lonigro for discussions. C.A.M. was supported by a National Institutes of Health Ruth L. Kirschstein post-doctoral training grant, and currently derives support from the American Association of Cancer Research Amgen Fellowship in Clinical/Translational Research, the Canary Foundation and American Cancer Society Early Detection Postdoctoral Fellowship. This work was supported in part by the National Institutes of Health (to A.M.C.), the Department of Defense (to A.M.C.), the Early Detection Research Network (to A.M.C.), and NCIBI (grant number U54 DA 021519).
Author Contributions C.A.M., C.K.-S. and A.M.C. wrote the manuscript. C.K.-S., X.C., X.J., B.H. and N.P. performed the sequencing and biochemical experiments. C.A.M., S.K.-S., L.S. and T.B. performed bioinformatics analysis.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains a Supplementary Discussion, Supplementary Materials and Methods, Supplementary Tables 1-9, Supplementary Figures 1-15 with Legends and Supplementary References (PDF 9467 kb)
Rights and permissions
About this article
Cite this article
Maher, C., Kumar-Sinha, C., Cao, X. et al. Transcriptome sequencing to detect gene fusions in cancer. Nature 458, 97–101 (2009). https://doi.org/10.1038/nature07638
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07638
This article is cited by
-
MALAT1 functions as a transcriptional promoter of MALAT1::GLI1 fusion for truncated GLI1 protein expression in cancer
BMC Cancer (2023)
-
Characterization and evaluation of gene fusions as a measure of genetic instability and disease prognosis in prostate cancer
BMC Cancer (2023)
-
JAFFAL: detecting fusion genes with long-read transcriptome sequencing
Genome Biology (2022)
-
Analysis of gene expression profiles in two spinal cord injury models
European Journal of Medical Research (2022)
-
Susceptibility of cytoskeletal-associated proteins for tumor progression
Cellular and Molecular Life Sciences (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.