Abstract
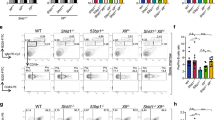
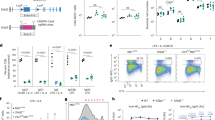
Variable, diversity and joining (V(D)J) recombination and class-switch recombination use overlapping but distinct non-homologous end joining pathways to repair DNA double-strand-break intermediates. 53BP1 is a DNA-damage-response protein that is rapidly recruited to sites of chromosomal double-strand breaks, where it seems to function in a subset of ataxia telangiectasia mutated (ATM) kinase-, H2A histone family member X (H2AX, also known as H2AFX)- and mediator of DNA damage checkpoint 1 (MDC1)-dependent events1,2. A 53BP1-dependent end-joining pathway has been described that is dispensable for V(D)J recombination but essential for class-switch recombination3,4. Here we report a previously unrecognized defect in the joining phase of V(D)J recombination in 53BP1-deficient lymphocytes that is distinct from that found in classical non-homologous-end-joining-, H2ax-, Mdc1- and Atm-deficient mice. Absence of 53BP1 leads to impairment of distal V–DJ joining with extensive degradation of unrepaired coding ends and episomal signal joint reintegration at V(D)J junctions. This results in apoptosis, loss of T-cell receptor α locus integrity and lymphopenia. Further impairment of the apoptotic checkpoint causes propagation of lymphocytes that have antigen receptor breaks. These data suggest a more general role for 53BP1 in maintaining genomic stability during long-range joining of DNA breaks.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Stucki, M. & Jackson, S. P. γH2AX and MDC1: anchoring the DNA-damage-response machinery to broken chromosomes. DNA Repair 5, 534–543 (2006)
Fernandez-Capetillo, O., Lee, A., Nussenzweig, M. & Nussenzweig, A. H2AX: the histone guardian of the genome. DNA Repair 3, 959–967 (2004)
Manis, J. P. et al. 53BP1 links DNA damage-response pathways to immunoglobulin heavy chain class-switch recombination. Nature Immunol. 5, 481–487 (2004)
Ward, I. M. et al. 53BP1 is required for class switch recombination. J. Cell Biol. 165, 459–464 (2004)
Petersen, S. et al. AID is required to initiate Nbs1/γ-H2AX focus formation and mutations at sites of class switching. Nature 414, 660–665 (2001)
Chen, H. T. et al. Response to RAG-mediated VDJ cleavage by NBS1 and γ-H2AX. Science 290, 1962–1965 (2000)
Reina-San-Martin, B., Chen, H. T., Nussenzweig, A. & Nussenzweig, M. C. ATM is required for efficient recombination between immunoglobulin switch regions. J. Exp. Med. 200, 1103–1110 (2004)
Callen, E. et al. ATM prevents the persistence and propagation of chromosome breaks in lymphocytes. Cell 130, 63–75 (2007)
Reina-San-Martin, B. et al. H2AX is required for recombination between immunoglobulin switch regions but not for intra-switch region recombination or somatic hypermutation. J. Exp. Med. 197, 1767–1778 (2003)
Ramiro, A. R. et al. Role of genomic instability and p53 in AID-induced c-myc–Igh translocations. Nature 440, 105–109 (2006)
Franco, S. et al. H2AX prevents DNA breaks from progressing to chromosome breaks and translocations. Mol. Cell 21, 201–214 (2006)
Reina-San-Martin, B., Chen, J., Nussenzweig, A. & Nussenzweig, M. C. Enhanced intra-switch region recombination during immunoglobulin class switch recombination in 53BP1-/- B cells. Eur. J. Immunol. 37, 235–239 (2007)
Ward, I. M., Minn, K., van Deursen, J. & Chen, J. p53 binding protein 53BP1 is required for DNA damage responses and tumor suppression in mice. Mol. Cell. Biol. 23, 2556–2563 (2003)
Celeste, A. et al. Genomic instability in mice lacking histone H2AX. Science 296, 922–927 (2002)
Lou, Z. et al. MDC1 maintains genomic stability by participating in the amplification of ATM-dependent DNA damage signals. Mol. Cell 21, 187–200 (2006)
Matei, I. R. et al. ATM deficiency disrupts Tcra locus integrity and the maturation of CD4+CD8+ thymocytes. Blood 109, 1887–1896 (2007)
Difilippantonio, S. et al. Distinct domains in Nbs1 regulate irradiation-induced checkpoints and apoptosis. J. Exp. Med. 204, 1003–1011 (2007)
Huang, C. Y. et al. Defects in coding joint formation in vivo in developing ATM-deficient B and T lymphocytes. J. Exp. Med. 204, 1371–1381 (2007)
Gu, Y. et al. Growth retardation and leaky SCID phenotype of Ku70-deficient mice. Immunity 7, 653–665 (1997)
Curry, J. D., Geier, J. K. & Schlissel, M. S. Single-strand recombination signal sequence nicks in vivo: evidence for a capture model of synapsis. Nature Immunol. 6, 1272–1279 (2005)
Jones, J. M. & Gellert, M. Ordered assembly of the V(D)J synaptic complex ensures accurate recombination. EMBO J. 21, 4162–4171 (2002)
Huyen, Y. et al. Methylated lysine 79 of histone H3 targets 53BP1 to DNA double-strand breaks. Nature 432, 406–411 (2004)
Botuyan, M. V. et al. Structural basis for the methylation state-specific recognition of histone H4-K20 by 53BP1 and Crb2 in DNA repair. Cell 127, 1361–1373 (2006)
Bekker-Jensen, S., Lukas, C., Melander, F., Bartek, J. & Lukas, J. Dynamic assembly and sustained retention of 53BP1 at the sites of DNA damage are controlled by Mdc1/NFBD1. J. Cell Biol. 170, 201–211 (2005)
Adams, M. M. et al. 53BP1 oligomerization is independent of its methylation by PRMT1. Cell Cycle 4, 1854–1861 (2005)
Lo, K. W. et al. The 8-kDa dynein light chain binds to p53-binding protein 1 and mediates DNA damage-induced p53 nuclear accumulation. J. Biol. Chem. 280, 8172–8179 (2005)
Jhunjhunwala, S. et al. The 3D structure of the immunoglobulin heavy-chain locus: implications for long-range genomic interactions. Cell 133, 265–279 (2008)
Talukder, S. R., Dudley, D. D., Alt, F. W., Takahama, Y. & Akamatsu, Y. Increased frequency of aberrant V(D)J recombination products in core RAG-expressing mice. Nucleic Acids Res. 32, 4539–4549 (2004)
Huang, C. Y. & Sleckman, B. P. Developmental stage-specific regulation of TCR-α-chain gene assembly by intrinsic features of the TEA promoter. J. Immunol. 179, 449–454 (2007)
Bredemeyer, A. L. et al. ATM stabilizes DNA double-strand-break complexes during V(D)J recombination. Nature 442, 466–470 (2006)
Acknowledgements
We are grateful to M. McAuliffe and co-workers for developing the three-dimensional-FISH measurement algorithm; D. G. Schatz and J. Haber for discussions; A. Wynshaw-Boris for Atm-/- mice and J. Chen for 53BP1-/- and Mdc1-/- mice; D. Venzon for help with statistical analysis; and members of the A. Nussenzweig laboratory (J. Daniel and A. Celeste) for comments on the manuscript. B.P.S. is supported by NIH grant R01AI074953. E.G. is supported by pre-doctoral fellowship from the Cancer Research Institute. M.C.N. is a HHMI investigator. A.N. is supported by the Intramural Research Program of the NIH, National Cancer Institute, Center for Cancer Research.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains contains Supplementary Figures 1-5 with Legends and Supplementary Tables 1-4 (PDF 1737 kb)
Rights and permissions
About this article
Cite this article
Difilippantonio, S., Gapud, E., Wong, N. et al. 53BP1 facilitates long-range DNA end-joining during V(D)J recombination. Nature 456, 529–533 (2008). https://doi.org/10.1038/nature07476
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07476
This article is cited by
-
DNA double-strand break end synapsis by DNA loop extrusion
Nature Communications (2023)
-
3D microenvironment attenuates simulated microgravity-mediated changes in T cell transcriptome
Cellular and Molecular Life Sciences (2022)
-
RepID-deficient cancer cells are sensitized to a drug targeting p97/VCP segregase
Molecular & Cellular Toxicology (2021)
-
Functional transcription promoters at DNA double-strand breaks mediate RNA-driven phase separation of damage-response factors
Nature Cell Biology (2019)
-
The H2B deubiquitinase Usp22 promotes antibody class switch recombination by facilitating non-homologous end joining
Nature Communications (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.