Abstract
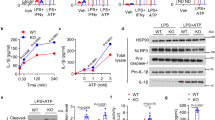
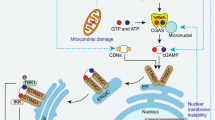
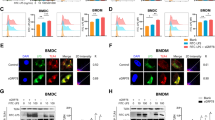
Systems for protein degradation are essential for tight control of the inflammatory immune response1,2. Autophagy, a bulk degradation system that delivers cytoplasmic constituents into autolysosomes, controls degradation of long-lived proteins, insoluble protein aggregates and invading microbes, and is suggested to be involved in the regulation of inflammation3,4,5. However, the mechanism underlying the regulation of inflammatory response by autophagy is poorly understood. Here we show that Atg16L1 (autophagy-related 16-like 1), which is implicated in Crohn's disease6,7, regulates endotoxin-induced inflammasome activation in mice. Atg16L1-deficiency disrupts the recruitment of the Atg12-Atg5 conjugate to the isolation membrane, resulting in a loss of microtubule-associated protein 1 light chain 3 (LC3) conjugation to phosphatidylethanolamine. Consequently, both autophagosome formation and degradation of long-lived proteins are severely impaired in Atg16L1-deficient cells. Following stimulation with lipopolysaccharide, a ligand for Toll-like receptor 4 (refs 8, 9), Atg16L1-deficient macrophages produce high amounts of the inflammatory cytokines IL-1β and IL-18. In lipopolysaccharide-stimulated macrophages, Atg16L1-deficiency causes Toll/IL-1 receptor domain-containing adaptor inducing IFN-β (TRIF)-dependent activation of caspase-1, leading to increased production of IL-1β. Mice lacking Atg16L1 in haematopoietic cells are highly susceptible to dextran sulphate sodium-induced acute colitis, which is alleviated by injection of anti-IL-1β and IL-18 antibodies, indicating the importance of Atg16L1 in the suppression of intestinal inflammation. These results demonstrate that Atg16L1 is an essential component of the autophagic machinery responsible for control of the endotoxin-induced inflammatory immune response.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Liu, Y. C., Penninger, J. & Karin, M. Immunity by ubiquitylation: A reversible process of modification. Nature Rev. Immunol. 5, 941–952 (2005)
Wang, Y. et al. Lysosome-associated small Rab GTPase Rab7b negatively regulates TLR4 signaling in macrophages by promoting lysosomal degradation of TLR4. Blood 110, 962–971 (2007)
Ohsumi, Y. Molecular dissection of autophagy: Two ubiquitin-like systems. Nature Rev. Mol. Cell Biol. 2, 211–216 (2001)
Mizushima, N., Levine, B., Cuervo, A. M. & Klionsky, D. J. Autophagy fights disease through cellular self-digestion. Nature 451, 1069–1075 (2008)
Levine, B. & Deretic, V. Unveiling the roles of autophagy in innate and adaptive immunity. Nature Rev. Immunol. 7, 767–777 (2007)
Hampe, J. et al. A genome-wide association scan of nonsynonymous SNPs identifies a susceptibility variant for Crohn disease in ATG16L1. Nature Genet. 39, 207–211 (2007)
Rioux, J. D. et al. Genome-wide association study identifies new susceptibility loci for Crohn disease and implicates autophagy in disease pathogenesis. Nature Genet. 39, 596–604 (2007)
Akira, S., Uematsu, S. & Takeuchi, O. Pathogen recognition and innate immunity. Cell 124, 783–801 (2006)
Yamamoto, M. et al. Role of adaptor TRIF in the MyD88-independent toll-like receptor signaling pathway. Science 301, 640–643 (2003)
Mizushima, N. et al. Mouse Apg16L, a novel WD-repeat protein, targets to the autophagic isolation membrane with the Apg12-Apg5 conjugate. J. Cell Sci. 116, 1679–1688 (2003)
Fujita, N., Itoh, T., Fukuda, M., Noda, T. & Yoshimori, T. The Atg16L complex specifies the site of LC3 lipidation for membrane biogenesis in autophagy. Mol. Biol. Cell 19, 2092–2100 (2008)
Kuma, A. et al. The role of autophagy during the early neonatal starvation period. Nature 432, 1032–1036 (2004)
Komatsu, M. et al. Impairment of starvation-induced and constitutive autophagy in Atg7-deficient mice. J. Cell Biol. 169, 425–434 (2005)
Lee, H. K., Lund, J. M., Ramanathan, B., Mizushima, N. & Iwasaki, A. Autophagy-dependent viral recognition by plasmacytoid dendritic cells. Science 315, 1398–1401 (2007)
Sanjuan, M. A. et al. Toll-like receptor signalling in macrophages links the autophagy pathway to phagocytosis. Nature 450, 1253–1257 (2007)
Kanneganti, T. D., Lamkanfi, M. & Núñez, G. Intracellular NOD-like receptors in host defense and disease. Immunity 27, 549–559 (2007)
Pétrilli, V., Dostert, C., Muruve, D. A. & Tschopp, J. The inflammasome: A danger sensing complex triggering innate immunity. Curr. Opin. Immunol. 19, 615–622 (2007)
Hsu, L. C. et al. The protein kinase PKR is required for macrophage apoptosis after activation of Toll-like receptor 4. Nature 428, 341–345 (2004)
Greten, F. R. et al. NF-κB is a negative regulator of IL-1β secretion as revealed by genetic and pharmacological inhibition of IKKβ. Cell 130, 918–931 (2007)
Dostert, C. et al. Innate immune activation through Nalp3 inflammasome sensing of asbestos and silica. Science 320, 674–677 (2008)
Hewinson, J., Moore, S. F., Glover, C., Watts, A. G. & MacKenzie, A. B. A key role for redox signaling in rapid P2X7 receptor-induced IL-1beta processing in human monocytes. J. Immunol. 180, 8410–8420 (2008)
Xu, Y. et al. Toll-like receptor 4 is a sensor for autophagy associated with innate immunity. Immunity 27, 135–144 (2007)
Delgado, M. A., Elmaoued, R. A., Davis, A. S., Kyei, G. & Deretic, V. Toll-like receptors control autophagy. EMBO J. 27, 1110–1121 (2008)
Hara, T. et al. Suppression of basal autophagy in neural cells causes neurodegenerative disease in mice. Nature 441, 885–889 (2006)
Komatsu, M. et al. Homeostatic levels of p62 control cytoplasmic inclusion body formation in autophagy-deficient mice. Cell 131, 1149–1163 (2007)
Paludan, C. et al. Endogenous MHC class II processing of a viral nuclear antigen after autophagy. Science 307, 593–596 (2005)
Maeda, S. et al. Nod2 mutation in Crohn's disease potentiates NF-κB activity and IL-1β processing. Science 307, 737–738 (2005)
Ishikura, T. et al. Interleukin-18 overproduction exacerbates the development of colitis with markedly infiltrated macrophages in interleukin-18 transgenic mice. J. Gastroenterol. Hepatol. 18, 960–969 (2003)
Izcue, A., Coombes, J. L. & Powrie, F. Regulatory T cells suppress systemic and mucosal immune activation to control intestinal inflammation. Immunol. Rev. 212, 256–271 (2006)
Nakagawa, I. et al. Autophagy defends cells against invading group A Streptococcus. Science 306, 1037–1040 (2004)
Fujita, N. et al. An Atg4B mutant hampers the lipidation of LC3 paralogues and causes defects in autophagosome closure. Mol. Biol. Cell advance online publication, doi: 10.1091/mbc.E08-03-0312 (3 September 2008)
Acknowledgements
We are grateful to T. Kitamura, S. Yamaoka and N. Mizushima for providing materials. We thank K. J. Ishii, M. Yamamoto and members of the Laboratory of Host Defense for discussions; Y. Fujiwara, M. Shiokawa, R. Nakayama and N. Kitagaki for technical assistance; and M. Hashimoto and E. Kamada for secretarial assistance. This work was in part supported by grants from NIH (AI070167) and the Ministry of Health, Labour and Welfare of Japan, and by Grant-in-Aid for Specially Promoted Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author Contributions T.S. generated the Atg16L1-deficient mice and performed the immunological experiments. N.F. performed the cell biology experiments. N.Y. generated the retroviral vector. M.K. and K.T. generated the Atg7-deficient mice. T.T. performed histological analysis of mice. M.H.J., S.U., B.-G.Y., T.S., H.O., T.N., T.K. and O.T. helped with experiments. T.Y. designed the cell biology research. S.A. supervised the overall research project.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-22 with Legends. (PDF 4004 kb)
Rights and permissions
About this article
Cite this article
Saitoh, T., Fujita, N., Jang, M. et al. Loss of the autophagy protein Atg16L1 enhances endotoxin-induced IL-1β production. Nature 456, 264–268 (2008). https://doi.org/10.1038/nature07383
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07383
This article is cited by
-
The application of nanoparticles-based ferroptosis, pyroptosis and autophagy in cancer immunotherapy
Journal of Nanobiotechnology (2024)
-
The role of the NLRP3 inflammasome and pyroptosis in cardiovascular diseases
Nature Reviews Cardiology (2024)
-
Autophagy: a necessary evil in cancer and inflammation
3 Biotech (2024)
-
Resveratrol and quercetin protect from Benzo(a)pyrene-induced autophagy in retinal pigment epithelial cells
International Ophthalmology (2024)
-
Erbin accelerates TFEB-mediated lysosome biogenesis and autophagy and alleviates sepsis-induced inflammatory responses and organ injuries
Journal of Translational Medicine (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.