Abstract
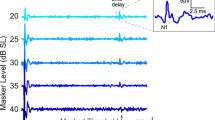
Although the cochlea is an amplifier and a remarkably sensitive and finely tuned detector of sounds, it also produces conspicuous mechanical and electrical waveform distortions1. These distortions reflect nonlinear mechanical interactions within the cochlea. By allowing one tone to suppress another (masking effect), they contribute to speech intelligibility2. Tones can also combine to produce sounds with frequencies not present in the acoustic stimulus3. These sounds compose the otoacoustic emissions that are extensively used to screen hearing in newborns. Because both cochlear amplification and distortion originate from the outer hair cells-one of the two types of sensory receptor cells-it has been speculated that they stem from a common mechanism. Here we show that the nonlinearity underlying cochlear waveform distortions relies on the presence of stereocilin, a protein defective in a recessive form of human deafness4. Stereocilin was detected in association with horizontal top connectors5,6,7, lateral links that join adjacent stereocilia within the outer hair cell's hair bundle. These links were absent in stereocilin-null mutant mice, which became progressively deaf. At the onset of hearing, however, their cochlear sensitivity and frequency tuning were almost normal, although masking was much reduced and both acoustic and electrical waveform distortions were completely lacking. From this unique functional situation, we conclude that the main source of cochlear waveform distortions is a deflection-dependent hair bundle stiffness resulting from constraints imposed by the horizontal top connectors, and not from the intrinsic nonlinear behaviour of the mechanoelectrical transducer channel.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Goldstein, J. L. Auditory nonlinearity. J. Acoust. Soc. Am. 41, 676–689 (1967)
Delgutte, B. in Auditory Computation (eds Hawkins, H. H., McMullen, T. A., Popper, A. N. & Fay, R. R.) 157–220 (Springer, 1996)
Kemp, D. T. Stimulated acoustic emissions from within the human auditory system. J. Acoust. Soc. Am. 64, 1386–1391 (1978)
Verpy, E. et al. Mutations in a new gene encoding a protein of the hair bundle cause non-syndromic deafness at the DFNB16 locus. Nature Genet. 29, 345–349 (2001)
Furness, D. N. & Hackney, C. M. Cross-links between stereocilia in the guinea pig cochlea. Hear. Res. 18, 177–188 (1985)
Tsuprun, V. & Santi, P. Structure of outer hair cell stereocilia side and attachment links in the chinchilla cochlea. J. Histochem. Cytochem. 50, 493–502 (2002)
Goodyear, R. J., Marcotti, W., Kros, C. J. & Richardson, G. P. Development and properties of stereociliary link types in hair cells of the mouse cochlea. J. Comp. Neurol. 485, 75–85 (2005)
Brownell, W. E., Bader, C. R., Bertrand, D. & de Ribaupierre, Y. Evoked mechanical responses of isolated cochlear outer hair cells. Science 227, 194–196 (1985)
Ashmore, J. F. A fast motile response in guinea-pig outer hair cells: the cellular basis of the cochlear amplifier. J. Physiol. (Lond.) 388, 323–347 (1987)
He, D. Z. & Dallos, P. Somatic stiffness of cochlear outer hair cells is voltage-dependent. Proc. Natl Acad. Sci. USA 96, 8223–8228 (1999)
Liberman, M. C. et al. Prestin is required for electromotility of the outer hair cell and for the cochlear amplifier. Nature 419, 300–304 (2002)
Hudspeth, A. J. How the ear’s works work. Nature 341, 397–404 (1989)
Kennedy, H. J., Crawford, A. C. & Fettiplace, R. Force generation by mammalian hair bundles supports a role in cochlear amplification. Nature 433, 880–883 (2005)
Dallos, P. & Harris, D. Properties of auditory nerve responses in absence of outer hair cells. J. Neurophysiol. 41, 365–383 (1978)
Liberman, M. C. & Dodds, L. W. Single-neuron labeling and chronic cochlear pathology. III. Stereocilia damage and alterations of threshold tuning curves. Hear. Res. 16, 55–74 (1984)
Robles, L. & Ruggero, M. A. Mechanics of the mammalian cochlea. Physiol. Rev. 81, 1305–1352 (2001)
Patuzzi, R. B., Yates, G. K. & Johnstone, B. M. Outer hair cell receptor current and sensorineural hearing loss. Hear. Res. 42, 47–72 (1989)
Dallos, P. & Cheatham, M. A. Compound action potential (AP) tuning curves. J. Acoust. Soc. Am. 59, 591–597 (1976)
Moore, B. C. J. Cochlear Hearing Loss (Whurr Publishers Ltd, 1998)
Ruggero, M. A., Robles, L. & Rich, N. C. Two-tone suppression in the basilar membrane of the cochlea: mechanical basis of auditory-nerve rate suppression. J. Neurophysiol. 68, 1087–1099 (1992)
Kim, D. O., Molnar, C. E. & Matthews, J. W. Cochlear mechanics: nonlinear behavior in two-tone responses as reflected in cochlear-nerve-fiber responses and in ear-canal sound pressure. J. Acoust. Soc. Am. 67, 1704–1721 (1980)
Robles, L., Ruggero, M. A. & Rich, N. C. Two-tone distortion on the basilar membrane of the chinchilla cochlea. J. Neurophysiol. 77, 2385–2399 (1997)
Kimura, R. S. Hairs of the cochlear sensory cells and their attachment to the tectorial membrane. Acta Otolaryngol. (Stockh.) 61, 55–72 (1966)
Legan, P. K. et al. A targeted deletion in α-tectorin reveals that the tectorial membrane is required for the gain and timing of cochlear feedback. Neuron 28, 273–285 (2000)
Lukashkin, A. N., Lukashkina, V. A., Legan, P. K., Richardson, G. P. & Russell, I. J. Role of the tectorial membrane revealed by otoacoustic emissions recorded from wild-type and transgenic TectaΔENT/ΔENT mice. J. Neurophysiol. 91, 163–171 (2004)
Patuzzi, R. in The Cochlea (eds Dallos, P., Popper, A. N. & Fay, R. R.) 186–257 (Springer, 1996)
van Netten, S. M. & Kros, C. J. Gating energies and forces of the mammalian hair cell transducer channel and related hair bundle mechanics. Proc. R. Soc. Lond. B. 267, 1915–1923 (2000)
Howard, J. & Hudspeth, A. J. Compliance of the hair bundle associated with gating of mechanoelectrical transduction channels in the bullfrog’s saccular hair cell. Neuron 1, 189–199 (1988)
Flock, A. & Strelioff, D. Graded and nonlinear mechanical properties of sensory hairs in the mammalian hearing organ. Nature 310, 597–599 (1984)
Fettiplace, R. Active hair bundle movements in auditory hair cells. J. Physiol. (Lond.) 576, 29–36 (2006)
Kress, C., Vandormael-Pournin, S., Baldacci, P., Cohen-Tannoudji, M. & Babinet, C. Nonpermissiveness for mouse embryonic stem (ES) cell derivation circumvented by a single backcross to 129/Sv strain: establishment of ES cell lines bearing the Omd conditional lethal mutation. Mamm. Genome 9, 998–1001 (1998)
Lallemand, Y., Luria, V., Haffner-Krausz, R. & Lonai, P. Maternally expressed PGK-Cre transgene as a tool for early and uniform activation of the Cre site-specific recombinase. Transgenic Res. 7, 105–112 (1998)
Michel, V. et al. Cadherin 23 is a component of the transient lateral links in the developing hair bundles of cochlear sensory cells. Dev. Biol. 280, 281–294 (2005)
Delmaghani, S. et al. Mutations in the gene encoding pejvakin, a newly identified protein of the afferent auditory pathway, cause DFNB59 auditory neuropathy. Nature Genet. 38, 770–778 (2006)
Acknowledgements
We thank M. Lenoir, S. Guadagnini and M.-C. Prévost for advice on SEM; E. Perret and P. Roux for advice on confocal microscopy, S. Chardenoux, S. Nouaille and A. Mallet for technical help, Y. Lallemand for providing us with PGK-Crem mice, N. Michalski for help with figure drawing and J. Boutet de Monvel for critical reading of the manuscript. This work was supported by the European Commission FP6 Integrated Project EuroHear, Fondation Raymonde et Guy Strittmatter, Région Ile-de-France (Programme Sésame), and the Wellcome Trust.
Author Contributions C.P. and P.A. contributed equally as co-senior authors. D.W. and M.L. are co-second authors. D.W. and G.H. produced the Strc knockout mice. P.A. conducted the auditory tests. M.L. performed the SEM experiments on wild type, Strc-/- and TectaΔENT/ΔENT mice, with help from C.H. G.M.L. carried out the SEM experiments in the mouse models of Usher syndrome. E.V. performed the immunofluorescence studies with help from C.H., and the RT–PCR analysis. G.P.R. and R.J.G. carried out the transmission electron microscopy analysis. E.V. supervised the stereocilin expression studies and the characterization of the morphological anomalies in the Strc knockout mice. C.P. supervised the whole project with help from G.P.R. and in collaboration with P.A. for the physiological studies. E.V., J.-P.H., G.P.R., P.A. and C.P. prepared the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-6 with Legends and Supplementary Notes (PDF 1653 kb)
Rights and permissions
About this article
Cite this article
Verpy, E., Weil, D., Leibovici, M. et al. Stereocilin-deficient mice reveal the origin of cochlear waveform distortions. Nature 456, 255–258 (2008). https://doi.org/10.1038/nature07380
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07380
This article is cited by
-
Digital live imaging of intraoperative electrocochleography during cochlear implantation: the first 50 patients
European Archives of Oto-Rhino-Laryngology (2024)
-
Microtubule-associated protein 1 A and tubby act independently in regulating the localization of stereocilin to the tips of inner ear hair cell stereocilia
Molecular Brain (2022)
-
Characterizing the Relationship Between Reflection and Distortion Otoacoustic Emissions in Normal-Hearing Adults
Journal of the Association for Research in Otolaryngology (2022)
-
Central auditory deficits associated with genetic forms of peripheral deafness
Human Genetics (2022)
-
Inner hair cell stereocilia are embedded in the tectorial membrane
Nature Communications (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.