Abstract
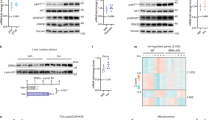
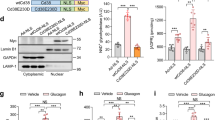
During early fasting, increases in skeletal muscle proteolysis liberate free amino acids for hepatic gluconeogenesis in response to pancreatic glucagon. Hepatic glucose output diminishes during the late protein-sparing phase of fasting, when ketone body production by the liver supplies compensatory fuel for glucose-dependent tissues1,2,3,4. Glucagon stimulates the gluconeogenic program by triggering the dephosphorylation and nuclear translocation of the CREB regulated transcription coactivator 2 (CRTC2; also known as TORC2), while parallel decreases in insulin signalling augment gluconeogenic gene expression through the dephosphorylation and nuclear shuttling of forkhead box O1 (FOXO1)5,6,7. Here we show that a fasting-inducible switch, consisting of the histone acetyltransferase p300 and the nutrient-sensing deacetylase sirtuin 1 (SIRT1), maintains energy balance in mice through the sequential induction of CRTC2 and FOXO1. After glucagon induction, CRTC2 stimulated gluconeogenic gene expression by an association with p300, which we show here is also activated by dephosphorylation at Ser 89 during fasting. In turn, p300 increased hepatic CRTC2 activity by acetylating it at Lys 628, a site that also targets CRTC2 for degradation after its ubiquitination by the E3 ligase constitutive photomorphogenic protein (COP1)8. Glucagon effects were attenuated during late fasting, when CRTC2 was downregulated owing to SIRT1-mediated deacetylation and when FOXO1 supported expression of the gluconeogenic program. Disrupting SIRT1 activity, by liver-specific knockout of the Sirt1 gene or by administration of a SIRT1 antagonist, increased CRTC2 activity and glucose output, whereas exposure to SIRT1 agonists reduced them. In view of the reciprocal activation of FOXO1 and its coactivator peroxisome proliferator-activated receptor-γ coactivator-1α (PGC-1α, encoded by Ppargc1a) by SIRT1 activators9,10,11,12, our results illustrate how the exchange of two gluconeogenic regulators during fasting maintains energy balance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Marliss, E. B., Aoki, T. T., Unger, R. H., Soeldner, J. S. & Cahill, G. F. Glucagon levels and metabolic effects in fasting man. J. Clin. Invest. 49, 2256–2270 (1970)
Goodman, M. N. et al. Starvation in the rat. II. Effect of age and obesity on protein sparing and fuel metabolism. Am. J. Physiol. 239, E277–E286 (1980)
Goodman, M. N., McElaney, M. A. & Ruderman, N. B. Adaptation to prolonged starvation in the rat: curtailment of skeletal muscle proteolysis. Am. J. Physiol. 241, E321–E327 (1981)
Cahill, G. F. Fuel metabolism in starvation. Annu. Rev. Nutr. 26, 1–22 (2006)
Puigserver, P. et al. Insulin-regulated hepatic gluconeogenesis through FOXO1–PGC-1α interaction. Nature 423, 550–555 (2003)
Matsumoto, M., Pocai, A., Rossetti, L., Depinho, R. A. & Accili, D. Impaired regulation of hepatic glucose production in mice lacking the forkhead transcription factor Foxo1 in liver. Cell Metab. 6, 208–216 (2007)
Koo, S. H. et al. The CREB coactivator TORC2 is a key regulator of fasting glucose metabolism. Nature 437, 1109–1111 (2005)
Dentin, R. et al. Insulin modulates gluconeogenesis by inhibition of the coactivator TORC2. Nature 449, 366–369 (2007)
Rodgers, J. T. et al. Nutrient control of glucose homeostasis through a complex of PGC-1α and SIRT1. Nature 434, 113–118 (2005)
Baur, J. A. et al. Resveratrol improves health and survival of mice on a high-calorie diet. Nature 444, 337–342 (2006)
Frescas, D., Valenti, L. & Accili, D. Nuclear trapping of the forkhead transcription factor FoxO1 via Sirt-dependent deacetylation promotes expression of glucogenetic genes. J. Biol. Chem. 280, 20589–20595 (2005)
Gan, L. et al. FoxO-dependent and -independent mechanisms mediate SirT1 effects on IGFBP-1 gene expression. Biochem. Biophys. Res. Commun. 337, 1092–1096 (2005)
Badman, M. K. et al. Hepatic fibroblast growth factor 21 is regulated by PPARα and is a key mediator of hepatic lipid metabolism in ketotic states. Cell Metab. 5, 426–437 (2007)
Nakae, J., Kitamura, T., Silver, D. L. & Accili, D. The forkhead transcription factor Foxo1 (Fkhr) confers insulin sensitivity onto glucose-6-phosphatase expression. J. Clin. Invest. 108, 1359–1367 (2001)
Barthel, A. et al. Differential regulation of endogenous glucose-6-phosphatase and phosphoenolpyruvate carboxykinase gene expression by the forkhead transcription factor FKHR in H4IIE-hepatoma cells. Biochem. Biophys. Res. Commun. 285, 897–902 (2001)
Hornbuckle, L. A. et al. Selective stimulation of G-6-Pase catalytic subunit but not G-6-P transporter gene expression by glucagon in vivo and cAMP in situ. Am. J. Physiol. Endocrinol. Metab. 286, E795–E808 (2004)
Ayala, J. E. et al. Conservation of an insulin response unit between mouse and human glucose-6-phosphatase catalytic subunit gene promoters: transcription factor FKHR binds the insulin response sequence. Diabetes 48, 1885–1889 (1999)
Schmoll, D. et al. Identification of a cAMP response element within the glucose-6-phosphatase hydrolytic subunit gene promoter which is involved in the transcriptional regulation by cAMP and glucocorticoids in H4IIE hepatoma cells. Biochem. J. 338, 457–463 (1999)
Freiman, R. N. & Tjian, R. Regulating the regulators: lysine modifications make their mark. Cell 112, 11–17 (2003)
Ravnskjaer, K. et al. Cooperative interactions between CBP and TORC2 confer selectivity to CREB target gene expression. EMBO J. 26, 2880–2889 (2007)
Yuan, L. W. & Gambee, J. E. Phosphorylation of p300 at serine 89 by protein kinase C. J. Biol. Chem. 275, 40946–40951 (2000)
Yang, W. et al. Regulation of transcription by AMP-activated protein kinase: phosphorylation of p300 blocks its interaction with nuclear receptors. J. Biol. Chem. 276, 38341–38344 (2001)
Guidez, F. et al. Histone acetyltransferase activity of p300 is required for transcriptional repression by the promyelocytic leukemia zinc finger protein. Mol. Cell. Biol. 25, 5552–5566 (2005)
Zheng, Y. et al. Synthesis and evaluation of a potent and selective cell-permeable p300 histone acetyltransferase inhibitor. J. Am. Chem. Soc. 127, 17182–17183 (2005)
Blander, G. & Guarente, L. The Sir2 family of protein deacetylases. Annu. Rev. Biochem. 73, 417–435 (2004)
Milne, J. C. et al. Small molecule activators of SIRT1 as therapeutics for the treatment of type 2 diabetes. Nature 450, 712–716 (2007)
Motta, M. C. et al. Mammalian SIRT1 represses forkhead transcription factors. Cell 116, 551–563 (2004)
Ferre, P., Pegorier, J. P., Marliss, E. B. & Girard, J. R. Influence of exogenous fat and gluconeogenic substrates on glucose homeostasis in the newborn rat. Am. J. Physiol. 234, E129–E136 (1978)
Cheng, H. L. et al. Developmental defects and p53 hyperacetylation in Sir2 homolog (SIRT1)-deficient mice. Proc. Natl Acad. Sci. USA 100, 10794–10799 (2003)
Miyabayashi, T. et al. Wnt/β-catenin/CBP signaling maintains long-term murine embryonic stem cell pluripotency. Proc. Natl Acad. Sci. USA 104, 5668–5673 (2007)
Acknowledgements
We thank M. Kahn for the gift of phospho-specific p300 antiserum and L. Vera for technical assistance. We also thank R. Shaw and M. Mihaylova for sharing results on p300 phosphorylation. This work was supported by grants from the National Institutes of Health, the Clayton Medical Research Foundation, Inc., the Hillblom Foundation (to Y.L.), and the Kieckhefer Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
L.G. is a member of the Advisory Board at Sirtris. J.M. was an employee at Sirtris.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-17 with Legends. (PDF 722 kb)
Rights and permissions
About this article
Cite this article
Liu, Y., Dentin, R., Chen, D. et al. A fasting inducible switch modulates gluconeogenesis via activator/coactivator exchange. Nature 456, 269–273 (2008). https://doi.org/10.1038/nature07349
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07349
This article is cited by
-
The CRTC-1 transcriptional domain is required for COMPASS complex-mediated longevity in C. elegans
Nature Aging (2023)
-
Proteogenomics of different urothelial bladder cancer stages reveals distinct molecular features for papillary cancer and carcinoma in situ
Nature Communications (2023)
-
Characterization and Optimization of Chitosan Production by Aspergillus terreus
Arabian Journal for Science and Engineering (2023)
-
Biological functions of CRTC2 and its role in metabolism-related diseases
Journal of Cell Communication and Signaling (2023)
-
A propolis-derived small molecule ameliorates metabolic syndrome in obese mice by targeting the CREB/CRTC2 transcriptional complex
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.