Abstract
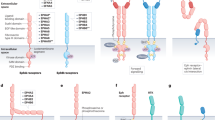
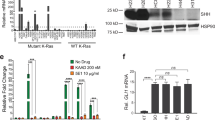
Ligand-dependent activation of the hedgehog (Hh) signalling pathway has been associated with tumorigenesis in a number of human tissues1,2,3,4,5,6,7. Here we show that, although previous reports have described a cell-autonomous role for Hh signalling in these tumours1,2,3,4,5,6,7, Hh ligands fail to activate signalling in tumour epithelial cells. In contrast, our data support ligand-dependent activation of the Hh pathway in the stromal microenvironment. Specific inhibition of Hh signalling using small molecule inhibitors, a neutralizing anti-Hh antibody or genetic deletion of smoothened (Smo) in the mouse stroma results in growth inhibition in xenograft tumour models. Taken together, these studies demonstrate a paracrine requirement for Hh ligand signalling in the tumorigenesis of Hh-expressing cancers and have important implications for the development of Hh pathway antagonists in cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Clement, V., Sanchez, P., de Tribolet, N., Radovanovic, I. & Ruiz i Altaba, A. HEDGEHOG–GLI1 signaling regulates human glioma growth, cancer stem cell self-renewal, and tumorigenicity. Curr. Biol. 17, 165–172 (2007)
Stecca, B. et al. Melanomas require HEDGEHOG–GLI signaling regulated by interactions between GLI1 and the RAS-MEK/AKT pathways. Proc. Natl Acad. Sci. USA 104, 5895–5900 (2007)
Yuan, Z. et al. Frequent requirement of hedgehog signaling in non-small cell lung carcinoma. Oncogene 26, 1046–1055 (2007)
Thayer, S. P. et al. Hedgehog is an early and late mediator of pancreatic cancer tumorigenesis. Nature 425, 851–856 (2003)
Karhadkar, S. S. et al. Hedgehog signalling in prostate regeneration, neoplasia and metastasis. Nature 431, 707–712 (2004)
Berman, D. M. et al. Widespread requirement for Hedgehog ligand stimulation in growth of digestive tract tumours. Nature 425, 846–851 (2003)
Watkins, D. N. et al. Hedgehog signalling within airway epithelial progenitors and in small-cell lung cancer. Nature 422, 313–317 (2003)
Ingham, P. W. & McMahon, A. P. Hedgehog signaling in animal development: paradigms and principles. Genes Dev. 15, 3059–3087 (2001)
van den Brink, G. R. Hedgehog signaling in development and homeostasis of the gastrointestinal tract. Physiol. Rev. 87, 1343–1375 (2007)
Romer, J. T. et al. Suppression of the Shh pathway using a small molecule inhibitor eliminates medulloblastoma in Ptc1+/-p53-/- mice. Cancer Cell 6, 229–240 (2004)
Frank-Kamenetsky, M. et al. Small-molecule modulators of Hedgehog signaling: identification and characterization of Smoothened agonists and antagonists. J. Biol. 1, 10 (2002)
Cooper, M. K., Porter, J. A., Young, K. E. & Beachy, P. A. Teratogen-mediated inhibition of target tissue response to Shh signaling. Science 280, 1603–1607 (1998)
Kayed, H. et al. Indian hedgehog signaling pathway: expression and regulation in pancreatic cancer. Int. J. Cancer 110, 668–676 (2004)
Fan, C. M. et al. Long-range sclerotome induction by sonic hedgehog: direct role of the amino-terminal cleavage product and modulation by the cyclic AMP signaling pathway. Cell 81, 457–465 (1995)
Sasai, K. et al. Shh pathway activity is down-regulated in cultured medulloblastoma cells: implications for preclinical studies. Cancer Res. 66, 4215–4222 (2006)
Duda, D. G. et al. Differential transplantability of tumor-associated stromal cells. Cancer Res. 64, 5920–5924 (2004)
Hayashi, S. & McMahon, A. P. Efficient recombination in diverse tissues by a tamoxifen-inducible form of Cre: a tool for temporally regulated gene activation/inactivation in the mouse. Dev. Biol. 244, 305–318 (2002)
Long, F., Zhang, X. M., Karp, S., Yang, Y. & McMahon, A. P. Genetic manipulation of hedgehog signaling in the endochondral skeleton reveals a direct role in the regulation of chondrocyte proliferation. Development 128, 5099–5108 (2001)
Beachy, P. A., Karhadkar, S. S. & Berman, D. M. Tissue repair and stem cell renewal in carcinogenesis. Nature 432, 324–331 (2004)
Fan, L. et al. Hedgehog signaling promotes prostate xenograft tumor growth. Endocrinology 145, 3961–3970 (2004)
Pola, R. et al. The morphogen Sonic hedgehog is an indirect angiogenic agent upregulating two families of angiogenic growth factors. Nature Med. 7, 706–711 (2001)
Ingram, W. J., Wicking, C. A., Grimmond, S. M., Forrest, A. R. & Wainwright, B. J. Novel genes regulated by Sonic Hedgehog in pluripotent mesenchymal cells. Oncogene 21, 8196–8205 (2002)
Bergmann, U., Funatomi, H., Yokoyama, M., Beger, H. G. & Korc, M. Insulin-like growth factor I overexpression in human pancreatic cancer: evidence for autocrine and paracrine roles. Cancer Res. 55, 2007–2011 (1995)
Durai, R., Yang, W., Gupta, S., Seifalian, A. M. & Winslet, M. C. The role of the insulin-like growth factor system in colorectal cancer: review of current knowledge. Int. J. Colorectal Dis. 20, 203–220 (2005)
Zeng, G. et al. Aberrant Wnt/β-catenin signaling in pancreatic adenocarcinoma. Neoplasia 8, 279–289 (2006)
Pasca di Magliano, M. et al. Common activation of canonical wnt signaling in pancreatic adenocarcinoma. PLoS ONE 2, e1155 (2007)
Bhowmick, N. A., Neilson, E. G. & Moses, H. L. Stromal fibroblasts in cancer initiation and progression. Nature 432, 332–337 (2004)
Orimo, A. & Weinberg, R. A. Stromal fibroblasts in cancer: a novel tumor-promoting cell type. Cell Cycle 5, 1597–1601 (2006)
Ericson, J., Morton, S., Kawakami, A., Roelink, H. & Jessell, T. M. Two critical periods of Sonic Hedgehog signaling required for the specification of motor neuron identity. Cell 87, 661–673 (1996)
Yoneda, T. & Pratt, R. M. Mesenchymal cells from the human embryonic palate are highly responsive to epidermal growth factor. Science 213, 563–565 (1981)
Acknowledgements
The authors thank A. McMahon and M. Scott for providing transgenic mice; T. Holcomb, K. Wagner, D. Lee and P. Wen for their assistance in cell line screening; J. Ernst for rSHH, M. Cole for assistance with imaging; S. Louie for assistance with graphics; P. Haverty for assistance with gene expression data; and M. Evangelista, C. Callahan and V. Dixit for comments and discussions. Tissue samples were provided by the Cooperative Human Tissue Network, which is funded by the National Cancer Institute, and the National Center for Research Resources, which is supported by the National Institutes of Health. Other investigators may have received samples from these same tissues.
Author Contributions F.J.d.S. and L.L.R. conceived and directed the project. S.E.G., R.L.Y., S.J.S., T.T., H.T. J.C.M. and K.K. designed and carried out experiments. C.P.A., D.M., L.F., T.J., D.K. and M.N.-P. carried out experiments. R.L.Y., S.E.G. and F.J.d.S. wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
R.L.Y., S.E.G., S.J.S., T.T., H.T., C.A., D.M., L.F., T.J., D.K., M.N.-P., J.C.M. and F.J.d.S. are employed by Genentech, Inc. and/or may have stocks or shares in Genentech, Inc.
Supplementary information
Supplementary Information 1
This file contains Supplementary Methods, Supplementary Tables 1-3 and Supplementary Figures 1-13. (PDF 3308 kb)
Supplementary Information 2
This file contains supplemental microarray data. (XLS 16739 kb)
Supplementary Information 3
This file contains supplemental microarray data. (XLS 14492 kb)
Rights and permissions
About this article
Cite this article
Yauch, R., Gould, S., Scales, S. et al. A paracrine requirement for hedgehog signalling in cancer. Nature 455, 406–410 (2008). https://doi.org/10.1038/nature07275
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07275
This article is cited by
-
Usability of serum hedgehog signalling proteins as biomarkers in canine mammary carcinomas
BMC Veterinary Research (2023)
-
Targeting the hedgehog pathway in MET mutation cancers and its effects on cells associated with cancer development
Cell Communication and Signaling (2023)
-
Hedgehog signaling in tissue homeostasis, cancers, and targeted therapies
Signal Transduction and Targeted Therapy (2023)
-
Epithelial and stromal co-evolution and complicity in pancreatic cancer
Nature Reviews Cancer (2023)
-
Fibroblast subtypes in pancreatic cancer and pancreatitis: from mechanisms to therapeutic strategies
Cellular Oncology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.