Abstract
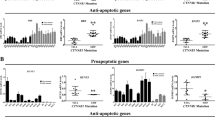
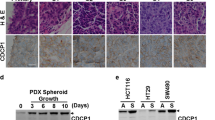
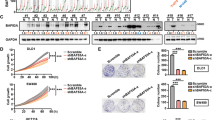
Aberrant activation of the canonical WNT/β-catenin pathway occurs in almost all colorectal cancers and contributes to their growth, invasion and survival1,2. Although dysregulated β-catenin activity drives colon tumorigenesis, further genetic perturbations are required to elaborate full malignant transformation3. To identify genes that both modulate β-catenin activity and are essential for colon cancer cell proliferation, we conducted two loss-of-function screens in human colon cancer cells and compared genes identified in these screens with an analysis of copy number alterations in colon cancer specimens. One of these genes, CDK8, which encodes a member of the mediator complex4, is located at 13q12.13, a region of recurrent copy number gain in a substantial fraction of colon cancers. Here we show that the suppression of CDK8 expression inhibits proliferation in colon cancer cells characterized by high levels of CDK8 and β-catenin hyperactivity. CDK8 kinase activity was necessary for β-catenin-driven transformation and for expression of several β-catenin transcriptional targets. Together these observations suggest that therapeutic interventions targeting CDK8 may confer a clinical benefit in β-catenin-driven malignancies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Bienz, M. & Clevers, H. Linking colorectal cancer to Wnt signaling. Cell 103, 311–320 (2000)
Camp, R. L., Chung, G. G. & Rimm, D. L. Automated subcellular localization and quantification of protein expression in tissue microarrays. Nature Med. 8, 1323–1327 (2002)
Vogelstein, B. et al. Genetic alterations during colorectal-tumor development. N. Engl. J. Med. 319, 525–532 (1988)
Conaway, R. C., Sato, S., Tomomori-Sato, C., Yao, T. & Conaway, J. W. The mammalian Mediator complex and its role in transcriptional regulation. Trends Biochem. Sci. 30, 250–255 (2005)
Smith, G. et al. Mutations in APC, Kirsten-ras, and p53—alternative genetic pathways to colorectal cancer. Proc. Natl Acad. Sci. USA 99, 9433–9438 (2002)
van de Wetering, M. et al. The β-catenin/TCF-4 complex imposes a crypt progenitor phenotype on colorectal cancer cells. Cell 111, 241–250 (2002)
Moffat, J. et al. A lentiviral RNAi library for human and mouse genes applied to an arrayed viral high-content screen. Cell 124, 1283–1298 (2006)
Korinek, V. et al. Constitutive transcriptional activation by a β-catenin–Tcf complex in APC-/- colon carcinoma. Science 275, 1784–1787 (1997)
Davidson, G. et al. Casein kinase 1γ couples Wnt receptor activation to cytoplasmic signal transduction. Nature 438, 867–872 (2005)
Hino, S., Michiue, T., Asashima, M. & Kikuchi, A. Casein kinase Iε enhances the binding of Dvl-1 to Frat-1 and is essential for Wnt-3a-induced accumulation of β-catenin. J. Biol. Chem. 278, 14066–14073 (2003)
Beroukhim, R. et al. Assessing the significance of chromosomal aberrations in cancer: methodology and application to glioma. Proc. Natl Acad. Sci. USA 104, 20007–20012 (2007)
Weir, B. A. et al. Characterizing the cancer genome in lung adenocarcinoma. Nature 450, 893–898 (2007)
Martin, E. S. et al. Common and distinct genomic events in sporadic colorectal cancer and diverse cancer types. Cancer Res. 67, 10736–10743 (2007)
Tsafrir, D. et al. Relationship of gene expression and chromosomal abnormalities in colorectal cancer. Cancer Res. 66, 2129–2137 (2006)
Morris E. J. et al. E2F1 represses β-catenin transcription and is antagonized by both pRB and CDK8. Nature 10.1038/nature07310 (this issue).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl Acad. Sci. USA 102, 15545–15550 (2005)
Garraway, L. A. et al. Integrative genomic analyses identify MITF as a lineage survival oncogene amplified in malignant melanoma. Nature 436, 117–122 (2005)
Gold, M. O. & Rice, A. P. Targeting of CDK8 to a promoter-proximal RNA element demonstrates catalysis-dependent activation of gene expression. Nucleic Acids Res. 26, 3784–3788 (1998)
Ilyas, M., Tomlinson, I. P., Rowan, A., Pignatelli, M. & Bodmer, W. F. β-Catenin mutations in cell lines established from human colorectal cancers. Proc. Natl Acad. Sci. USA 94, 10330–10334 (1997)
Sparks, A. B., Morin, P. J., Vogelstein, B. & Kinzler, K. W. Mutational analysis of the APC/β-catenin/Tcf pathway in colorectal cancer. Cancer Res. 58, 1130–1134 (1998)
Rowan, A. J. et al. APC mutations in sporadic colorectal tumors: A mutational “hotspot” and interdependence of the “two hits”. Proc. Natl Acad. Sci. USA 97, 3352–3357 (2000)
Tassan, J. P., Jaquenoud, M., Leopold, P., Schultz, S. J. & Nigg, E. A. Identification of human cyclin-dependent kinase 8, a putative protein kinase partner for cyclin C. Proc. Natl Acad. Sci. USA 92, 8871–8875 (1995)
Sansom, O. J. et al. Myc deletion rescues Apc deficiency in the small intestine. Nature 446, 676–679 (2007)
Murakami, T. et al. Constitutive activation of Wnt/β-catenin signaling pathway in migration-active melanoma cells: role of LEF-1 in melanoma with increased metastatic potential. Biochem. Biophys. Res. Commun. 288, 8–15 (2001)
He, T. C. et al. Identification of c-MYC as a target of the APC pathway. Science 281, 1509–1512 (1998)
Fryer, C. J., White, J. B. & Jones, K. A. Mastermind recruits CycC:CDK8 to phosphorylate the Notch ICD and coordinate activation with turnover. Mol. Cell 16, 509–520 (2004)
Yook, J. I. et al. A Wnt–Axin2–GSK3β cascade regulates Snail1 activity in breast cancer cells. Nature Cell Biol. 8, 1398–1406 (2006)
Chiang, M. Y. et al. Identification of a conserved negative regulatory sequence that influences the leukemogenic activity of NOTCH1. Mol. Cell. Biol. 26, 6261–6271 (2006)
Kolligs, F. T., Hu, G., Dang, C. V. & Fearon, E. R. Neoplastic transformation of RK3E by mutant β-catenin requires deregulation of Tcf/Lef transcription but not activation of c-myc expression. Mol. Cell. Biol. 19, 5696–5706 (1999)
Boehm, J. S. et al. Integrative genomic approaches identify IKBKE as a breast cancer oncogene. Cell 129, 1065–1079 (2007)
Beroukhim, R. et al. Assessing the significance of chromosomal aberrations in cancer: methodology and application to glioma. Proc. Natl Acad. Sci. USA 104, 20007–20012 (2007)
Damalas, A. et al. Deregulated β-catenin induces a p53- and ARF-dependent growth arrest and cooperates with Ras in transformation. EMBO J. 20, 4912–4922 (2001)
Weir, B. A. et al. Characterizing the cancer genome in lung adenocarcinoma. Nature 450, 893–898 (2007)
Acknowledgements
We thank E. Shin for technical assistance with immunohistochemistry, M. Miri for assistance with sample collection and G. Getz for assistance with SNP array analysis. This work was supported in part by a grant from the US NCI/NIH R33CA128625 (W.C.H.), T32 NIH grant (R.F.) and a GI SPORE Career Development Grant (P50CA127003; R.F.), a Harvard-MIT Clinical Investigator Training Program Fellowship (A.J.B), Department of Defense Prostate Cancer Postdoctoral Fellowships (I.G. and S.Y.K.), Warren-Whitman-Richardson, Hagerty Foundation Research Fellowships (I.F.D.) and K12 award (M.G.C.). J.B. is a Beatriu de Pinos Fellow of the Departament d’Educació i Universitats de la Generalitat de Catalunya.
Author Contributions R.F., A.J.B., M.M. and W.C.H. designed the approach. R.F., A.J.B., E.B., E.F., I.F.D., I.G., J.S.B., N.V., S.J.S., S.O., S.Y.K. and Y.S. performed the experiments. J. Barretina, J. Baselga, J.T. and R.A.S. provided samples and analysis. A.H.L., A.J.B., C.M., C.S.F., D.E.R., E.B., E.F., I.F.D., I.G., J.A.C., M.G.C., M.L., M.M., P.T., R.A.S., R.F., S.F., S.J.S., S.O., S.Y.K. and W.C.H. analysed data. R.F., A.J.B., R.A.S. and W.C.H. wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
In accordance with Harvard Medical School guidelines, we disclose that W.C.H., M.M., M.L. and R.A.S. are consultants for Novartis.
Additional information
The SNP data can be found at http://research3.dfci.harvard.edu/cdk8colon/index.php.
Supplementary information
Supplementary Information
This file contains Supplementary Methods, Supplementary Figures 1-6 with legends, and legends for Supplementary Tables 1-6. (PDF 3727 kb)
Supplementary Table 1
This file contains Supplementary Table 1 (PDF 2184 kb)
Supplementary Table 2
This file contains Supplementary Table 2 (PDF 2312 kb)
Supplementary Table 3
This file contains Supplementary Table 3 (PDF 248 kb)
Supplementary Table 4
This file contains Supplementary Table 4 (PDF 278 kb)
Supplementary Table 5
This file contains Supplementary Table 5 (PDF 316 kb)
Supplementary Table 6
This file contains Supplementary Table 6 (PDF 262 kb)
Rights and permissions
About this article
Cite this article
Firestein, R., Bass, A., Kim, S. et al. CDK8 is a colorectal cancer oncogene that regulates β-catenin activity. Nature 455, 547–551 (2008). https://doi.org/10.1038/nature07179
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07179
This article is cited by
-
Hit discovery of potential CDK8 inhibitors and analysis of amino acid mutations for cancer therapy through computer-aided drug discovery
BMC Chemistry (2024)
-
Identifying regulators of aberrant stem cell and differentiation activity in colorectal cancer using a dual endogenous reporter system
Nature Communications (2024)
-
Non-hippo kinases: indispensable roles in YAP/TAZ signaling and implications in cancer therapy
Molecular Biology Reports (2023)
-
Cyclins and cyclin-dependent kinases: from biology to tumorigenesis and therapeutic opportunities
Journal of Cancer Research and Clinical Oncology (2023)
-
Discovery of small molecule degraders for modulating cell cycle
Frontiers of Medicine (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.