Abstract
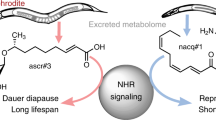
In many organisms, population-density sensing and sexual attraction rely on small-molecule-based signalling systems1,2. In the nematode Caenorhabditis elegans, population density is monitored through specific glycosides of the dideoxysugar ascarylose (the ‘ascarosides’) that promote entry into an alternative larval stage, the non-feeding and highly persistent dauer stage3,4. In addition, adult C. elegans males are attracted to hermaphrodites by a previously unidentified small-molecule signal5,6. Here we show, by means of combinatorial activity-guided fractionation of the C. elegans metabolome, that the mating signal consists of a synergistic blend of three dauer-inducing ascarosides, which we call ascr#2, ascr#3 and ascr#4. This blend of ascarosides acts as a potent male attractant at very low concentrations, whereas at the higher concentrations required for dauer formation the compounds no longer attract males and instead deter hermaphrodites. The ascarosides ascr#2 and ascr#3 carry different, but overlapping, information, as ascr#3 is more potent as a male attractant than ascr#2, whereas ascr#2 is slightly more potent than ascr#3 in promoting dauer formation7. We demonstrate that ascr#2, ascr#3 and ascr#4 are strongly synergistic, and that two types of neuron, the amphid single-ciliated sensory neuron type K (ASK) and the male-specific cephalic companion neuron (CEM), are required for male attraction by ascr#3. On the basis of these results, male attraction and dauer formation in C. elegans appear as alternative behavioural responses to a common set of signalling molecules. The ascaroside signalling system thus connects reproductive and developmental pathways and represents a unique example of structure- and concentration-dependent differential activity of signalling molecules.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Camilli, A. & Bassler, B. L. Bacterial small-molecule signaling pathways. Science 311, 1113–1116 (2006)
Dulac, C. & Torello, A. T. Molecular detection of pheromone signals in mammals: from genes to behaviour. Nature Rev. Neurosci. 4, 551–562 (2003)
Golden, J. W. & Riddle, D. L. A pheromone influences larval development in the nematode Caenorhabditis elegans . Science 218, 578–580 (1982)
Golden, J. W. & Riddle, D. L. A pheromone-induced developmental switch in Caenorhabditis elegans: Temperature-sensitive mutants reveal a wild-type temperature-dependent process. Proc. Natl Acad. Sci. USA 81, 819–823 (1984)
Simon, J. M. & Sternberg, P. W. Evidence of a mate-finding cue in the hermaphrodite nematode Caenorhabditis elegans . Proc. Natl Acad. Sci. USA 99, 1598–1603 (2002)
White, J. Q. et al. The sensory circuitry for sexual attraction in C. elegans males. Curr. Biol. 17, 1847–1857 (2007)
Butcher, R. A., Fujita, M., Schroeder, F. C. & Clardy, J. Small-molecule pheromones that control dauer development in Caenorhabditis elegans . Nature Chem. Biol. 3, 420–422 (2007)
Hirsh, D., Oppenheim, D. & Klass, M. Development of the reproductive system of Caenorhabditis elegans . Dev. Biol. 49, 200–219 (1976)
Brey, W. W. et al. Design, construction, and validation of a 1-mm triple-resonance high-temperature-superconducting probe for NMR. J. Magn. Reson. 179, 290–293 (2006)
Carde, R. T. & Elkinton, J. S. Field Trapping with Attractants: Methods and Interpretation (eds Hummel, H. E. & Miller, T. A.) 111–129 (Springer, 1984)
Chasnov, J. R., So, W. K., Chan, C. M. & Chow, K. L. The species, sex, and stage specificity of a Caenorhabditis sex pheromone. Proc. Natl Acad. Sci. USA 104, 6730–6735 (2007)
Jeong, P. Y. et al. Chemical structure and biological activity of the Caenorhabditis elegans dauer-inducing pheromone. Nature 433, 541–545 (2005)
Gershenzon, J. & Dudareva, N. The function of terpene natural products in the natural world. Nature Chem. Biol. 3, 408–414 (2007)
Lehar, J. et al. Chemical combination effects predict connectivity in biological systems. Mol. Syst. Biol. 3 10.1038/msb4100116 (2007)
Golden, J. W. & Riddle, D. L. A gene affecting production of the Caenorhabditis elegans dauer-inducing pheromone. Mol. Gen. Genet. 198, 534–536 (1985)
Cronin, C. J. et al. An automated system for measuring parameters of nematode sinusoidal movement. BMC Genet. 6 10.1186/1471-2156-6-5 (2005)
Collet, J., Spike, C. A., Lundquist, E. A., Shaw, J. E. & Herman, R. K. Analysis of osm-6, a gene that affects sensory cilium structure and sensory neuron function in Caenorhabditis elegans . Genetics 148, 187–200 (1998)
Bae, Y. K. et al. General and cell-type specific mechanisms target TRPP2/PKD-2 to cilia. Development 133, 3859–3870 (2006)
Tabish, M., Siddiqui, Z. K., Nishikawa, K. & Siddiqui, S. S. Exclusive expression of C. elegans osm-3 kinesin gene in chemosensory neurons open to the external environment. J. Mol. Biol. 247, 377–389 (1995)
Zwaal, R. R., Mendel, J. E., Sternberg, P. W. & Plasterk, R. H. Two neuronal G proteins are involved in chemosensation of the Caenorhabditis elegans Dauer-inducing pheromone. Genetics 145, 715–727 (1997)
Gems, D. & Riddle, D. L. Longevity in Caenorhabditis elegans reduced by mating but not gamete production. Nature 379, 723–725 (1996)
Partridge, L., Gems, D. & Withers, D. J. Sex and death: what is the connection? Cell 120, 461–472 (2005)
Tissenbaum, H. A. & Ruvkun, G. An insulin-like signaling pathway affects both longevity and reproduction in Caenorhabditis elegans . Genetics 148, 703–717 (1998)
Acknowledgements
This work was supported by the Human Frontiers Science Program (A.S.E., P.W.S. and P.E.A.T.), a US National Institutes of Health grant (P41 GM079571) to F.C.S., and the Howard Hughes Medical Institute, of which J.S. is an associate and P.W.S. an investigator. NMR data were collected in the UF-AMRIS facility; we thank J. Rocca for assistance. We thank E. Peden and D. Xue for the ceh-30 strains, C. J. Cronin and A. Choe for advice on behavioural assays, L. R. Baugh for liquid-culture dauer formation assays, B. Fox for assistance with the synthesis of ascr#2, ascr#3 and ascr#4, and M. de Bono, A. Dossey and M. Stadler for discussions. E. Hallem, J. Bungert and D. Hutchinson provided comments on the manuscript.
Author Contributions J.S. and P.W.S. designed the biological experiments and J.S. performed all the biological experiments; F.K. developed the procedure for collecting secreted worm metabolites and designed the chemical experiments and fractionation; F.K. and R.A. produced worm-conditioned water and performed chromatography; F.K., F.C.S., R.U.M., C.Z. and A.S.E. performed structure elucidation by NMR; H.T.A. performed structure elucidation by LC-MS; F.C.S. synthesized ascr#2, ascr#3 and ascr#4; and J.S., F.K., F.C.S., A.S.E., P.E.A.T. and P.W.S. analysed the data and wrote the paper.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Information 1
The file contains Supplementary Figures 1-15 with Legends, Supplementary Methods Supplementary Table 1 and legends to Supplementary Movies 1-2. (PDF 2761 kb)
nature07168-s2.mov
The file contains Supplementary Movie 1 (MOV 4557 kb)
nature07168-s3.mov
The file contains Supplementary Movie 2 (MOV 4154 kb)
Rights and permissions
About this article
Cite this article
Srinivasan, J., Kaplan, F., Ajredini, R. et al. A blend of small molecules regulates both mating and development in Caenorhabditis elegans. Nature 454, 1115–1118 (2008). https://doi.org/10.1038/nature07168
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07168
This article is cited by
-
The nematode-trapping fungus Arthrobotrys oligospora detects prey pheromones via G protein-coupled receptors
Nature Microbiology (2024)
-
Plant parasitic nematode-fungus interactions: recent concepts and mechanisms
Plant Physiology Reports (2024)
-
Investigation into the communication between unheated and heat-stressed Caenorhabditis elegans via volatile stress signals
Scientific Reports (2023)
-
Pathogenic bacteria modulate pheromone response to promote mating
Nature (2023)
-
Distinct neuropeptide-receptor modules regulate a sex-specific behavioral response to a pheromone
Communications Biology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.