Abstract
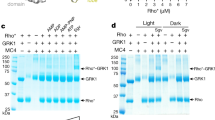
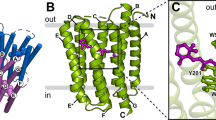
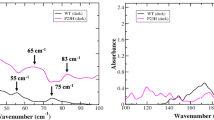
In the G-protein-coupled receptor (GPCR) rhodopsin, the inactivating ligand 11-cis-retinal is bound in the seven-transmembrane helix (TM) bundle and is cis/trans isomerized by light to form active metarhodopsin II. With metarhodopsin II decay, all-trans-retinal is released, and opsin is reloaded with new 11-cis-retinal. Here we present the crystal structure of ligand-free native opsin from bovine retinal rod cells at 2.9 ångström (Å) resolution. Compared to rhodopsin, opsin shows prominent structural changes in the conserved E(D)RY and NPxxY(x)5,6F regions and in TM5–TM7. At the cytoplasmic side, TM6 is tilted outwards by 6–7 Å, whereas the helix structure of TM5 is more elongated and close to TM6. These structural changes, some of which were attributed to an active GPCR state, reorganize the empty retinal-binding pocket to disclose two openings that may serve the entry and exit of retinal. The opsin structure sheds new light on ligand binding to GPCRs and on GPCR activation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Lagerstrom, M. C. & Schioth, H. B. Structural diversity of G protein-coupled receptors and significance for drug discovery. Nature Rev. Drug Discov. 7, 339–357 (2008)
Palczewski, K. G protein-coupled receptor rhodopsin. Annu. Rev. Biochem. 75, 743–767 (2006)
Okada, T., Ernst, O. P., Palczewski, K. & Hofmann, K. P. Activation of rhodopsin: new insights from structural and biochemical studies. Trends Biochem. Sci. 26, 318–324 (2001)
Farrens, D. L., Altenbach, C., Yang, K., Hubbell, W. L. & Khorana, H. G. Requirement of rigid-body motion of transmembrane helices for light activation of rhodopsin. Science 274, 768–770 (1996)
Sheikh, S. P., Zvyaga, T. A., Lichtarge, O., Sakmar, T. P. & Bourne, H. R. Rhodopsin activation blocked by metal-ion-binding sites linking transmembrane helices C and F. Nature 383, 347–350 (1996)
Fritze, O. et al. Role of the conserved NPxxY(x)5,6F motif in the rhodopsin ground state and during activation. Proc. Natl Acad. Sci. USA 100, 2290–2295 (2003)
Knierim, B., Hofmann, K. P., Ernst, O. P. & Hubbell, W. L. Sequence of late molecular events in the activation of rhodopsin. Proc. Natl Acad. Sci. USA 104, 20290–20295 (2007)
Menon, S. T., Han, M. & Sakmar, T. P. Rhodopsin: structural basis of molecular physiology. Physiol. Rev. 81, 1659–1688 (2001)
Lamb, T. D. & Pugh, E. N. Dark adaptation and the retinoid cycle of vision. Prog. Retin. Eye Res. 23, 307–380 (2004)
McBee, J. K., Palczewski, K., Baehr, W. & Pepperberg, D. R. Confronting complexity: the interlink of phototransduction and retinoid metabolism in the vertebrate retina. Prog. Retin. Eye Res. 20, 469–529 (2001)
Vogel, R. & Siebert, F. Conformations of the active and inactive states of opsin. J. Biol. Chem. 276, 38487–38493 (2001)
Palczewski, K. et al. Crystal structure of rhodopsin: A G protein-coupled receptor. Science 289, 739–745 (2000)
Okada, T. et al. The retinal conformation and its environment in rhodopsin in light of a new 2.2 Å crystal structure. J. Mol. Biol. 342, 571–583 (2004)
Li, J., Edwards, P. C., Burghammer, M., Villa, C. & Schertler, G. F. Structure of bovine rhodopsin in a trigonal crystal form. J. Mol. Biol. 343, 1409–1438 (2004)
Salom, D. et al. Crystal structure of a photoactivated deprotonated intermediate of rhodopsin. Proc. Natl Acad. Sci. USA 103, 16123–16128 (2006)
Standfuss, J. et al. Crystal structure of a thermally stable rhodopsin mutant. J. Mol. Biol. 372, 1179–1188 (2007)
Nakamichi, H. & Okada, T. Local peptide movement in the photoreaction intermediate of rhodopsin. Proc. Natl Acad. Sci. USA 103, 12729–12734 (2006)
Cherezov, V. et al. High-resolution crystal structure of an engineered human β2-adrenergic G protein-coupled receptor. Science 318, 1258–1265 (2007)
Rosenbaum, D. M. et al. GPCR engineering yields high-resolution structural insights into β2-adrenergic receptor function. Science 318, 1266–1273 (2007)
Rasmussen, S. G. et al. Crystal structure of the human β2 adrenergic G-protein-coupled receptor. Nature 450, 383–387 (2007)
Xie, G., Gross, A. K. & Oprian, D. D. An opsin mutant with increased thermal stability. Biochemistry 42, 1995–2001 (2003)
Okada, T., Takeda, K. & Kouyama, T. Highly selective separation of rhodopsin from bovine rod outer segment membranes using combination of divalent cation and alkyl(thio)glucoside. Photochem. Photobiol. 67, 495–499 (1998)
Krebs, A., Edwards, P. C., Villa, C., Li, J. & Schertler, G. F. The three-dimensional structure of bovine rhodopsin determined by electron cryomicroscopy. J. Biol. Chem. 278, 50217–50225 (2003)
Ruprecht, J. J., Mielke, T., Vogel, R., Villa, C. & Schertler, G. F. Electron crystallography reveals the structure of metarhodopsin I. EMBO J. 23, 3609–3620 (2004)
Liu, W., Eilers, M., Patel, A. B. & Smith, S. O. Helix packing moments reveal diversity and conservation in membrane protein structure. J. Mol. Biol. 337, 713–729 (2004)
Cohen, G. B., Oprian, D. D. & Robinson, P. R. Mechanism of activation and inactivation of opsin: role of Glu113 and Lys296. Biochemistry 31, 12592–12601 (1992)
Ballesteros, J. A. & Weinstein, H. Integrated methods for the construction of three-dimensional models and computational probing of structure–function relations in G-protein coupled receptors. Methods Neurosci. 25, 366–428 (1995)
Ballesteros, J. A. et al. Activation of the β2-adrenergic receptor involves disruption of an ionic lock between the cytoplasmic ends of transmembrane segments 3 and 6. J. Biol. Chem. 276, 29171–29177 (2001)
Filipek, S., Stenkamp, R. E., Teller, D. C. & Palczewski, K. G protein-coupled receptor rhodopsin: a prospectus. Annu. Rev. Physiol. 65, 851–879 (2003)
Crocker, E. et al. Location of Trp265 in metarhodopsin II: implications for the activation mechanism of the visual receptor rhodopsin. J. Mol. Biol. 357, 163–172 (2006)
Shi, L. et al. β2 adrenergic receptor activation. Modulation of the proline kink in transmembrane 6 by a rotamer toggle switch. J. Biol. Chem. 277, 40989–40996 (2002)
Lüdeke, S. et al. The role of Glu181 in the photoactivation of rhodopsin. J. Mol. Biol. 353, 345–356 (2005)
Schädel, S. A. et al. Ligand channeling within a G-protein-coupled receptor. The entry and exit of retinals in native opsin. J. Biol. Chem. 278, 24896–24903 (2003)
Heck, M. et al. Signaling states of rhodopsin. Formation of the storage form, metarhodopsin III, from active metarhodopsin II. J. Biol. Chem. 278, 3162–3169 (2003)
Bartl, F. J. & Vogel, R. Structural and functional properties of metarhodopsin III: Recent spectroscopic studies on deactivation pathways of rhodopsin. Phys. Chem. Chem. Phys. 9, 1648–1658 (2007)
Farrens, D. L. & Khorana, H. G. Structure and function in rhodopsin. Measurement of the rate of metarhodopsin II decay by fluorescence spectroscopy. J. Biol. Chem. 270, 5073–5076 (1995)
Hofmann, K. P., Pulvermüller, A., Buczylko, J., Van Hooser, P. & Palczewski, K. The role of arrestin and retinoids in the regeneration pathway of rhodopsin. J. Biol. Chem. 267, 15701–15706 (1992)
Jäger, S., Palczewski, K. & Hofmann, K. P. Opsin/all-trans-retinal complex activates transducin by different mechanisms than photolyzed rhodopsin. Biochemistry 35, 2901–2908 (1996)
Sachs, K., Maretzki, D., Meyer, C. K. & Hofmann, K. P. Diffusible ligand all-trans-retinal activates opsin via a palmitoylation-dependent mechanism. J. Biol. Chem. 275, 6189–6194 (2000)
De Lean, A., Stadel, J. M. & Lefkowitz, R. J. A ternary complex model explains the agonist-specific binding properties of the adenylate cyclase-coupled beta-adrenergic receptor. J. Biol. Chem. 255, 7108–7117 (1980)
Scheer, A., Fanelli, F., Costa, T., De Benedetti, P. G. & Cotecchia, S. The activation process of the α1B-adrenergic receptor: potential role of protonation and hydrophobicity of a highly conserved aspartate. Proc. Natl Acad. Sci. USA 94, 808–813 (1997)
Kefalov, V. J., Crouch, R. K. & Cornwall, M. C. Role of noncovalent binding of 11-cis-retinal to opsin in dark adaptation of rod and cone photoreceptors. Neuron 29, 749–755 (2001)
Sachs, K., Maretzki, D. & Hofmann, K. P. Assays for activation of opsin by all-trans-retinal. Methods Enzymol. 315, 238–251 (2000)
Jancarik, J. & Kim, S.-H. Sparse matrix sampling: a screening method for crystallization of proteins. J. Appl. Crystallogr. 24, 409–411 (1991)
Surya, A., Foster, K. W. & Knox, B. E. Transducin activation by the bovine opsin apoprotein. J. Biol. Chem. 270, 5024–5031 (1995)
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997)
Collaborative Computational Project, Number 4. The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994)
Brunger, A. T. et al. Crystallography & NMR system: A new software suite for macromolecular structure determination. Acta Crystallogr. D 54, 905–921 (1998)
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004)
Laskowski, R. A., MacArthur, M. W., Moss, D. S. & Thornton, J. M. PROCHECK: a program to check the stereochemical quality of protein structures. J. Appl. Crystallogr. 26, 283–291 (1993)
Hooft, R. W., Vriend, G., Sander, C. & Abola, E. E. Errors in protein structures. Nature 381, 272 (1996)
DeLano, W. L. The PyMOL Molecular Graphics System. <http://www.pymol.org> (2002)
Acknowledgements
We thank J. Engelmann and C. Koch for technical assistance; Y. Li for help in the early stage of the project; Y. J. Kim for help with data collection; M. Sommer, C. Enenkel and M. Heck for critically reading the manuscript; and N. Krauss for support and sharing his expertise in crystallography. We thank U. Müller and the scientific staff of the Protein Structure Factory and the Freie Universität Berlin at beamlines BL 14.1 and BL 14.2 at BESSY for continuous support of the project. This work was supported by the Deutsche Forschungsgemeinschaft Sfb449 (to O.P.E.), Sfb740 (to O.P.E. and K.P.H.), DFG-KOSEF international cooperation ER 294/1-1 (to O.P.E.) and F01-2004-000-10054-0 (to H.-W.C.). H-.W.C. was supported by CBNU funds for overseas research 2006–2007.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Information
This file contains Supplementary Table 1, Supplementary Discussion and Supplementary Figure 1. Supplementary Table 1 includes X-ray data collection and refinement statistics. A model of rhodopsin regeneration shown in Supplementary Figure 1 is discussed. (PDF 468 kb)
Rights and permissions
About this article
Cite this article
Park, J., Scheerer, P., Hofmann, K. et al. Crystal structure of the ligand-free G-protein-coupled receptor opsin. Nature 454, 183–187 (2008). https://doi.org/10.1038/nature07063
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07063
This article is cited by
-
Interdisciplinary biophysical studies of membrane proteins bacteriorhodopsin and rhodopsin
Biophysical Reviews (2023)
-
New insights into the molecular mechanism of rhodopsin retinitis pigmentosa from the biochemical and functional characterization of G90V, Y102H and I307N mutations
Cellular and Molecular Life Sciences (2022)
-
Characteristic structural difference between inactive and active states of orexin 2 receptor determined using molecular dynamics simulations
Biophysical Reviews (2022)
-
Identification of OPN3 as associated with non-syndromic oligodontia in a Japanese population
Journal of Human Genetics (2021)
-
Structural aspects of rod opsin and their implication in genetic diseases
Pflügers Archiv - European Journal of Physiology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.