Abstract
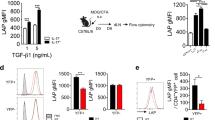
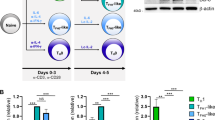
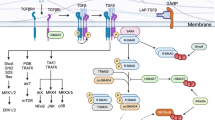
The recent discovery of CD4+ T cells characterized by secretion of interleukin (IL)-17 (TH17 cells) and the naturally occurring regulatory FOXP3+ CD4 T cell (nTreg) has had a major impact on our understanding of immune processes not readily explained by the TH1/TH2 paradigm. TH17 and nTreg cells have been implicated in the pathogenesis of human autoimmune diseases, including multiple sclerosis, rheumatoid arthritis, inflammatory bowel disease and psoriasis1,2. Our recent data and the work of others demonstrated that transforming growth factor-β (TGF-β) and IL-6 are responsible for the differentiation of naive mouse T cells into TH17 cells, and it has been proposed that IL-23 may have a critical role in stabilization of the TH17 phenotype3,4,5. A second pathway has been discovered in which a combination of TGF-β and IL-21 is capable of inducing differentiation of mouse TH17 cells in the absence of IL-6 (refs 6–8). However, TGF-β and IL-6 are not capable of differentiating human TH17 cells2,9 and it has been suggested that TGF-β may in fact suppress the generation of human TH17 cells10. Instead, it has been recently shown that the cytokines IL-1β, IL-6 and IL-23 are capable of driving IL-17 secretion in short-term CD4+ T cell lines isolated from human peripheral blood11, although the factors required for differentiation of naive human CD4 to TH17 cells are still unknown. Here we confirm that whereas IL-1β and IL-6 induce IL-17A secretion from human central memory CD4+ T cells, TGF-β and IL-21 uniquely promote the differentiation of human naive CD4+ T cells into TH17 cells accompanied by expression of the transcription factor RORC2. These data will allow the investigation of this new population of TH17 cells in human inflammatory disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Kikly, K., Liu, L., Na, S. & Sedgwick, J. D. The IL-23/Th(17) axis: therapeutic targets for autoimmune inflammation. Curr. Opin. Immunol. 18, 670–675 (2006)
Wilson, N. J. et al. Development, cytokine profile and function of human interleukin 17-producing helper T cells. Nature Immunol. 8, 950–957 (2007)
Bettelli, E. et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 441, 235–238 (2006)
Mangan, P. R. et al. Transforming growth factor-β induces development of the TH17 lineage. Nature 441, 231–234 (2006)
Veldhoen, M., Hocking, R. J., Atkins, C. J., Locksley, R. M. & Stockinger, B. TGFβ in the context of an inflammatory cytokine milieu supports de novo differentiation of IL-17-producing T cells. Immunity 24, 179–189 (2006)
Korn, T. et al. IL-21 initiates an alternative pathway to induce proinflammatory TH17 cells. Nature 448, 484–487 (2007)
Nurieva, R. et al. Essential autocrine regulation by IL-21 in the generation of inflammatory T cells. Nature 448, 480–483 (2007)
Zhou, L. et al. IL-6 programs TH-17 cell differentiation by promoting sequential engagement of the IL-21 and IL-23 pathways. Nature Immunol. 8, 967–974 (2007)
Acosta-Rodriguez, E. V., Napolitani, G., Lanzavecchia, A. & Sallusto, F. Interleukins 1β and 6 but not transforming growth factor-β are essential for the differentiation of interleukin 17-producing human T helper cells. Nature Immunol. 8, 942–949 (2007)
Evans, H. G., Suddason, T., Jackson, I., Taams, L. S. & Lord, G. M. Optimal induction of T helper 17 cells in humans requires T cell receptor ligation in the context of Toll-like receptor-activated monocytes. Proc. Natl Acad. Sci. USA 104, 17034–17039 (2007)
Laurence, A. & O’Shea, J. J. TH-17 differentiation: of mice and men. Nature Immunol. 8, 903–905 (2007)
McGeachy, M. J. et al. TGF-β and IL-6 drive the production of IL-17 and IL-10 by T cells and restrain TH-17 cell-mediated pathology. Nature Immunol. 8, 1390–1397 (2007)
Yang, X. O. et al. T helper 17 lineage differentiation is programmed by orphan nuclear receptors RORα and RORγ. Immunity 28, 29–39 (2008)
Wei, L., Laurence, A., Elias, K. M. & O’Shea, J. J. IL-21 is produced by Th17 cells and drives IL-17 production in a STAT3-dependent manner. J. Biol. Chem. 282, 34605–34610 (2007)
Acknowledgements
We thank D. Kozoriz for assistance with flow cytometric cell sorting. Supported by grants from the NIH (D.A.H., V.K.K.), the National Multiple Sclerosis Society (D.A.H., V.K.K.), the Juvenile Diubetes Research Foundation (D.A.H., V.K.K.), and the American Cancer Society (D.E.A.). D.A.H. and V.K.K. are recipients of a Javit2 Investigator award from the NIH.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Methods
The file contains more detailed Supplementary Methods describing how to differentiate naïve human CD4+ T cells in the presence of TGF-β1 and IL-21. (PDF 89 kb)
Rights and permissions
About this article
Cite this article
Yang, L., Anderson, D., Baecher-Allan, C. et al. IL-21 and TGF-β are required for differentiation of human TH17 cells. Nature 454, 350–352 (2008). https://doi.org/10.1038/nature07021
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07021
This article is cited by
-
Lymphocyte apoptosis and its association with the inflammatory markers and disease severity in juvenile-onset systemic lupus erythematosus patients
Pediatric Rheumatology (2024)
-
Role and Interrelationship Between Myeloid-Derived Suppressor Cells and CD4+ T Cells in Different Types of Infections: A Review
Proceedings of the Zoological Society (2024)
-
Metabolites of Trichoderma harzianum re-balance T-reg/Th-17 cytokine axis in epileptic rats
Beni-Suef University Journal of Basic and Applied Sciences (2023)
-
Differentiation of IL-26+ TH17 intermediates into IL-17A producers via epithelial crosstalk in psoriasis
Nature Communications (2023)
-
Transforming growth factors β and their signaling pathway in renal cell carcinoma and peritumoral space—transcriptome analysis
Clinical and Translational Oncology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.