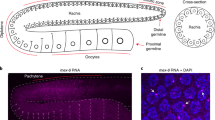
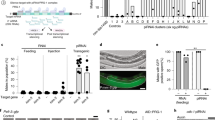
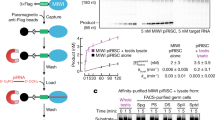
Abstract
In contrast to microRNAs and Piwi-associated RNAs, short interfering RNAs (siRNAs) are seemingly dispensable for host-directed gene regulation in Drosophila. This notion is based on the fact that mutants lacking the core siRNA-generating enzyme Dicer-2 or the predominant siRNA effector Argonaute 2 are viable, fertile and of relatively normal morphology1,2. Moreover, endogenous Drosophila siRNAs have not yet been identified. Here we report that siRNAs derived from long hairpin RNA genes (hpRNAs) programme Slicer complexes that can repress endogenous target transcripts. The Drosophila hpRNA pathway is a hybrid mechanism that combines canonical RNA interference factors (Dicer-2, Hen1 (known as CG12367) and Argonaute 2) with a canonical microRNA factor (Loquacious) to generate ∼21-nucleotide siRNAs. These novel regulatory RNAs reveal unexpected complexity in the sorting of small RNAs, and open a window onto the biological usage of endogenous RNA interference in Drosophila.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Lee, Y. S. et al. Distinct roles for Drosophila Dicer-1 and Dicer-2 in the siRNA/miRNA silencing pathways. Cell 117, 69–81 (2004)
Okamura, K., Ishizuka, A., Siomi, H. & Siomi, M. C. Distinct roles for Argonaute proteins in small RNA-directed RNA cleavage pathways. Genes Dev. 18, 1655–1666 (2004)
Kennerdell, J. R. & Carthew, R. W. Heritable gene silencing in Drosophila using double-stranded RNA. Nature Biotechnol. 18, 896–898 (2000)
Rice, P., Longden, I. & Bleasby, A. EMBOSS: the European Molecular Biology Open Software Suite. Trends Genet. 16, 276–277 (2000)
Wilson, R. J., Goodman, J. L. & Strelets, V. B. FlyBase: integration and improvements to query tools. Nucleic Acids Res. 36, D588–D593 (2008)
Tupy, J. L. et al. Identification of putative noncoding polyadenylated transcripts in Drosophila melanogaster . Proc. Natl Acad. Sci. USA 102, 5495–5500 (2005)
Jones-Rhoades, M. W., Bartel, D. P. & Bartel, B. MicroRNAs and their regulatory roles in plants. Annu. Rev. Plant Biol. 57, 19–53 (2006)
Liu, X. et al. Dicer-1, but not Loquacious, is critical for assembly of miRNA-induced silencing complexes. RNA 13, 2324–2329 (2007)
Leuschner, P. J., Ameres, S. L., Kueng, S. & Martinez, J. Cleavage of the siRNA passenger strand during RISC assembly in human cells. EMBO Rep. 7, 314–320 (2006)
Matranga, C., Tomari, Y., Shin, C., Bartel, D. P. & Zamore, P. D. Passenger-strand cleavage facilitates assembly of siRNA into Ago2-containing RNAi enzyme complexes. Cell 123, 607–620 (2005)
Diederichs, S. & Haber, D. A. Dual role for Argonautes in microRNA processing and posttranscriptional regulation of microRNA expression. Cell 131, 1097–1108 (2007)
Grishok, A. et al. Genes and mechanisms related to RNA interference regulate expression of the small temporal RNAs that control C. elegans developmental timing. Cell 106, 23–34 (2001)
Vagin, V. V. et al. A distinct small RNA pathway silences selfish genetic elements in the germline. Science 313, 320–324 (2006)
Saito, K. et al. Pimet, the Drosophila homolog of HEN1, mediates 2'-O-methylation of Piwi- interacting RNAs at their 3′ ends. Genes Dev. 21, 1603–1608 (2007)
Horwich, M. D. et al. The Drosophila RNA methyltransferase, DmHen1, modifies germline piRNAs and single-stranded siRNAs in RISC. Curr. Biol. 17, 1265–1272 (2007)
Seitz, H., Ghildiyal, M. & Zamore, P. D. Argonaute loading improves the 5′ precision of both microRNAs and their miRNA strands in flies. Curr. Biol. 18, 147–151 (2008)
Forstemann, K., Horwich, M. D., Wee, L., Tomari, Y. & Zamore, P. D. Drosophila microRNAs are sorted into functionally distinct Argonaute complexes after production by Dicer-1. Cell 130, 287–297 (2007)
Harris, P. V. et al. Molecular cloning of Drosophila mus308, a gene involved in DNA cross-link repair with homology to prokaryotic DNA polymerase I genes. Mol. Cell. Biol. 16, 5764–5771 (1996)
Dunoyer, P., Himber, C. & Voinnet, O. DICER-LIKE 4 is required for RNA interference and produces the 21-nucleotide small interfering RNA component of the plant cell-to-cell silencing signal. Nature Genet. 37, 1356–1360 (2005)
Rajagopalan, R., Vaucheret, H., Trejo, J. & Bartel, D. P. A diverse and evolutionarily fluid set of microRNAs in Arabidopsis thaliana . Genes Dev. 20, 3407–3425 (2006)
Reinhart, B. J., Weinstein, E. G., Rhoades, M. W., Bartel, B. & Bartel, D. P. MicroRNAs in plants. Genes Dev. 16, 1616–1626 (2002)
Ghildiyal, M. et al. Endogenous siRNAs derived from transposons and mRNAs in Drosophila somatic cells. Science 10.1126/science.1157396; published online 10 April 2008
Czech, B. et al. An endogenous small interfering RNA pathway in Drosophila . Nature 10.1038/nature07007 (this issue)
Kawamura, Y. et al. Drosophila endogenous small RNAs bind to Argonaute 2 in somatic cells. Nature 10.1038/nature06938 (this issue)
Okamura, K., Hagen, J. W., Duan, H., Tyler, D. M. & Lai, E. C. The mirtron pathway generates microRNA-class regulatory RNAs in Drosophila . Cell 130, 89–100 (2007)
Stark, A., Brennecke, J., Russell, R. B. & Cohen, S. M. Identification of Drosophila microRNA targets. PLoS Biol. 1, E60 (2003)
Ruby, J. G. et al. Evolution, biogenesis, expression, and target predictions of a substantially expanded set of Drosophila microRNAs. Genome Res. 17, 1850–1864 (2007)
Karolchik, D. et al. The UCSC Genome Browser Database: 2008 update. Nucleic Acids Res. 36, D773–D779 (2008)
Forstemann, K. et al. Normal microRNA maturation and germ-line stem cell maintenance requires Loquacious, a double-stranded RNA-binding domain protein. PLoS Biol. 3, e236 (2005)
Park, J. K., Liu, X., Strauss, T. J., McKearin, D. M. & Liu, Q. The miRNA pathway intrinsically controls self-renewal of Drosophila germline stem cells. Curr. Biol. 17, 533–538 (2007)
Acknowledgements
We are grateful to R. Carthew, Q. Liu, H. Siomi and S. Cohen for plasmids and Drosophila strains. K.O. was supported by the Charles H. Revson Foundation. H.G. was supported by A*STAR, Singapore. D.P.B. is an HHMI investigator, and work in his laboratory was supported by a grant from the NIH (GM067031). E.C.L. was supported by grants from the Leukemia and Lymphoma Foundation, the Burroughs Wellcome Foundation, the V Foundation for Cancer Research, the Sidney Kimmel Foundation for Cancer Research, and the NIH (GM083300).
Author Contributions J.G.R. identified hp-CG4068 and hpRNA1. W.-J.C. performed the EINVERTED analysis and identified the additional hpRNA loci and their targets. H.G. performed initial hpRNA northern analysis; all other experiments were designed and carried out by K.O. All authors contributed to the preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Figures
The file contains Supplementary Figures S1-S13 with Legends. (PDF 2913 kb)
Rights and permissions
About this article
Cite this article
Okamura, K., Chung, WJ., Ruby, J. et al. The Drosophila hairpin RNA pathway generates endogenous short interfering RNAs. Nature 453, 803–806 (2008). https://doi.org/10.1038/nature07015
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07015
This article is cited by
-
Mitochondrially mediated RNA interference, a retrograde signaling system affecting nuclear gene expression
Heredity (2023)
-
Low dose rate γ-irradiation protects fruit fly chromosomes from double strand breaks and telomere fusions by reducing the esi-RNA biogenesis factor Loquacious
Communications Biology (2022)
-
Structure of the Dicer-2–R2D2 heterodimer bound to a small RNA duplex
Nature (2022)
-
Single-molecule analysis of processive double-stranded RNA cleavage by Drosophila Dicer-2
Nature Communications (2021)
-
Conserved chromosomal functions of RNA interference
Nature Reviews Genetics (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.