Abstract
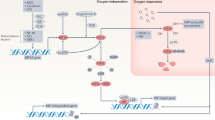
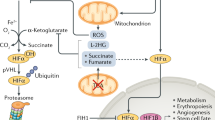
The hypoxic response is an ancient stress response triggered by low ambient oxygen (O2) (ref. 1) and controlled by hypoxia-inducible transcription factor-1 (HIF-1), whose α subunit is rapidly degraded under normoxia but stabilized when O2-dependent prolyl hydroxylases (PHDs) that target its O2-dependent degradation domain are inhibited2,3,4. Thus, the amount of HIF-1α, which controls genes involved in energy metabolism and angiogenesis, is regulated post-translationally. Another ancient stress response is the innate immune response, regulated by several transcription factors, among which NF-κB plays a central role5,6. NF-κB activation is controlled by IκB kinases (IKK), mainly IKK-β, needed for phosphorylation-induced degradation of IκB inhibitors in response to infection and inflammation7. IKK-β is modestly activated in hypoxic cell cultures when PHDs that attenuate its activation are inhibited8. However, defining the relationship between NF-κB and HIF-1α has proven elusive. Using in vitro systems, it was reported that HIF-1α activates NF-κB9, that NF-κB controls HIF-1α transcription10 and that HIF-1α activation may be concurrent with inhibition of NF-κB11. Here we show, with the use of mice lacking IKK-β in different cell types, that NF-κB is a critical transcriptional activator of HIF-1α and that basal NF-κB activity is required for HIF-1α protein accumulation under hypoxia in cultured cells and in the liver and brain of hypoxic animals. IKK-β deficiency results in defective induction of HIF-1α target genes including vascular endothelial growth factor. IKK-β is also essential for HIF-1α accumulation in macrophages experiencing a bacterial infection. Hence, IKK-β is an important physiological contributor to the hypoxic response, linking it to innate immunity and inflammation.
Similar content being viewed by others
Main
Hypoxia is characterized by a decreased O2 tension within cells and can occur under several pathophysiological situations including ischaemia, cancer and inflammation12. During ischaemia, the flow of nutrients and O2 to damaged tissues is decreased and HIF-1α activation induces genes whose products restore blood supply, nutrients and energy production, thereby maintaining tissue integrity and homeostasis13,14. The hypoxic response is important for the proper function of tissue macrophages and infiltrating neutrophils that encounter low O2 tension in infected tissues and after bacterial replication15. HIF-1α was also suggested to promote the expression of inflammatory cytokines, which are known to be regulated by NF-κB16, in lipopolysaccharide (LPS)-stimulated macrophages17 and mediate NF-κB activation in anoxic neutrophils9. However, it was also reported that hypoxia leads to modest IKK-β activation by inhibiting PHDs that negatively modulate IKK-β activity8. We therefore decided to critically explore the relationship between IKK-β, NF-κB and HIF-1α under in vivo conditions in IKK-β-deficient mice and primary macrophages.
We first examined bone marrow-derived macrophages (BMDM) from either IKKβF/F or IKKβF/F/Mx1Cre mice challenged with poly(I)˙poly(C), which induces interferon (IFN) and thereby drives CRE recombinase expression from the Mx1 promoter to delete IKKβ in IFN-responsive cells of the resulting IKKβΔ mice18. BMDM were incubated with Gram-positive (group A Streptococcus; GAS) and with Gram-negative (Pseudomonas aeruginosa) bacteria. Both species induced HIF-1α accumulation in an IKK-β-dependent manner (Fig. 1a). The induction of HIF-1 target genes involved in the hypoxic and innate immune responses was also dependent on IKK-β (Fig. 1b). These genes included Cox-2, which is directly regulated by NF-κB and HIF-1α, Cnlp, which encodes the murine antimicrobial peptide mCRAMP, whose expression is not directly responsive to NF-κB19, and Glut-1, encoding a glucose transporter. Moreover, Hif1a mRNA was markedly downregulated in IKK-β-deficient cells even before infection (Fig. 1b). IκB degradation and the nuclear accumulation of RelA/NF-κB preceded HIF-1α expression (Fig. 1c), indicating that NF-κB may control Hif1a gene transcription. Indeed, chromatin immunoprecipitation (ChIP) in LPS-stimulated macrophages revealed that RelA is recruited to the Hif1a promoter, which contains a classical κB site at -197/-188 base pairs, conserved between mice and humans (Fig. 1d). Furthermore, the basal levels of Hif1a mRNA were decreased in RelA-deficient fibroblasts even under resting conditions (Supplementary Fig. 1), suggesting that NF-κB activity is required for effective Hif1a mRNA expression even in non-stimulated cells.
a, BMDM from either IKKβF/F (IKKβ+/+) or poly(I)˙poly(C)-injected IKKβF/F/Mx1-Cre (IKKβΔ ; IKKβ-/-) mice were incubated with either with GAS or P. aeruginosa (MOI of 10 for 4 h). Expression of the indicated proteins was analysed by immunoblotting. b, RNA was extracted from BMDM incubated with GAS and gene expression was analysed by quantitative RT–PCR. Results are averages of three separate experiments done in triplicate, and are shown as means and s.e.m. Values are normalized relative to 18S rRNA. Light grey bars, control; dark grey bars, GAS. Cramp, gene encoding cathelicidin antimicrobial peptide. c, RAW264.7 macrophages were incubated with GAS and protein expression was analysed by immunoblotting at the indicated time points. d, ChIP was performed with an anti-RelA antibody using fixed and sheared chromatin isolated from RAW264.7 mouse macrophages incubated for 1 h with or without LPS. The Hif1a promoter fragment, which contains a κB site at -197/-188 base pairs, was detected by PCR amplification.
As found elsewhere8, hypoxia modestly activated IKK in macrophages (Fig. 2a), induced the phosphorylation of IKK-α/β and IκBα and promoted IκBα degradation (Fig. 2b). Hypoxia also induced the nuclear translocation of RelA, which preceded HIF-1α accumulation (Fig. 2c), as occurred in bacteria-infected macrophages (Fig. 1c). Binding of NF-κB to a canonical κB DNA site was also induced by hypoxia (Fig. 2d). We examined whether IKK-β was required for hypoxia-induced HIF-1α accumulation, a response that is thought to be dependent mainly on inhibition of HIF-1α degradation3,4. IKK-β was required for the optimal accumulation of HIF-1α, but not of HIF-2α, in BMDM incubated with the hypoxia mimetic desferrioxamine (DFX) as well as in response to actual hypoxia (Fig. 3a, b). IKK-β also did not affect HIF-2α expression in infected macrophages (Fig. 1a). The overexpression of a non-degradable IκBα (IκB superrepressor) also blocked HIF-1α accumulation induced by hypoxia in HEK-293 cells (Supplementary Fig. 2). The hypoxia-dependent induction of HIF-1 target genes, such as those encoding vascular endothelial growth factor (VEGF) and GLUT-1, was nearly abolished in IKK-β-deficient macrophages (Fig. 3c) or fibroblasts (Supplementary Fig. 3). Expression of Hif1a, but not Hif2α, mRNA was substantially decreased in the absence of IKK-β even under normoxia (Fig. 3c), further supporting the notion that basal NF-κB activity is required for the expression of enough Hif1a mRNA at all times to result in the rapid accumulation of HIF-1α protein, which occurs only under hypoxic conditions. Activation of NF-κB by LPS induced Hif1a promoter activity (Supplementary Fig. 4), elevated HIF-1α expression in hypoxic cells (Fig. 3d) and potentiated the induction of Vegf mRNA (Supplementary Fig. 5). Despite substantial expression of Hif1a mRNA in LPS-stimulated normoxic macrophages (Supplementary Fig. 5), these cells do not accumulate HIF-1α protein (Fig. 3d), which echoes findings in T cells stimulated with anti-CD3 antibody20. Hence, NF-κB activation without hypoxic inhibition of PHDs is insufficient for HIF-1α protein accumulation. In mouse fibroblasts, IKK-β was required for basal Hif1a promoter activity and its stimulation by treatment with DFX (Fig. 3e).
RAW264.7 macrophages were incubated with or without LPS under normoxia or were placed under hypoxia (0.5% O2). a, At the indicated time points, IKK activity was measured by an immunocomplex kinase assay with GST–IκBα as a substrate. b, Cell lysates were prepared and IKK-β and IκBα phosphorylation (P) and amounts were analysed by immunoblotting. c, Nuclear extracts were prepared at the indicated time points and analysed by immunoblotting for the nuclear accumulation of RelA and HIF-1α. d, Nuclear extracts were prepared after 2 h of stimulation with LPS or hypoxia, and binding activity for DNA for NF-κB was examined by a mobility-shift assay. Antibody inhibition was performed with an anti-RelA antibody.
a, BMDM from IKKβF/F (IKK-β+/+) or IKKβΔ (IKK-β-/-) mice were incubated with DFX for 4 h. Expression of the indicated proteins was analysed by immunoblotting for nuclear (NE) and cytosolic (CE) extracts. b, BMDM were obtained as above and cultured under normoxia or hypoxia (0.5% O2 for 4 h). Protein expression was analysed by immunoblotting. c, BMDM were treated as above and mRNA expression was analysed by quantitative RT–PCR. Light grey bars, normoxia; dark grey bars, hypoxia. Results are means and s.e.m. for three separate experiments performed in triplicate. *, P < 0.05 versus normoxic IKKβ+/+ cells; †, P < 0.05 versus hypoxic IKKβ+/+ cells. PGK, phosphoglucokinase; iNOS, inducible nitric oxide synthase. d, RAW264.7 macrophages were cultured in the absence or presence of LPS under the indicated O2 tensions for 2 h. Protein expression was analysed by immunoblotting. e, Murine embryonic fibroblasts from IKKβ+/+ or IKKβ-/- embryos were transfected with a luciferase reporter gene driven by the Hif1a promoter. After 36 h the cells were incubated for 3 h with DFX. Light grey bars, control; dark grey bars, DFX. Results are means and s.e.m. for three separate experiments performed in triplicate.
We next examined the role of IKK-β in HIF-1 activation in intact mice. Administration of DFX induced HIF-1α expression in liver of IKKβF/F mice but not in IKKβΔ mice (Fig. 4a), which lack IKK-β in both hepatocytes and Kupffer cells21. IKKβΔ mice also contained less Hif1a and Vegf mRNA in their livers (Fig. 4b). We also examined the role of IKK-β in the response to actual hypoxia. Mice were placed in a chamber with an ambient O2 concentration of 8% (thus mimicking an altitude of 7,000 m (ref. 22)). Under these conditions we observed hypoxia-induced HIF-1α accumulation in liver (Fig. 4c) and brain (Fig. 4d) and in both cases it was dependent on IKK-β in CRE-expressing cells. In the brain the predominant CRE-expressing cells were astrocytes (Supplementary Fig. 6) and not neurons (data not shown), thus explaining the partial deletion of IKK-β in this tissue (Fig. 4d). Despite this, hypoxia-induced VEGF protein (Fig. 4e) and Vegf mRNA (Fig. 4f) were IKK-β dependent. IKKβΔ mice showed a profound increase in cerebellar astrocyte activation, marked by glial fibrillary acidic protein, relative to IKKβF/F mice (Supplementary Fig. 7). This may have been due to defective production of VEGF, a cytokine with anti-inflammatory properties that has been shown to promote tissue repair23. VEGF is also a potent neuroprotective factor24 whose decreased production may potentiate hypoxia-induced neuronal damage and thereby augment astrocyte activation. This situation may be akin to a loss of IKK-β in intestinal epithelial cells, which has previously been found to exacerbate ischaemic damage to the intestinal mucosa25. These results suggest that IKK-β inhibitors may not be useful in the treatment of neuroinflammatory disorders.
IKKβF/F (CRE-) or IKKβΔ (CRE+) mice were treated with vehicle (control) or DFX (600 mg kg-1). After 15 h, livers were removed for protein (a) and RNA (b) analysis. a, HIF-1α and IKK-β expression in nuclear (NE) or cytosolic (CE) extracts was analysed by immunoblotting. b, Expression of HIF-1α and Vegf mRNA was examined by quantitative RT–PCR. Light grey bars, control; dark grey bars, DFX. Results are means and s.e.m. (n = 3). Values are normalized relative to 18S rRNA. *, P < 0.05 versus normoxic CRE- mice; †, P < 0.05 versus DFX-treated CRE- mice. c, d, IKKβF/F and IKKβΔ mice were kept under normoxia or hypoxia (8% O2) for 24 h and HIF-1α and IKK-β expression was analysed by immunoblotting of liver (c) or brain (d) nuclear and cytosolic extracts, respectively. e, VEGF expression in brain of mice from the above experiment was analysed by ELISA. Light grey bars, normoxia; dark grey bars, hypoxia. Results are means and s.e.m. (n = 3). *, P < 0.05 versus normoxic CRE- mice; †, P < 0.05 versus hypoxic CRE- mice. f, VEGF and Hif1a mRNA expression were analysed by quantitative RT–PCR of total brain RNA. Light grey bars, normoxia; dark grey bars, hypoxia. Results are means and s.e.m. *, P < 0.05 versus normoxic CRE- mice; †, P < 0.05 versus hypoxic CRE- mice (n = 3).
Although early studies demonstrated the induction of Hif1a mRNA in experimental animals during development and hypoxia26,27, numerous in vitro studies led to the current model that the accumulation of HIF-1α is regulated predominantly at the post-translational level through the inhibition of O2-dependent PHDs that drive HIF-1α degradation under normoxia3,4. Our results show clearly that transcriptional activation of the Hif1a gene by IKK-β-responsive NF-κB, which precedes HIF-1α protein accumulation, is of critical importance under pathophysiologically relevant conditions ex vivo and in vivo. Both macrophages infected with bacteria and mice subjected to hypoxia reveal a pronounced defect in HIF-1α expression on loss of IKK-β. The IKK-β/NF-κB–HIF-1α crosstalk is not critical during normal embryonic development, because the respective gene deletions result in different phenotypes. Whereas Hif1a-/- embryos die prematurely at embryonic day 9.5, mainly as a result of defects in neural fold closure and capillary development13,14, IKK-β-/- embryos die later, at embryonic day 13.5, from massive liver apoptosis driven by TNF28,29.
Previous findings identified a connection between HIF-1α and innate immunity and inflammation, but it was not clear how microbial infection or inflammation led to HIF-1α activation15,19. Our results, together with the previous finding that IKK-β catalytic activity is controlled by O2-sensitive PHDs8, establish NF-κB as a hypoxia-regulated transcription factor that controls Hif1a mRNA expression both under basal conditions and during hypoxia, thereby serving as a regulator of the hypoxic response. Our findings demonstrate that this depends on NF-κB activation, which controls Hif1a mRNA expression, but accumulation of HIF-1α protein requires hypoxia, which in bacterial infection may be due to depletion of intracellular oxygen by replicating bacteria. These findings have far-reaching physiological implications because they indicate the existence of coupling between two evolutionary ancient stress responses: innate immunity and the hypoxic response. By controlling HIF-1α activation in macrophages during microbial infections, which may lower local O2 tension, NF-κB can enhance glycolytic energy metabolism and the production of angiogenic factors, in addition to its well-established role in the expression of proinflammatory cytokines, chemokines and antimicrobial peptides. In addition to more effective execution of the host-defence response, the ability of NF-κB to promote HIF-1α activation expands its pro-survival function because the HIF-1-dependent hypoxic response is critical for providing cells and tissues undergoing ischaemia with sufficient energy supplies and allows them to resist cell death.
By serving as an essential component of the hypoxic response in vivo, IKK-β also performs a homeostatic function in the brain, an organ that is extremely sensitive to deprivation of oxygen and glucose30.
Methods Summary
To delete IKK-β in IKKβF/F/Mx1Cre mice, 250 μg of poly(I)˙poly(C) (Sigma) was injected intraperitoneally on three alternate days, three weeks before exposure to hypoxia or isolation of myeloid cells18. To induce hypoxia in vivo, mice were placed in a special chamber in which N2 and O2 were injected to achieve an O2 concentration of 8 ± 0.1%. This was controlled by the Oxycycler hydraulic system (Model A44x0; BioSpherix) and ANA-Win2 software (Version 2.4.17; Watlow Anafaze). Control mice were kept in the same room under normal atmospheric O2 and were exposed to the same level of noise and light during each experiment. After 24 h of normoxia or hypoxia, mice were killed and their livers and brains were rapidly removed and frozen in liquid N2 or OCT with a solid CO2/2-methylpropan-1-ol bath.
Online Methods
Quantitative RT–PCR
Total RNA was extracted with Trizol (Invitrogen) and reverse-transcribed with random hexamers and SuperScript II Kit (Invitrogen). Real-time PCR was performed with SYBR Green PCR Master Mix Kit (Applied Biosystems). The following primer pairs were used: VEGF, 5′-CCACGTCAGAGAGCAACATCA-3′ and 5′-TCATTCTCTCTATGTGCTGGCTTT-3′; PGK, 5′-GGAAGCGGGTCGTGATGA-3′ and 5′-GCCTTGATCCTTTGGTTGTTTG-3′; GLUT-1, 5′-CATCCTTATTGCCCAGGTGTTT-3′ and 5′-GAAGACGACACTGAGCAGCAGA-3′; iNOS, 5′-GGCAGCCTGTGAGACCTTTG-3′ and 5′-CATTGGAAGTGAAGCGTTTCG-3′; COX-2, 5′-GTGGAAAAACCTCGTCCAGA-3′ and 5′- GCTCGGCTTCCAGTATTGAG-3′; HIF-1α, 5′-ACAAGTCACCACAGGACAG-3′ and 5′-AGGGAGAAAATCAAGTCG-3′; HIF-2α, 5′-CAACCTGCAGCCTCAGTGTATC-3′ and 5′-CACCACGTCGTTCTTCTCGAT-3′; 18S rRNA, 5′-CGCCGCTAGAGGTGAAATTCT-3′ and 5′- CGAACCTCCGACTTTCGTTCT-3′.
Immunoblotting
Whole-cell extracts were obtained by lysing cells in 1% SDS, 10 mM Tris-HCl pH 7.4. Cytoplasmic and nuclear extracts were obtained as described2. Proteins were separated by SDS–PAGE and detected by immunoblotting. Blots were incubated with antibodies against phosphorylated IKK-α/β, phosphorylated IκBα, IKK-α, IKK-β, IκBα, RelA and histone H3 (all from Santa Cruz Biotechnology), actin (Sigma), HIF-1α, HIF-2α and HIF-1β (Novus).
Chromatin immunoprecipitation
Chromatin immunoprecipitation (ChIP) was performed with ChIP-IT Express Kit (Active Motif) in accordance with the manufacturer’s instructions. Chromatin was precipitated with RelA antibodies (Santa Cruz Biotechnology). Samples were analysed by PCR. The murine HIF-1α and actin promoters were amplified with the primer pairs 5′-CACCCCCATCTCCTTTCTCT-3′ and 5′-GGGTTCCTCGAGATCCAATG- 3′, and 5′-TGCACTGTGCGGCGAAGC-3′ and 5′-TCGAGCCATAAAAGGCAA-3′, respectively.
Luciferase assay
A murine HIF-1α-luciferase reporter, pHIF-1α/Luc, was kindly provided by S. W. Ebbinghaus. pHIF-1α/Luc was co-transfected with the internal control pRL-TK into either IKKβ+/+ or IKKβ-/- MEFs with Lipofectamine 2000 (Invitrogen). Luciferase activity was measured with the Dual-luciferase reporter assay system (Promega). Results are presented as relative reporter activity after normalization to the internal control pRL-TK.
Statistical analysis
Results are expressed as means and s.e.m. A Stat View II (Abacus Concepts) statistical package was used for all analyses: multiple groups were compared by one-factor analysis of variance, followed by Fisher’s protected least-squares difference to assess specific group differences.
References
Wang, G. L. & Semenza, G. L. General involvement of hypoxia-inducible factor 1 in transcriptional response to hypoxia. Proc. Natl Acad. Sci. USA 90, 4304–4308 (1993)
Maxwell, P. H. et al. The tumour suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis. Nature 399, 271–275 (1999)
Semenza, G. HIF-1, O2, and the 3 PHDs: how animal cells signal hypoxia to the nucleus. Cell 107, 1–3 (2001)
Schofield, C. J. & Ratcliffe, P. J. Oxygen sensing by HIF hydroxylase. Nature Rev. Mol. Cell Biol. 5, 343–354 (2004)
Akira, S., Uematsu, S. & Takeuchi, O. Pathogen recognition and innate immunity. Cell 124, 783–801 (2006)
Karin, M. & Ben-Neriah, Y. Phosphorylation meets ubiquitination: the control of NF-κB activity. Annu. Rev. Immunol. 18, 621–663 (2000)
Häcker, H. & Karin, M. Regulation and function of IKK and IKK-related kinases. Sci. STKE 357, re13 (2006)
Cummins, E. P. et al. Prolyl hydroxylase-1 negatively regulates IκB kinase-β, giving insight into hypoxia-induced NFκB activity. Proc. Natl Acad. Sci. USA 103, 18154–18159 (2006)
Walmsley, S. R. et al. Hypoxia-induced neutrophil survival is mediated by HIF-1α-dependent NF-κB activity. J. Exp. Med. 201, 105–115 (2005)
Belaiba, R. S. et al. Hypoxia up-regulates HIF-1α transcription by involving PI-3 kinase and NFκB in pulmonary artery smooth muscle cells. Mol. Biol. Cell 18, 4691–4697 (2007)
Carbia-Nagashima, A. et al. RSUME, a small RWD-containing protein, enhances SUMO conjugation and stabilizes HIF-1α during hypoxia. Cell 131, 309–323 (2007)
Paul, S. A., Simons, J. W. & Mabjeesh, N. J. HIF at the crossroads between ischemia and carcinogenesis. J. Cell. Physiol. 200, 20–30 (2004)
Ryan, H. E., Lo, J. & Johnson, R. S. HIF-1α is required for solid tumor formation and embryonic vascularization. EMBO J. 17, 3005–3015 (1998)
Iyer, N. V. et al. Cellular and developmental control of O2 homeostasis by hypoxia-inducible factor 1α. Genes Dev. 12, 149–162 (1998)
Cramer, T. et al. HIF-1α is essential for myeloid cell-mediated inflammation. Cell 112, 645–657 (2003)
Barnes, P. J. & Karin, M. Nuclear factor-κB: a pivotal transcription factor in chronic inflammatory diseases. N. Engl. J. Med. 336, 1066–1071 (1997)
Peyssonnaux, C. et al. Cutting edge: Essential role of hypoxia inducible factor-1α in development of lipopolysaccharide-induced sepsis. J. Immunol. 178, 7516–7519 (2007)
Greten, F. R. et al. NF-κB is a negative regulator of IL-1β secretion as revealed by genetic and pharmacological inhibition of IKKβ. Cell 130, 918–931 (2007)
Peyssonnaux, C. et al. HIF-1α expression regulates the bactericidal capacity of phagocytes. J. Clin. Invest. 115, 1806–1815 (2005)
Makino, Y. et al. Hypoxia-inducible factor regulates survival of antigen receptor-driven T cells. J. Immunol. 171, 6534–6540 (2003)
Maeda, S., Kamata, H., Luo, J. L., Leffert, H. & Karin, M. IKKβ couples hepatocyte death to cytokine-driven compensatory proliferation that promotes chemical hepatocarcinogenesis. Cell 121, 977–990 (2005)
Schoch, H. J., Fischer, S. & Marti, H. H. Hypoxia-induced vascular endothelial growth factor expression causes vascular leakage in the brain. Brain 125, 2549–2557 (2002)
Riboldi, E. et al. Cutting edge: proangiogenic properties of alternatively activated dendritic cells. J. Immunol. 175, 2788–2792 (2005)
Storkebaum, E., Lambrechts, D. & Carmeliet, P. VEGF: once regarded as a specific angiogenic factor, now implicated in neuroprotection. BioEssays 26, 943–954 (2004)
Chen, L. W. et al. The two faces of IKK and NF-κB inhibition: prevention of systemic inflammation but increased local injury following intestinal ischemia–reperfusion. Nature Med. 9, 575–581 (2003)
Jain, S., Maltepe, E., Lu, M. M., Simon, C. & Bradfield, C. A. Expression of ARNT, ARNT2, HIF1 α, HIF2 α and Ah receptor mRNAs in the developing mouse. Mech. Dev. 73, 117–123 (1998)
Elson, D. A., Ryan, H. E., Snow, J. W., Johnson, R. S. & Arbeit, J. M. Coordinate up-regulation of hypoxia inducible factor (HIF)-1α and HIF-1 target genes during multi-stage epidermal carcinogenesis and wound healing. Cancer Res. 60, 6189–6195 (2000)
Li, Z. W. et al. The IKKβ subunit of IκB kinase (IKK) is essential for nuclear factor κB activation and prevention of apoptosis. J. Exp. Med. 189, 1839–1845 (1999)
Li, Q., Van Antwerp, D., Mercurio, F., Lee, K. F. & Verma, I. M. Severe liver degeneration in mice lacking the IκB kinase 2 gene. Science 284, 321–325 (1999)
Leach, R. M. & Treacher, D. F. Oxygen transport-2. Tissue hypoxia. Br. Med. J. 317, 1370–1373 (1998)
Acknowledgements
J.R. and M.G. were supported by a postdoctoral fellowship from the Spanish Ministry of Education and Science. Work in the laboratories of M.K., R.S.J., K.A., V.N. and G.G.H. was supported by grants from the National Institutes of Health. M.K. is an American Cancer Society Research Professor.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary information
The file contains Supplementary Figures S1-S7 with Legends and additional references. (PDF 466 kb)
Rights and permissions
About this article
Cite this article
Rius, J., Guma, M., Schachtrup, C. et al. NF-κB links innate immunity to the hypoxic response through transcriptional regulation of HIF-1α. Nature 453, 807–811 (2008). https://doi.org/10.1038/nature06905
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature06905
This article is cited by
-
Bronchial epithelial transcriptomics and experimental validation reveal asthma severity-related neutrophilc signatures and potential treatments
Communications Biology (2024)
-
Expression analysis of lymphocyte subsets and lymphocyte-to-monocyte ratio: reveling immunosuppression and chronic inflammation in breast cancer
Journal of Cancer Research and Clinical Oncology (2024)
-
Submersion and hypoxia inhibit alveolar epithelial Na+ transport through ERK/NF-κB signaling pathway
Respiratory Research (2023)
-
TMT-based quantitative proteomics reveals the targets of andrographolide on LPS-induced liver injury
BMC Veterinary Research (2023)
-
Polo-like kinase 1 promotes pulmonary hypertension
Respiratory Research (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.