Abstract
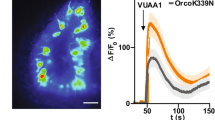
From worm to man, many odorant signals are perceived by the binding of volatile ligands to odorant receptors1 that belong to the G-protein-coupled receptor (GPCR) family2. They couple to heterotrimeric G-proteins, most of which induce cAMP production3. This second messenger then activates cyclic-nucleotide-gated ion channels to depolarize the olfactory receptor neuron, thus providing a signal for further neuronal processing. Recent findings, however, have challenged this concept of odorant signal transduction in insects, because their odorant receptors, which lack any sequence similarity to other GPCRs4, are composed of conventional odorant receptors (for example, Or22a), dimerized with a ubiquitously expressed chaperone protein5, such as Or83b in Drosophila6. Or83b has a structure akin to GPCRs, but has an inverted orientation in the plasma membrane4,7. However, G proteins are expressed in insect olfactory receptor neurons8, and olfactory perception is modified by mutations affecting the cAMP transduction pathway9. Here we show that application of odorants to mammalian cells co-expressing Or22a and Or83b results in non-selective cation currents activated by means of an ionotropic and a metabotropic pathway, and a subsequent increase in the intracellular Ca2+ concentration. Expression of Or83b alone leads to functional ion channels not directly responding to odorants, but being directly activated by intracellular cAMP or cGMP. Insect odorant receptors thus form ligand-gated channels as well as complexes of odorant-sensing units and cyclic-nucleotide-activated non-selective cation channels. Thereby, they provide rapid and transient as well as sensitive and prolonged odorant signalling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Buck, L. & Axel, R. A novel multigene family may encode odorant receptors: a molecular basis for odor recognition. Cell 65, 175–187 (1991)
Mombaerts, P. Seven-transmembrane proteins as odorant and chemosensory receptors. Science 286, 707–711 (1999)
Ronnett, G. V. & Moon, C. G proteins and olfactory signal transduction. Annu. Rev. Physiol. 64, 189–222 (2002)
Benton, R., Sachse, S., Michnick, S. W. & Vosshall, L. B. Atypical membrane topology and heteromeric function of Drosophila odorant receptors in vivo . PLoS Biol. 4, e20 (2006)
Neuhaus, E. M. et al. Odorant receptor heterodimerization in the olfactory system of Drosophila melanogaster . Nature Neurosci. 8, 15–17 (2005)
Larsson, M. C. et al. Or83b encodes a broadly expressed odorant receptor essential for Drosophila olfaction. Neuron 43, 703–714 (2004)
Wistrand, M., Kall, L. & Sonnhammer, E. L. A general model of G protein-coupled receptor sequences and its application to detect remote homologs. Protein Sci. 15, 509–521 (2006)
Rützler, M., Lu, T. & Zwiebel, L. J. Gα encoding gene family of the malaria vector mosquito Anopheles gambiae: expression analysis and immunolocalization of AGαq and AGαo in female antennae. J. Comp. Neurol. 499, 533–545 (2006)
Martin, F., Charro, M. J. & Alcorta, E. Mutations affecting the cAMP transduction pathway modify olfaction in Drosophila . J. Comp. Physiol. A 187, 359–370 (2001)
Wetzel, C. H. et al. Functional expression and characterization of a Drosophila odorant receptor in a heterologous cell system. Proc. Natl Acad. Sci. USA 98, 9377–9380 (2001)
Katada, S., Nakagawa, T., Kataoka, H. & Touhara, K. Odorant response assays for a heterologously expressed olfactory receptor. Biochem. Biophys. Res. Commun. 305, 964–969 (2003)
Dobritsa, A. A., van der Goes van Naters, W., Warr, C. G., Steinbrecht, R. A. & Carlson, J. R. Integrating the molecular and cellular basis of odor coding in the Drosophila antenna. Neuron 37, 827–841 (2003)
Pelz, D., Roeske, T., Syed, Z., de Bruyne, M. & Galizia, C. G. The molecular receptive range of an olfactory receptor in vivo (Drosophila melanogaster Or22a). J. Neurobiol. 66, 1544–1563 (2006)
Wicher, D. et al. Differential receptor activation by cockroach adipokinetic hormones produces differential effects on ion currents, neuronal activity and locomotion. J. Neurophysiol. 95, 2314–2325 (2006)
Barry, P. H. The relative contributions of cAMP and InsP3 pathways to olfactory responses in vertebrate olfactory receptor neurons and the specificity of odorants for both pathways. J. Gen. Physiol. 122, 247–250 (2003)
Ludwig, A. et al. Two pacemaker channels from human heart with profoundly different activation kinetics. EMBO J. 18, 2323–2329 (1999)
Dhallan, R. S., Yau, K. W., Schrader, K. A. & Reed, R. R. Primary structure and functional expression of a cyclic nucleotide-activated channel from olfactory neurons. Nature 347, 184–187 (1990)
Craven, K. B. & Zagotta, W. N. CNG and HCN channels: two peas, one pod. Annu. Rev. Physiol. 68, 375–401 (2006)
Hallem, E. A., Ho, M. G. & Carlson, J. R. The molecular basis of odor coding in the Drosophila antenna. Cell 117, 965–979 (2004)
Kaupp, U. B. & Seifert, R. Cyclic nucleotide-gated ion channels. Physiol. Rev. 82, 769–824 (2002)
Nagel, G. et al. Channelrhodopsin-1: a light-gated proton channel in green algae. Science 296, 2395–2398 (2002)
Nagel, G. et al. Channelrhodopsin-2, a directly light-gated cation-selective membrane channel. Proc. Natl Acad. Sci. USA 100, 13940–13945 (2003)
Doyle, D. A. et al. The structure of the potassium channel: molecular basis of K+ conduction and selectivity. Science 280, 69–77 (1998)
Sato, K. et al. Insect olfactory receptors are heteromeric ligand-gated ion channels. Nature doi: 10.1038/nature06850 (this issue)
Wicher, D., Agricola, H. J., Schönherr, R., Heinemann, S. H. & Derst, C. TRPγ channels are inhibited by cAMP and contribute to pacemaking in neurosecretory insect neurons. J. Biol. Chem. 281, 3227–3236 (2006)
Hille, B. Ion Channels of Excitable Membranes 441–470 (Sinauer Associates, Sunderland, 2001)
Bugaj, V. et al. Functional properties of endogenous receptor- and store-operated calcium influx channels in HEK293 cells. J. Biol. Chem. 280, 16790–16797 (2005)
Messutat, S., Heine, M. & Wicher, D. Calcium-induced calcium release in neurosecretory insect neurons: fast and slow responses. Cell Calcium 30, 199–211 (2001)
Patton, C., Thompson, S. & Epel, D. Some precautions in using chelators to buffer metals in biological solutions. Cell Calcium 35, 427–431 (2004)
Acknowledgements
We thank S. Arend, S. Dietel, S. Kaltofen, A. Roßner and R. Stieber for technical assistance, E. Grosse-Wilde for discussion, E. Neuhaus and H. Hatt for providing the Or22a and Or83b DNA, and M. Biel for providing human HCN2 DNA. This study was supported by the Max Planck Society (D.W., M.C.S. and B.S.H.) and SFB 604 (TP 4, S.H.H.).
Author Contributions D.W. designed the experiments, performed the whole-cell patch-clamp and the calcium-imaging experiments, and analysed the data. R.B. and R.S. made the laser scanning recordings and contributed to the electrophysiological experiments. R.H. performed the cAMP radioreceptor assay. M.C.S. contributed to the bioinformatics and molecular biology effort. D.W. and S.H.H. wrote the paper. B.S.H. and M.C.S. contributed to the manuscript. All authors discussed the concepts and results, and commented on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary information
The file contains Supplementary Figures 1-5 with Legends and Supplementray Tables 1-2. (PDF 1295 kb)
Rights and permissions
About this article
Cite this article
Wicher, D., Schäfer, R., Bauernfeind, R. et al. Drosophila odorant receptors are both ligand-gated and cyclic-nucleotide-activated cation channels. Nature 452, 1007–1011 (2008). https://doi.org/10.1038/nature06861
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature06861
This article is cited by
-
Identification of an adult attractant for Anomala corpulenta by the reverse chemical ecology approach
Journal of Pest Science (2024)
-
High-quality chromosome-level scaffolds of the plant bug Pachypeltis micranthus provide insights into the availability of Mikania micrantha control
BMC Genomics (2023)
-
High-throughput ligand profile characterization in novel cell lines expressing seven heterologous insect olfactory receptors for the detection of volatile plant biomarkers
Scientific Reports (2023)
-
Olfactory navigation in arthropods
Journal of Comparative Physiology A (2023)
-
Reduced olfactory acuity in recently flightless insects suggests rapid regressive evolution
BMC Ecology and Evolution (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.