Abstract
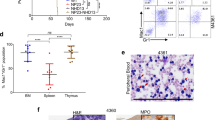
During haematopoiesis, pluripotent haematopoietic stem cells are sequentially restricted to give rise to a variety of lineage-committed progenitors. The classical model of haematopoiesis postulates that, in the first step of differentiation, the stem cell generates common myelo-erythroid progenitors and common lymphoid progenitors (CLPs). However, our previous studies in fetal mice showed that myeloid potential persists even as the lineage branches segregate towards T and B cells1,2,3,4,5,6. We therefore proposed the ‘myeloid-based’ model of haematopoiesis7,8, in which the stem cell initially generates common myelo-erythroid progenitors and common myelo-lymphoid progenitors. T-cell and B-cell progenitors subsequently arise from common myelo-lymphoid progenitors through myeloid-T and myeloid-B stages, respectively. However, it has been unclear whether this myeloid-based model is also valid for adult haematopoiesis. Here we provide clonal evidence that the early cell populations in the adult thymus contain progenitors that have lost the potential to generate B cells but retain substantial macrophage potential as well as T-cell, natural killer (NK)-cell and dendritic-cell potential. We also show that such T-cell progenitors can give rise to macrophages in the thymic environment in vivo. Our findings argue against the classical dichotomy model in which T cells are derived from CLPs; instead, they support the validity of the myeloid-based model for both adult and fetal haematopoiesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kawamoto, H., Ohmura, K. & Katsura, Y. Direct evidence for the commitment of hematopoietic stem cells to T, B and myeloid lineages in murine fetal liver. Int. Immunol. 9, 1011–1019 (1997)
Kawamoto, H., Ohmura, K. & Katsura, Y. Presence of progenitors restricted to T, B, or myeloid lineage, but absence of multipotent stem cells, in the murine fetal thymus. J. Immunol. 161, 3799–3802 (1998)
Ohmura, K. et al. Emergence of T, B and myeloid lineage-committed as well as multipotent hematopoietic progenitors in the aorta–gonad–mesonephros region of day 10 fetuses of the mouse. J. Immunol. 163, 4788–4795 (1999)
Kawamoto, H., Ikawa, T., Ohmura, K., Fujimoto, S. & Katsura, Y. T cell progenitors emerge earlier than B cell progenitors in the murine fetal liver. Immunity 12, 441–450 (2000)
Ikawa, T. et al. Identification of the earliest prethymic T-cell progenitors in murine fetal blood. Blood 103, 530–537 (2004)
Lu, M., Kawamoto, H., Katsube, Y., Ikawa, T. & Katsura, Y. The common myelolymphoid progenitor: a key intermediate stage in hemopoiesis generating T and B cells. J. Immunol. 169, 3519–3525 (2002)
Katsura, Y. Redefinition of lymphoid progenitors. Nature Rev. Immunol. 2, 127–132 (2002)
Kawamoto, H. A close developmental relationship between the lymphoid and myeloid lineages. Trends Immunol. 27, 169–175 (2006)
Kondo, M., Weissman, I. L. & Akashi, K. Identification of clonogenic common lymphoid progenitors in mouse bone marrow. Cell 91, 661–672 (1997)
Kincade, P. W. et al. Nature or nurture? Steady-state lymphocyte formation in adults does not recapitulate ontogeny. Immunol. Rev. 187, 116–125 (2002)
Spangrude, G. J. Divergent models of lymphoid lineage specification: do clonal assays provide all the answers? Immunol. Rev. 187, 40–47 (2002)
Laiosa, C. V., Stadtfeld, M. & Graf, T. Determinants of lymphoid–myeloid lineage diversification. Annu. Rev. Immunol. 24, 705–738 (2006)
Buza-Vidas, N., Luc, S. & Jacobsen, S. E. Delineation of the earliest lineage commitment steps of haematopoietic stem cells: new developments, controversies and major challenges. Curr. Opin. Hematol. 14, 315–321 (2007)
Lee, C. K. et al. Generation of macrophages from early T progenitors in vitro. J. Immunol. 166, 5964–5969 (2001)
Balciunaite, G., Ceredig, R. & Rolink, A. G. The earliest subpopulation of mouse thymocytes contains potent T, significant macrophage, and natural killer cell but no B-lymphocyte potential. Blood 105, 1930–1936 (2005)
Benz, C. & Bleul, C. C. A multipotent precursor in the thymus maps to the branching point of the T versus B lineage decision. J. Exp. Med. 202, 21–31 (2005)
Nishikawa, S., Ogawa, M., Nishikawa, S., Kunisada, T. & Kodama, H. B lymphopoiesis on stromal cell clone: stromal cell clones acting on different stages of B cell differentiation. Eur. J. Immunol. 18, 1767–1771 (1988)
Anderson, M. K., Weiss, A. H., Hernandez-Hoyos, G., Dionne, C. J. & Rothenberg, E. V. Constitutive expression of PU.1 in fetal hematopoietic progenitors blocks T cell development at the pro-T cell stage. Immunity 16, 285–296 (2002)
Masuda, K. et al. Prethymic T-cell development defined by the expression of paired immunoglobulin-like receptors. EMBO J. 24, 4052–4060 (2005)
Ikawa, M., Yamada, S., Nakanishi, T. & Okabe, M. ‘Green mice’ and their potential usage in biological research. FEBS Lett. 430, 83–87 (1998)
Ardavin, C., Wu, L., Li, C. L. & Shortman, K. Thymic dendritic cell and T cells develop simultaneously in the thymus from a common precursor population. Nature 362, 761–763 (1993)
Lu, M. et al. The earliest thymic progenitors in adults are restricted to T, NK, and dendritic cell lineage and have a potential to form more diverse TCRβ chains than fetal progenitors. J. Immunol. 175, 5848–5856 (2005)
Wang, B. et al. A block in both early T lymphocyte and natural killer cell development in transgenic mice with high-copy numbers of the human CD3E gene. Proc. Natl Acad. Sci. USA 91, 9402–9406 (1994)
Hollander, G. A. et al. Developmental control point in induction of thymic cortex regulated by a subpopulation of prothymocytes. Nature 373, 350–353 (1995)
Surh, C. D. & Sprent, J. T-cell apoptosis detected in situ during positive and negative selection in the thymus. Nature 372, 100–103 (1994)
Lind, E. F., Prockop, S. E., Porritt, H. E. & Petrie, H. T. Mapping precursor movement through the postnatal thymus reveals specific microenvironments supporting defined stages of early lymphoid development. J. Exp. Med. 194, 127–134 (2001)
Bell, J. J. & Bhandoola, A. The earliest thymic progenitors for T cells possess myeloid lineage potential. Nature doi: 10.1038/nature06840 (this issue)
Acknowledgements
We thank P. Burrows, W. van Ewijk and W. T. V. Germeraad for critical reading of the manuscript, and O. Kawamoto for valuable advise.
Author Contributions H.W. performed most of experiments, with the assistance of R.S. and K.K. for immunohistochemical analysis and stromal cell co-cultures, respectively. Experiments shown in Fig. 1 and Supplementary Figs 2 and 3 were performed by K.M., and those in Supplementary Fig. 4d by T.I. Y.K. gave critical advice and comments in designing the experiments and writing the paper. H.K. designed the experiments and wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary information
This file contains Supplementary Figures S1-S8 with Legends and additional references. (PDF 2225 kb)
Rights and permissions
About this article
Cite this article
Wada, H., Masuda, K., Satoh, R. et al. Adult T-cell progenitors retain myeloid potential. Nature 452, 768–772 (2008). https://doi.org/10.1038/nature06839
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature06839
This article is cited by
-
Effectiveness of venetoclax and azacytidine against myeloid/natural killer cell precursor acute leukemia
International Journal of Hematology (2024)
-
Lineage switch in a pediatric patient with KMT2A-MLLT3 from acute megakaryoblastic leukemia to T cell acute lymphoblastic leukemia at the fourth relapse after allo-HSCT: with literature review
International Journal of Hematology (2023)
-
Acetylation licenses Th1 cell polarization to constrain Listeria monocytogenes infection
Cell Death & Differentiation (2022)
-
How transcription factors drive choice of the T cell fate
Nature Reviews Immunology (2021)
-
Co-occurrence of immature T-lymphoblastic lymphoma and acute myeloid leukemia—microenvironment-dependent lineage differentiation derived from a common progenitor?
Journal of Hematopathology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.