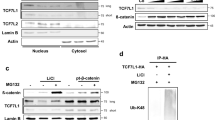
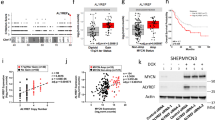
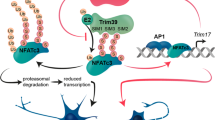
Abstract
The RE1-silencing transcription factor (REST, also known as NRSF) is a master repressor of neuronal gene expression and neuronal programmes in non-neuronal lineages1,2,3. Recently, REST was identified as a human tumour suppressor in epithelial tissues4, suggesting that its regulation may have important physiological and pathological consequences. However, the pathways controlling REST have yet to be elucidated. Here we show that REST is regulated by ubiquitin-mediated proteolysis, and use an RNA interference (RNAi) screen to identify a Skp1-Cul1-F-box protein complex containing the F-box protein β-TRCP (SCFβ-TRCP) as an E3 ubiquitin ligase responsible for REST degradation. β-TRCP binds and ubiquitinates REST and controls its stability through a conserved phospho-degron. During neural differentiation, REST is degraded in a β-TRCP-dependent manner. β-TRCP is required for proper neural differentiation only in the presence of REST, indicating that β-TRCP facilitates this process through degradation of REST. Conversely, failure to degrade REST attenuates differentiation. Furthermore, we find that β-TRCP overexpression, which is common in human epithelial cancers, causes oncogenic transformation of human mammary epithelial cells and that this pathogenic function requires REST degradation. Thus, REST is a key target in β-TRCP-driven transformation and the β-TRCP–REST axis is a new regulatory pathway controlling neurogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Chong, J. A. et al. REST: a mammalian silencer protein that restricts sodium channel gene expression to neurons. Cell 80, 949–957 (1995)
Schoenherr, C. J. & Anderson, D. J. The neuron-restrictive silencer factor (NRSF): a coordinate repressor of multiple neuron-specific genes. Science 267, 1360–1363 (1995)
Ballas, N. & Mandel, G. The many faces of REST oversee epigenetic programming of neuronal genes. Curr. Opin. Neurobiol. 15, 500–506 (2005)
Westbrook, T. F. et al. A genetic screen for candidate tumor suppressors identifies REST. Cell 121, 837–848 (2005)
Ballas, N., Grunseich, C., Lu, D. D., Speh, J. C. & Mandel, G. REST and its corepressors mediate plasticity of neuronal gene chromatin throughout neurogenesis. Cell 121, 645–657 (2005)
Petroski, M. D. & Deshaies, R. J. Function and regulation of cullin-RING ubiquitin ligases. Nature Rev. Mol. Cell Biol. 6, 9–20 (2005)
Bai, C. et al. SKP1 connects cell cycle regulators to the ubiquitin proteolysis machinery through a novel motif, the F-box. Cell 86, 263–274 (1996)
Skowyra, D., Craig, K. L., Tyers, M., Elledge, S. J. & Harper, J. W. F-box proteins are receptors that recruit phosphorylated substrates to the SCF ubiquitin-ligase complex. Cell 91, 209–219 (1997)
Guardavaccaro, D. et al. Control of meiotic and mitotic progression by the F box protein β-Trcp1 in vivo. Dev. Cell 4, 799–812 (2003)
Shirogane, T., Jin, J., Ang, X. L. & Harper, J. W. SCFβ-TRCP controls clock-dependent transcription via casein kinase 1-dependent degradation of the mammalian period-1 (Per1) protein. J. Biol. Chem. 280, 26863–26872 (2005)
Busino, L. et al. Degradation of Cdc25A by β-TrCP during S phase and in response to DNA damage. Nature 426, 87–91 (2003)
Jin, J. et al. SCFβ-TRCP links Chk1 signaling to degradation of the Cdc25A protein phosphatase. Genes Dev. 17, 3062–3074 (2003)
Winston, J. T. et al. The SCFβ-TRCP-ubiquitin ligase complex associates specifically with phosphorylated destruction motifs in IκBα- and β-catenin and stimulates IκBα- ubiquitination in vitro. Genes Dev. 13, 270–283 (1999)
Fuchs, S. Y., Spiegelman, V. S. & Kumar, K. G. The many faces of β-TrCP E3 ubiquitin ligases: reflections in the magic mirror of cancer. Oncogene 23, 2028–2036 (2004)
Wu, G. et al. Structure of a β-TrCP1-Skp1-β-catenin complex: destruction motif binding and lysine specificity of the SCF(β-TrCP1) ubiquitin ligase. Mol. Cell 11, 1445–1456 (2003)
Zhao, J. J. et al. Human mammary epithelial cell transformation through the activation of phosphatidylinositol 3-kinase. Cancer Cell 3, 483–495 (2003)
Kudo, Y. et al. Role of F-box protein β-Trcp1 in mammary gland development and tumorigenesis. Mol. Cell. Biol. 24, 8184–8194 (2004)
Nishikawa, S., Jakt, L. M. & Era, T. Embryonic stem-cell culture as a tool for developmental cell biology. Nature Rev. Mol. Cell Biol. 8, 502–507 (2007)
Ying, Q. L., Stavridis, M., Griffiths, D., Li, M. & Smith, A. Conversion of embryonic stem cells into neuroectodermal precursors in adherent monoculture. Nature Biotechnol. 21, 183–186 (2003)
Aubert, J. et al. Screening for mammalian neural genes via fluorescence-activated cell sorter purification of neural precursors from Sox1-gfp knock-in mice. Proc. Natl Acad. Sci. USA 100 (Suppl 1). 11836–11841 (2003)
Bain, G., Kitchens, D., Yao, M., Huettner, J. E. & Gottlieb, D. I. Embryonic stem cells express neuronal properties in vitro. Dev. Biol. 168, 342–357 (1995)
Fuller, G. N. et al. Many human medulloblastoma tumors overexpress repressor element-1 silencing transcription (REST)/neuron-restrictive silencer factor, which can be functionally countered by REST-VP16. Mol. Cancer Ther. 4, 343–349 (2005)
Su, X. et al. Abnormal expression of REST/NRSF and Myc in neural stem/progenitor cells causes cerebellar tumors by blocking neuronal differentiation. Mol. Cell. Biol. 26, 1666–1678 (2006)
Saitoh, T. & Katoh, M. Expression profiles of β-TRCP1 and β-TRCP2, and mutation analysis of β-TRCP2 in gastric cancer. Int. J. Oncol. 18, 959–964 (2001)
Noubissi, F. K. et al. CRD-BP mediates stabilization of β-TrCP1 and c-myc mRNA in response to β-catenin signalling. Nature 441, 898–901 (2006)
Jin, J. et al. Systematic analysis and nomenclature of mammalian F-box proteins. Genes Dev. 18, 2573–2580 (2004)
Ying, Q. L. & Smith, A. G. Defined conditions for neural commitment and differentiation. Methods Enzymol. 365, 327–341 (2003)
Acknowledgements
We thank G. Mandel and R. Truant for providing REST-C antibodies and mRFP-REST cDNA, respectively. We thank J. Luo, D. Nguyen and D. Lee for suggestions and reading of the manuscript. We are grateful to R. Mulligan, A. Balazs, J. Jin, M. Sheng and V. Dixit for providing reagents, and to D. Guardavaccaro and M. Pagano for communicating results before publication. T.F.W. is a fellow of the Susan G. Komen for the Cure. G.H. is a Helen Hay Whitney post-doctoral fellow. X.L.A. is supported by a National Institutes of Health predoctoral fellowship. This work was supported by National Institutes of Health grants to Y.S., J.W.H., and a US Army Innovator Award to S.J.E. S.J.E. is an Investigator with the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Information
The file contains Supplementary Notes with additional references and Supplementary Figures S1-S11 with Legends. (PDF 1131 kb)
Rights and permissions
About this article
Cite this article
Westbrook, T., Hu, G., Ang, X. et al. SCFβ-TRCP controls oncogenic transformation and neural differentiation through REST degradation. Nature 452, 370–374 (2008). https://doi.org/10.1038/nature06780
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature06780
This article is cited by
-
CRISPR/Cas9-based genome-wide screening of the deubiquitinase subfamily identifies USP3 as a protein stabilizer of REST blocking neuronal differentiation and promotes neuroblastoma tumorigenesis
Journal of Experimental & Clinical Cancer Research (2023)
-
A heterozygous mutation in UBE2H in a patient with developmental delay leads to an aberrant brain development in zebrafish
Human Genomics (2023)
-
SCFβTrCP-mediated degradation of SHARP1 in triple-negative breast cancer
Cell Death & Disease (2023)
-
Connecting telomere maintenance and regulation to the developmental origin and differentiation states of neuroblastoma tumor cells
Journal of Hematology & Oncology (2022)
-
Revealing β-TrCP activity dynamics in live cells with a genetically encoded biosensor
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.