Abstract
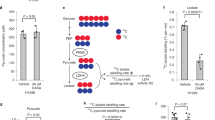
Many tumour cells have elevated rates of glucose uptake but reduced rates of oxidative phosphorylation. This persistence of high lactate production by tumours in the presence of oxygen, known as aerobic glycolysis, was first noted by Otto Warburg more than 75 yr ago1. How tumour cells establish this altered metabolic phenotype and whether it is essential for tumorigenesis is as yet unknown. Here we show that a single switch in a splice isoform of the glycolytic enzyme pyruvate kinase is necessary for the shift in cellular metabolism to aerobic glycolysis and that this promotes tumorigenesis. Tumour cells have been shown to express exclusively the embryonic M2 isoform of pyruvate kinase2. Here we use short hairpin RNA to knockdown pyruvate kinase M2 expression in human cancer cell lines and replace it with pyruvate kinase M1. Switching pyruvate kinase expression to the M1 (adult) isoform leads to reversal of the Warburg effect, as judged by reduced lactate production and increased oxygen consumption, and this correlates with a reduced ability to form tumours in nude mouse xenografts. These results demonstrate that M2 expression is necessary for aerobic glycolysis and that this metabolic phenotype provides a selective growth advantage for tumour cells in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Warburg, O. On the origin of cancer cells. Science 123, 309–314 (1956)
Mazurek, S., Boschek, C. B., Hugo, F. & Eigenbrodt, E. Pyruvate kinase type M2 and its role in tumor growth and spreading. Semin. Cancer Biol. 15, 300–308 (2005)
Fantin, V. R., St-Pierre, J. & Leder, P. Attenuation of LDH-A expression uncovers a link between glycolysis, mitochondrial physiology, and tumor maintenance. Cancer Cell 9, 425–434 (2006)
Majumder, P. K. et al. mTOR inhibition reverses Akt-dependent prostate intraepithelial neoplasia through regulation of apoptotic and HIF-1-dependent pathways. Nature Med. 10, 594–601 (2004)
Altenberg, B. & Greulich, K. O. Genes of glycolysis are ubiquitously overexpressed in 24 cancer classes. Genomics 84, 1014–1020 (2004)
Jurica, M. S. et al. The allosteric regulation of pyruvate kinase by fructose-1,6-bisphosphate. Structure 6, 195–210 (1998)
Dombrauckas, J. D., Santarsiero, B. D. & Mesecar, A. D. Structural basis for tumor pyruvate kinase M2 allosteric regulation and catalysis. Biochemistry 44, 9417–9429 (2005)
Muller, W. J., Sinn, E., Pattengale, P. K., Wallace, R. & Leder, P. Single-step induction of mammary adenocarcinoma in transgenic mice bearing the activated c-neu oncogene. Cell 54, 105–115 (1988)
Gatenby, R. A. & Gillies, R. J. Why do cancers have high aerobic glycolysis? Nature Rev. Cancer 4, 891–899 (2004)
Christofk, H. R., Vander Heiden, M. G., Wu, N., Asara, J. M. & Cantley, L. C. Pyruvate kinase M2 is a phosphotyrosine binding protein. Nature doi: 10.1038/nature06667 (this issue)
Zheng, L., Roeder, R. G. & Luo, Y. S phase activation of the histone H2B promoter by OCA-S, a coactivator complex that contains GAPDH as a key component. Cell 114, 255–266 (2003)
Stetak, A. et al. Nuclear translocation of the tumor marker pyruvate kinase M2 induces programmed cell death. Cancer Res. 67, 1602–1608 (2007)
DeBerardinis, R. J. et al. Beyond aerobic glycolysis: transformed cells can engage in glutamine metabolism that exceeds the requirement for protein and nucleotide synthesis. Proc. Natl Acad. Sci. USA 104, 19345–19350 (2007)
Manes, N. P. & El-Maghrabi, M. R. The kinase activity of human brain 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase is regulated via inhibition by phosphoenolpyruvate. Arch. Biochem. Biophys. 438, 125–136 (2005)
Telang, S. et al. Ras transformation requires metabolic control by 6-phosphofructo-2-kinase. Oncogene 25, 7225–7234 (2006)
Vander Heiden, M. G. et al. Growth factors can influence cell growth and survival through effects on glucose metabolism. Mol. Cell. Biol. 21, 5899–5912 (2001)
Budinger, G. R. et al. Cellular energy utilization and supply during hypoxia in embryonic cardiac myocytes. Am. J. Physiol. 270, L44–L53 (1996)
Sabatine, M. S. et al. Metabolomic identification of novel biomarkers of myocardial ischemia. Circulation 112, 3868–3875 (2005)
Engelman, J. A. et al. Allelic dilution obscures detection of a biologically significant resistance mutation in EGFR-amplified lung cancer. J. Clin. Invest. 116, 2695–2706 (2006)
Root, D. E., Hacohen, N., Hahn, W. C., Lander, E. S. & Sabatini, D. M. Genome-scale loss-of-function screening with a lentiviral RNAi library. Nature Methods 3, 715–719 (2006)
Acknowledgements
We thank M. Bentires-Alj for the MMTV-NeuNT tissue lysates and W. Hahn for the lentiviral shRNA constructs. We thank S. Soltoff for use of the anaerobic chamber and oxygen electrode, and Q. Song for use of the hypoxia chamber. We thank I. Rhee and T. Yuan for help with the nude mouse injections. We thank M. Liu for technical assistance. R.E.G. is supported by funding from the National Institutes of Health, the Donald W. Reynolds Foundation, the Fondation Leducq, and the Broad Institute Scientific Planning and Allocation of Resources Committee. M.G.V.H. is a Damon Runyon Fellow supported by the Damon Runyon Cancer Research Foundation. This research was supported by funding to L.C.C. from the National Institutes of Health. S.L.S. is an Investigator with the Howard Hughes Medical Institute.
Author Contributions M.H.H. and M.D.F. contributed the immunohistochemistry data (Fig. 1c). A.R., R.E.G., R.W. and S.L.S. contributed the metabolite measurement data (Fig. 3d). H.R.C. and M.G.V.H. performed all other experiments. H.R.C., M.G.V.H. and L.C.C. designed the study and wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
L.C.C. is a founder of a company (Cell Metabolism Therapeutics) that proposes to target enzymes in metabolism for cancer therapy. The results of this paper support this concept.
Supplementary information
Supplementary Figures
The file contains Supplementary Figures 1-2 with Legends. The Supplementary Figure 1 shows the peptide sequences used to generate PKM1- and PKM2-specific polyclonal antibodies as well as an immunoblot confirming antibody specificity. The Supplementary Figure 2 shows data from control experiments indicating that changes in PK activity in M1- and M2-expressing cells do not result in changes in adenine nucleotide levels. (PDF 193 kb)
Rights and permissions
About this article
Cite this article
Christofk, H., Vander Heiden, M., Harris, M. et al. The M2 splice isoform of pyruvate kinase is important for cancer metabolism and tumour growth. Nature 452, 230–233 (2008). https://doi.org/10.1038/nature06734
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature06734
This article is cited by
-
Ribosome profiling: a powerful tool in oncological research
Biomarker Research (2024)
-
PKM2 promotes lymphatic metastasis of hypopharyngeal carcinoma via regulating epithelial-mesenchymal transition: an experimental research
Diagnostic Pathology (2024)
-
Autophagy-related biomarkers in preeclampsia: the underlying mechanism, correlation to the immune microenvironment and drug screening
BMC Pregnancy and Childbirth (2024)
-
LINC01852 inhibits the tumorigenesis and chemoresistance in colorectal cancer by suppressing SRSF5-mediated alternative splicing of PKM
Molecular Cancer (2024)
-
Metabolic regulation of tumor-associated macrophage heterogeneity: insights into the tumor microenvironment and immunotherapeutic opportunities
Biomarker Research (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.