Abstract
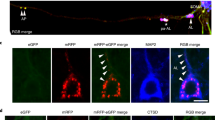
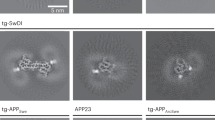
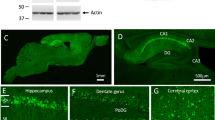
Senile plaques accumulate over the course of decades in the brains of patients with Alzheimer’s disease. A fundamental tenet of the amyloid hypothesis of Alzheimer’s disease is that the deposition of amyloid-β precedes and induces the neuronal abnormalities that underlie dementia1. This idea has been challenged, however, by the suggestion that alterations in axonal trafficking and morphological abnormalities precede and lead to senile plaques2. The role of microglia in accelerating or retarding these processes has been uncertain. To investigate the temporal relation between plaque formation and the changes in local neuritic architecture, we used longitudinal in vivo multiphoton microscopy to sequentially image young APPswe/PS1d9xYFP (B6C3-YFP) transgenic mice3. Here we show that plaques form extraordinarily quickly, over 24 h. Within 1–2 days of a new plaque’s appearance, microglia are activated and recruited to the site. Progressive neuritic changes ensue, leading to increasingly dysmorphic neurites over the next days to weeks. These data establish plaques as a critical mediator of neuritic pathology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Hardy, J. & Selkoe, D. J. The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science 297, 353–356 (2002)
Stokin, G. B. et al. Axonopathy and transport deficits early in the pathogenesis of Alzheimer’s disease. Science 307, 1282–1288 (2005)
Jankowsky, J. L. et al. Co-expression of multiple transgenes in mouse CNS: a comparison of strategies. Biomol. Eng. 17, 157–165 (2001)
Jankowsky, J. L. et al. Mutant presenilins specifically elevate the levels of the 42 residue beta-amyloid peptide in vivo: evidence for augmentation of a 42-specific gamma secretase. Hum. Mol. Genet. 13, 159–170 (2004)
Kawai, M., Kalaria, R. N., Harik, S. I. & Perry, G. The relationship of amyloid plaques to cerebral capillaries in Alzheimer’s disease. Am. J. Pathol. 137, 1435–1446 (1990)
Kumar-Singh, S. et al. Dense-core plaques in Tg2576 and PSAPP mouse models of Alzheimer’s disease are centered on vessel walls. Am. J. Pathol. 167, 527–543 (2005)
Hsiao, K. et al. Correlative memory deficits, Aβ elevation, and amyloid plaques in transgenic mice. Science 274, 99–102 (1996)
Hyman, B. T. et al. Quantitative analysis of senile plaques in Alzheimer disease: observation of log-normal size distribution and molecular epidemiology of differences associated with apolipoprotein E genotype and trisomy 21 (Down syndrome). Proc. Natl Acad. Sci. USA 92, 3586–3590 (1995)
Itagaki, S., McGeer, P. L., Akiyama, H., Zhu, S. & Selkoe, D. Relationship of microglia and astrocytes to amyloid deposits of Alzheimer disease. J. Neuroimmunol. 24, 173–182 (1989)
Frautschy, S. A. et al. Microglial response to amyloid plaques in APPsw transgenic mice. Am. J. Pathol. 152, 307–317 (1998)
Combs, C. K., Karlo, J. C., Kao, S. C. & Landreth, G. E. β-Amyloid stimulation of microglia and monocytes results in TNFα-dependent expression of inducible nitric oxide synthase and neuronal apoptosis. J. Neurosci. 21, 1179–1188 (2001)
Qin, S. et al. System Xc- and apolipoprotein E expressed by microglia have opposite effects on the neurotoxicity of amyloid-β peptide 1–40. J. Neurosci. 26, 3345–3356 (2006)
Schenk, D. et al. Immunization with amyloid-β attenuates Alzheimer-disease-like pathology in the PDAPP mouse. Nature 400, 173–177 (1999)
Nagele, R. G., Wegiel, J., Venkataraman, V., Imaki, H. & Wang, K. C. Contribution of glial cells to the development of amyloid plaques in Alzheimer’s disease. Neurobiol. Aging 25, 663–674 (2004)
Simard, A. R., Soulet, D., Gowing, G., Julien, J. P. & Rivest, S. Bone marrow-derived microglia play a critical role in restricting senile plaque formation in Alzheimer’s disease. Neuron 49, 489–502 (2006)
Games, D. et al. Alzheimer-type neuropathology in transgenic mice overexpressing V717F β-amyloid precursor protein. Nature 373, 523–527 (1995)
Jung, S. et al. Analysis of fractalkine receptor CX(3)CR1 function by targeted deletion and green fluorescent protein reporter gene insertion. Mol. Cell. Biol. 20, 4106–4114 (2000)
Bacskai, B. J. et al. Imaging of amyloid-β deposits in brains of living mice permits direct observation of clearance of plaques with immunotherapy. Nature Med. 7, 369–372 (2001)
Geula, C. et al. Aging renders the brain vulnerable to amyloid β-protein neurotoxicity. Nature Med. 4, 827–831 (1998)
Knowles, R. B. et al. Plaque-induced neurite abnormalities: implications for disruption of neural networks in Alzheimer’s disease. Proc. Natl Acad. Sci. USA 96, 5274–5279 (1999)
Le, R. et al. Plaque-induced abnormalities in neurite geometry in transgenic models of Alzheimer disease: implications for neural system disruption. J. Neuropathol. Exp. Neurol. 60, 753–758 (2001)
Jarrett, J. T. & Lansbury, P. T. Seeding ‘one-dimensional crystallization’ of amyloid: a pathogenic mechanism in Alzheimer’s disease and scrapie? Cell 73, 1055–1058 (1993)
Meyer-Luehmann, M. et al. Exogenous induction of cerebral β-amyloidogenesis is governed by agent and host. Science 313, 1781–1784 (2006)
Walsh, D. M. et al. Naturally secreted oligomers of amyloid-β protein potently inhibit hippocampal long-term potentiation in vivo . Nature 416, 535–539 (2002)
Lesne, S. et al. A specific amyloid-β protein assembly in the brain impairs memory. Nature 440, 352–357 (2006)
Spires, T. L. et al. Dendritic spine abnormalities in amyloid precursor protein transgenic mice demonstrated by gene transfer and intravital multiphoton microscopy. J. Neurosci. 25, 7278–7287 (2005)
Klunk, W. E. et al. Imaging Aβ plaques in living transgenic mice with multiphoton microscopy and methoxy-X04, a systemically administered Congo red derivative. J. Neuropathol. Exp. Neurol. 61, 797–805 (2002)
Acknowledgements
This work was supported by a grant from the National Institutes of Health and an award from the Alzheimer’s Association. We also thank S. Freeman (Harvard Medical School, Boston) for technical advice and W. E. Klunk (University of Pittsburgh) for the gift of methoxy-XO4.
Author Contributions M.M.-L. and B.T.H. designed the study; M.M.-L., T.L.S.-J., C.P., M.G.-A., A.de C. and A.R. performed experiments; J.K.-T. and D.M.H. provided mice; M.M.-L. and B.T.H. wrote the manuscript; B.J.B. gave technical support and conceptual advice; B.T.H. supervised the study.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Figures
The file contains Supplementary Figures 1-7 with Legends. (PDF 639 kb)
Rights and permissions
About this article
Cite this article
Meyer-Luehmann, M., Spires-Jones, T., Prada, C. et al. Rapid appearance and local toxicity of amyloid-β plaques in a mouse model of Alzheimer’s disease. Nature 451, 720–724 (2008). https://doi.org/10.1038/nature06616
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature06616
This article is cited by
-
Neuritic Plaques — Gateways to Understanding Alzheimer’s Disease
Molecular Neurobiology (2024)
-
Amyloid fibril proteomics of AD brains reveals modifiers of aggregation and toxicity
Molecular Neurodegeneration (2023)
-
Effect of disturbances in neuronal calcium and IP3 dynamics on β-amyloid production and degradation
Cognitive Neurodynamics (2023)
-
The role of A\(\beta \) and Tau proteins in Alzheimer’s disease: a mathematical model on graphs
Journal of Mathematical Biology (2023)
-
Role of NLRP3 Inflammasome and Its Inhibitors as Emerging Therapeutic Drug Candidate for Alzheimer’s Disease: a Review of Mechanism of Activation, Regulation, and Inhibition
Inflammation (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.