Abstract
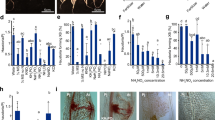
Calcium controls a number of critical events, including motility, secretion, cell invasion and egress by apicomplexan parasites1. Compared to animal2 and plant cells3, the molecular mechanisms that govern calcium signalling in parasites are poorly understood. Here we show that the production of the phytohormone abscisic acid (ABA) controls calcium signalling within the apicomplexan parasite Toxoplasma gondii, an opportunistic human pathogen. In plants, ABA controls a number of important events, including environmental stress responses, embryo development and seed dormancy4,5. ABA induces production of the second-messenger cyclic ADP ribose (cADPR), which controls release of intracellular calcium stores in plants6. cADPR also controls intracellular calcium release in the protozoan parasite T. gondii7,8; however, previous studies have not revealed the molecular basis of this pathway9. We found that addition of exogenous ABA induced formation of cADPR in T. gondii, stimulated calcium-dependent protein secretion, and induced parasite egress from the infected host cell in a density-dependent manner. Production of endogenous ABA within the parasite was confirmed by purification (using high-performance liquid chromatography) and analysis (by gas chromatography-mass spectrometry). Selective disruption of ABA synthesis by the inhibitor fluridone delayed egress and induced development of the slow-growing, dormant cyst stage of the parasite. Thus, ABA-mediated calcium signalling controls the decision between lytic and chronic stage growth, a developmental switch that is central in pathogenesis and transmission. The pathway for ABA production was probably acquired with an algal endosymbiont that was retained as a non-photosynthetic plastid known as the apicoplast. The plant-like nature of this pathway may be exploited therapeutically, as shown by the ability of a specific inhibitor of ABA synthesis to prevent toxoplasmosis in the mouse model.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Moreno, S. N. J. & Docampo, R. Calcium regulation in protozoan parasites. Curr. Opin. Microbiol. 6, 359–364 (2003)
Berridge, M. J., Bootman, M. D. & Roderick, H. L. Calcium signaling: Dynamics, homeostasis and remodelling. Nature Rev. Mol. Cell Biol. 4, 517–529 (2003)
Bothwell, J. H. F. & Ng, C. K. Y. The evolution of Ca2+ signaling in photosynthetic eukaryotes. New Phytol. 166, 21–38 (2005)
Xiong, L. & Zhu, J. K. Regulation of abscisic acid biosynthesis. Plant Physiol. 133, 29–36 (2003)
Schwartz, S. H., Qin, X. & Zeevaart, J. A. D. Elucidation of the indirect pathway of abscisic acid biosynthesis by mutants, genes, and enzymes. Plant Physiol. 131, 1591–1601 (2003)
Wu, Y. et al. Abscisic acid signaling through cyclic ADP ribose in plants. Science 278, 2126–2130 (1997)
Lovett, J. L., Marchesini, N., Moreno, S. N. & Sibley, L. D. Toxoplasma gondii microneme secretion involves intracellular Ca2+ release from IP3 / ryanodine sensitive stores. J. Biol. Chem. 277, 25870–25876 (2002)
Chini, E. N., Nagamune, K., Wetzel, D. M. & Sibley, L. D. Evidence that the cADPR signaling pathway controls calcium-mediated secretion in Toxoplasma gondii . Biochem. J. 389, 269–277 (2005)
Nagamune, K. & Sibley, L. D. Comparative genomic and phylogenetic analyses of calcium ATPases and calcium-regulated proteins in the Apicomplexa. Mol. Biol. Evol. 23, 1613–1627 (2006)
Puce, S. et al. Abscisic acid signaling through cyclic ADP-ribose in hydroid regeneration. J. Biochem. 279, 39783–39788 (2004)
Zocchi, E. et al. The temperature-signaling cascade in sponges involves a heat-gated cation channel, abscisic acid, and cyclic ADP ribose. Proc. Natl Acad. Sci. USA 98, 14859–14864 (2001)
de Almeida, J. A. S., Kascheres, C. & Pereira, M. D. A. Ethylene and abscisic acid in the control of development of the rhizome of Koleria eriantha (Benth.) Hanst. (Gesneriaceae). Braz. J. Plant Physiol. 17, 391–399 (2005)
Moudy, R., Manning, T. J. & Beckers, C. J. The loss of cytoplasmic potassium upon host cell breakdown triggers egress of Toxoplasma gondii . J. Biol. Chem. 276, 41492–41501 (2001)
Endo, T., Sethi, K. K. & Piekarski, G. Toxoplasma gondii: Calcium ionophore A23187-mediated exit of trophozoites from infected murine macrophages. Exp. Parasitol. 53, 179–188 (1982)
Zhang, Y. W., Halonen, S. K., Ma, Y. F., Wittner, M. & Weiss, L. M. Initial characterization of CST1, a Toxoplasma gondii cyst wall glycoprotein. Infect. Immun. 69, 501–507 (2001)
Zhang, Y. W. et al. Disruption of the Toxoplasma gondii bradyzoite-specific gene BAG1 decreases in vivo cyst formation. Mol. Microbiol. 31, 691–701 (1999)
Fux, B. et al. Toxoplasma gondii strains defective in oral transmission are also defective in developmental stage differentiation. Infect. Immun. 75, 2580–2590 (2007)
Leef, J. L. & Carlson, P. S. Carotenoid synthesis inhibiting herbicides and fatty acid synthesis oxime herbicides as anti-apicomplexa protozoan parasite agents. Patent 847932, 1–8 (Potomax Ltd. Prtn). (1999)
Jomaa, H. et al. Inhibitors of the non-mevalonate pathway of isoprenoid biosynthesis as antimalarial drugs. Science 285, 1573–1576 (1999)
Ralph, S. A. et al. Metabolic maps and functions of the Plasmodium falciparum apicoplast. Nature Rev. Microbiol. 2, 203–216 (2004)
Waller, R. F. & McFadden, G. I. The apicoplast: A review of the derived plastid of apicomplexan parasites. Curr. Iss. Mol. Biol. 7, 57–79 (2005)
Hirsch, R., Hartung, W. & Gimmler, H. Abscisic acid content of algae under stress. Bot. Acta 102, 326–334 (1989)
Cowan, A. K. & Rose, P. D. Abscisic acid metabolism in salt stressed cells of Dunaliella salina . Plant Physiol. 97, 798–803 (1991)
Kobayashi, M., Hirai, N., Kurimura, Y., Ohigashi, H. & Tsuji, Y. Abscisic acid-dependent algal morphogenesis in the unicellular green alga Haematococcus pluvialis . Plant Growth Reg. 22, 79–85 (1997)
Razem, F. A., El-Kereamy, A., Abrams, S. R. & Hill, R. D. The RNA-binding protein FCA is an abscisic acid receptor. Nature 439, 290–294 (2006)
Liu, X. et al. A G protein-coupled receptor is a plasma membrane receptor for the plant hormone abscisic acid. Science 315, 1712–1716 (2007)
Carruthers, V. B., Sherman, G. D. & Sibley, L. D. The Toxoplasma adhesive protein MIC2 is proteolytically processed at multiple sites by two parasite-derived proteases. J. Biol. Chem. 275, 14346–14353 (2000)
Tian, L., DellaPenna, D. & Zeevaart, J. A. D. Effect of hydroxylated carotenoid deficiency on ABA accumulation in Arabidopsis . Physiol. Plant. 122, 314–320 (2004)
Rodrigues, C. O., Ruiz, F. A., Rohloff, P., Scott, D. A. & Moreno, S. N. J. Characterization of isolated acidocalcisomes from Toxoplasma gondii tachyzoites reveals a novel pool of hydrolyzable polyphosphate. J. Biol. Chem. 277, 48650–48656 (2002)
Saeij, J. P., Boyle, J. P., Grigg, M. E., Arrizabalaga, G. & Boothroyd, J. C. Bioluminescence imaging of Toxoplasma gondii infection in living mice reveals dramatic differences between strains. Infect. Immun. 73, 695–702 (2005)
Acknowledgements
We thank J. Zeevaart for conducting the initial purification and analysis of ABA and for supplying standards; J. Boothroyd and L. Weiss for providing reagents; W. Beatty, S. Moreno, B. Striepen, A. Waters and L. Xiong for comments; and J. Nawas and D. Gill for technical assistance. This work was supported by the Uehara Medical Foundation (K.N.), the Mayo Clinic and American Heart Association (E.N.C.) and the NIH (L.D.S.).
Author Contributions K.N. performed the experiments on the effects of ABA and fluridone on the parasite, L.M.H. performed the MS studies, B.F. performed the animal studies, F.B. contributed to the analysis of ABA genes, E.N.C. performed the measurements of cADPR, L.D.S. supervised the project and wrote the manuscript with input from all the authors.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
The file contains Supplementary Figures 1-3 with Legends and additional references. (PDF 3529 kb)
Rights and permissions
About this article
Cite this article
Nagamune, K., Hicks, L., Fux, B. et al. Abscisic acid controls calcium-dependent egress and development in Toxoplasma gondii. Nature 451, 207–210 (2008). https://doi.org/10.1038/nature06478
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature06478
This article is cited by
-
Phytohormones regulate asexual Toxoplasma gondii replication
Parasitology Research (2023)
-
Contemporary exploitation of natural products for arthropod-borne pathogen transmission-blocking interventions
Parasites & Vectors (2022)
-
Integration of a multi-step heterologous pathway in Saccharomyces cerevisiae for the production of abscisic acid
Microbial Cell Factories (2019)
-
Phosphatidic acid governs natural egress in Toxoplasma gondii via a guanylate cyclase receptor platform
Nature Microbiology (2019)
-
The plant hormone abscisic acid regulates the growth and metabolism of endophytic fungus Aspergillus nidulans
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.