Abstract
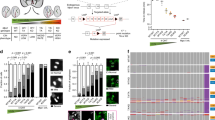
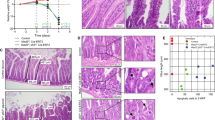
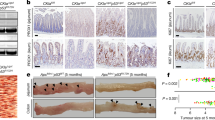
Epidemiological studies spanning more than 50 yr reach conflicting conclusions as to whether there is a lower incidence of solid tumours in people with trisomy 21 (Down’s syndrome)1,2. We used mouse models of Down’s syndrome and of cancer in a biological approach to investigate the relationship between trisomy and the incidence of intestinal tumours. ApcMin -mediated tumour number was determined in aneuploid mouse models Ts65Dn, Ts1Rhr and Ms1Rhr. Trisomy for orthologues of about half of the genes on chromosome 21 (Hsa21) in Ts65Dn mice or just 33 of these genes in Ts1Rhr mice resulted in a significant reduction in the number of intestinal tumours. In Ms1Rhr, segmental monosomy for the same 33 genes that are triplicated in Ts1Rhr resulted in an increased number of tumours. Further studies demonstrated that the Ets2 gene contributed most of the dosage-sensitive effect on intestinal tumour number. The action of Ets2 as a repressor when it is overexpressed differs from tumour suppression, which requires normal gene function to prevent cellular transformation. Upregulation of Ets2 and, potentially, other genes involved in this kind of protective effect may provide a prophylactic effect in all individuals, regardless of ploidy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Satgé, D. et al. A tumor profile in Down syndrome. Am. J. Med. Genet. 78, 207–216 (1998)
Yang, Q., Rasmussen, S. A. & Friedman, J. M. Mortality associated with Down's syndrome in the USA from 1983 to 1997: a population-based study. Lancet 359, 1019–1025 (2002)
Reeves, R. H. et al. A mouse model for Down Syndrome exhibits learning and behaviour deficits. Nature Genet. 11, 177–183 (1995)
Gardiner, K., Fortna, A., Bechtel, L. & Davisson, M. T. Mouse models of Down syndrome: how useful can they be? Comparison of the gene content of human chromosome 21 with orthologous mouse genomic regions. Gene 318, 137–147 (2003)
Su, L. K. et al. Multiple intestinal neoplasia caused by a mutation in the murine homolog of the APC gene. Science 256, 668–670 (1992)
Yamada, Y. & Mori, H. Multistep carcinogenesis of the colon in ApcMin/+) mouse. Cancer Sci. 98, 6–10 (2007)
Dietrich, W. F. et al. Genetic identification of Mom-1, a major modifier locus affecting Min-induced intestinal neoplasia in the mouse. Cell 75, 631–639 (1993)
MacPhee, M. et al. The secretory phospholipase A2 gene is a candidate for the Mom1 locus, a major modifier of ApcMin -induced intestinal neoplasia. Cell 81, 957–966 (1995)
Olson, L. E., Richtsmeier, J. T., Leszl, J. & Reeves, R. H. A chromosome 21 critical region does not cause specific Down syndrome phenotypes. Science 306, 687–690 (2004)
Hsu, T., Trojanowska, M. & Watson, D. K. Ets proteins in biological control and cancer. J. Cell. Biochem. 91, 896–903 (2004)
Wolvetang, E. J. et al. Overexpression of the chromosome 21 transcription factor Ets2 induces neuronal apoptosis. Neurobiol. Dis. 14, 349–356 (2003)
Roberts, R. B. et al. Importance of epidermal growth factor receptor signaling in establishment of adenomas and maintenance of carcinomas during intestinal tumorigenesis. Proc. Natl Acad. Sci. USA 99, 1521–1526 (2002)
Day, S. M., Strauss, D. J., Shavelle, R. M. & Reynolds, R. J. Mortality and causes of death in persons with Down syndrome in California. Dev. Med. Child Neurol. 47, 171–176 (2005)
Hill, D. A. et al. Mortality and cancer incidence among individuals with Down syndrome. Arch. Intern. Med. 163, 705–711 (2003)
Patja, K., Pukkala, E., Sund, R., Iivanainen, M. & Kaski, M. Cancer incidence of persons with Down syndrome in Finland: a population-based study. Int. J. Cancer 118, 1769–1772 (2006)
Goldacre, M. J., Wotton, C. J., Seagroatt, V. & Yeates, D. Cancers and immune related diseases associated with Down's syndrome: a record linkage study. Arch. Dis. Child. 89, 1014–1017 (2004)
O'Reilly, M. S. et al. Endostatin: an endogenous inhibitor of angiogenesis and tumor growth. Cell 88, 277–285 (1997)
Minami, T. et al. Vascular endothelial growth factor- and thrombin-induced termination factor, Down syndrome critical region-1, attenuates endothelial cell proliferation and angiogenesis. J. Biol. Chem. 279, 50537–50554 (2004)
Moore, C. S. et al. Integration of cytogenetic with recombinational and physical maps of mouse chromosome 16. Genomics 59, 1–5 (1999)
The Jackson Laboratory. Genotyping protocol for Apc. 〈http://jaxmice.jax.org/pub-cgi/protocols/protocols.sh?objtype=protocol &protocol_id=529#133〉
Smith, J. L. et al. ets-2 is a target for an akt (Protein kinase B)/jun N-terminal kinase signaling pathway in macrophages of motheaten-viable mutant mice. Mol. Cell. Biol. 20, 8026–8034 (2000)
Acknowledgements
The authors thank L. Siracusa for advice regarding the ApcMin system, C. Dang for important discussions regarding cancer models, and R. Roper for statistical advice. This work was supported by National Institute of Child Health and Development and National Cancer Institute awards (M.C.O. and R.H.R.).
Author Contributions T.E.S. and R.H.R. designed the experiments. T.E.S. and A.Y. managed husbandry and collected tumour data, which were analysed by T.E.S., A.Y. and R.H.R.; F.L. and M.C.O. designed the Ets2 conditional knockout mice; and A.Y., F.L. and M.C.O. analysed Ets2 expression. R.H.R. wrote the paper with substantial input from all authors.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
The file contains Supplementary Tables 1-2 and Supplementary Figures 1-4 with Legends. (PDF 319 kb)
Rights and permissions
About this article
Cite this article
Sussan, T., Yang, A., Li, F. et al. Trisomy represses ApcMin -mediated tumours in mouse models of Down’s syndrome. Nature 451, 73–75 (2008). https://doi.org/10.1038/nature06446
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature06446
This article is cited by
-
A reassessment of Jackson’s checklist and identification of two Down syndrome sub-phenotypes
Scientific Reports (2022)
-
Aneuploidy as a promoter and suppressor of malignant growth
Nature Reviews Cancer (2021)
-
Transient PLK4 overexpression accelerates tumorigenesis in p53-deficient epidermis
Nature Cell Biology (2016)
-
SHH desmoplastic/nodular medulloblastoma and Gorlin syndrome in the setting of Down syndrome: case report, molecular profiling, and review of the literature
Child's Nervous System (2016)
-
Aneuploidy and chromosomal instability in cancer: a jackpot to chaos
Cell Division (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.