Abstract
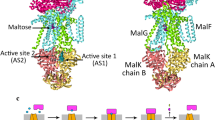
The maltose uptake system of Escherichia coli is a well-characterized member of the ATP-binding cassette transporter superfamily. Here we present the 2.8-Å crystal structure of the intact maltose transporter in complex with the maltose-binding protein, maltose and ATP. This structure, stabilized by a mutation that prevents ATP hydrolysis, captures the ATP-binding cassette dimer in a closed, ATP-bound conformation. Maltose is occluded within a solvent-filled cavity at the interface of the two transmembrane subunits, about halfway into the lipid bilayer. The binding protein docks onto the entrance of the cavity in an open conformation and serves as a cap to ensure unidirectional translocation of the sugar molecule. These results provide direct evidence for a concerted mechanism of transport in which solute is transferred from the binding protein to the transmembrane subunits when the cassette dimer closes to hydrolyse ATP.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Higgins, C. F. ABC transporters: from microorganisms to man. Annu. Rev. Cell Biol. 8, 67–113 (1992)
Boos, W. & Lucht, J. M. in Escherichia coli and Salmonella: Cellular and Molecular Biology (eds Neidhardt, F. C. et al.) 1175–1209 (ASM Press, Washington DC, 1996)
Holland, I. B. & Blight, M. A. ABC-ATPases, adaptable energy generators fuelling transmembrane movement of a variety of molecules in organisms from bacteria to humans. J. Mol. Biol. 293, 381–399 (1999)
Walker, J. E., Saraste, M., Runswick, M. J. & Gay, N. J. Distantly related sequences in the α- and β-subunits of ATP synthase, myosin, kinases and other ATP-requiring enzymes and a common nucleotide binding fold. EMBO J. 1, 945–951 (1982)
Pinkett, H. W., Lee, A. T., Lum, P., Locher, K. P. & Rees, D. C. An inward-facing conformation of a putative metal-chelate-type ABC transporter. Science 315, 373–377 (2007)
Locher, K. P., Lee, A. T. & Rees, D. C. The E. coli BtuCD structure: a framework for ABC transporter architecture and mechanism. Science 296, 1091–1098 (2002)
Hollenstein, K., Frei, D. C. & Locher, K. P. Structure of an ABC transporter in complex with its binding protein. Nature 446, 213–216 (2007)
Dawson, R. J. & Locher, K. P. Structure of a bacterial multidrug ABC transporter. Nature 443, 180–185 (2006)
Ferenci, T. The recognition of maltodextrins by Escherichia coli. . Eur. J. Biochem. 108, 613–636 (1980)
Raibaud, O., Roa, M., Braun-Breton, C. & Schwartz, M. Structure of the malB region in Escherichia coli K-12. I. Genetic map of the malK-lamB operon. Mol. Gen. Genet. 174, 241–248 (1979)
Silhavy, T. J. et al. Structure of the malB region in Escherichia coli K12. II. Genetic map of the malE,F,G operon. Mol. Gen. Genet. 174, 249–259 (1979)
Davidson, A. L. & Nikaido, H. Purification and characterization of the membrane-associated components of the maltose transport system from Escherichia coli . J. Biol. Chem. 266, 8946–8951 (1991)
Davidson, A. L. & Nikaido, H. Overproduction, solubilization, and reconstitution of the maltose transport system from Escherichia coli . J. Biol. Chem. 265, 4254–4260 (1990)
Landmesser, H. et al. Large-scale purification, dissociation and functional reassembly of the maltose ATP-binding cassette transporter (MalFGK(2)) of Salmonella typhimurium . Biochim. Biophys. Acta 1565, 64–72 (2002)
Davidson, A. L., Shuman, H. A. & Nikaido, H. Mechanism of maltose transport in Escherichia coli: Transmembrane signalling by periplasmic binding proteins. Proc. Natl Acad. Sci. USA 89, 2360–2364 (1992)
Sharff, A. J., Rodseth, L. E., Spurlino, J. E. & Quiocho, F. A. Crystallographic evidence of a large ligand-induced hinge-twist motion between the two domains of the maltodextrin binding protein involved in active transport and chemotaxis. Biochemistry 31, 10657–10663 (1992)
Quiocho, F. A., Spurlino, J. C. & Rodseth, L. E. Extensive features of tight oligosaccharide binding revealed in high-resolution structures of the maltodextrin transport/chemosensory receptor. Structure 5, 997–1015 (1997)
Duan, X. & Quiocho, F. A. Structural evidence for the dominant role of nonpolar interactions in the binding of a transport/chemonsensory receptor to its highly polar ligands. Biochemistry 41, 706–712 (2002)
Chen, J., Sharma, S., Quiocho, F. A. & Davidson, A. L. Trapping the transition state of an ATP-binding-cassette transporter: Evidence for a concerted mechanism of maltose transport. Proc. Natl Acad. Sci. USA 98, 1525–1530 (2001)
Chen, J., Lu, G., Lin, J., Davidson, A. L. & Quiocho, F. A. A tweezers-like motion of the ATP-binding cassette dimer in an ABC transport cycle. Mol. Cell 12, 651–661 (2003)
Lu, G., Westbrooks, J. M., Davidson, A. L. & Chen, J. ATP hydrolysis is required to reset the ATP-binding cassette dimer into the resting-state conformation. Proc. Natl Acad. Sci. USA 102, 17969–17974 (2005)
Orelle, C., Dalmas, O., Gros, P., Di Pietro, A. & Jault, J. M. The conserved glutamate residue adjacent to the Walker-B motif is the catalytic base for ATP hydrolysis in the ATP-binding cassette transporter BmrA. J. Biol. Chem. 278, 47002–47008 (2003)
Moody, J. E., Millen, L., Binns, D., Hunt, J. F. & Thomas, P. J. Cooperative, ATP-dependent association of the nucleotide binding cassettes during the catalytic cycle of ATP-binding cassette transporters. J. Biol. Chem. 277, 21111–21114 (2002)
Smith, P. C. et al. ATP binding to the motor domain from an ABC transporter drives formation of a nucleotide sandwich dimer. Mol. Cell 10, 139–149 (2002)
Rossmann, M. G. The molecular replacement method. Acta Crystallogr. A 46, 73–82 (1990)
Covitz, K.-M. Y., Panagiotidis, C. H., Reyes, M., Treptow, N. A. & Shuman, H. A. Mutations that alter the transmembrane signalling pathway in an ATP binding cassette (ABC) transporter. EMBO J. 13, 1752–1759 (1994)
Dassa, E. & Muir, S. Membrane topology of MalG, an inner membrane protein from the maltose transport system of Escherichia coli . Mol. Microbiol. 7, 29–38 (1993)
Froshauer, S., Green, G. N., Boyd, D., McGovern, K. & Beckwith, J. Genetic analysis of the membrane insertion and topology of MalF, a cytoplasmic membrane protein of Escherichia coli . J. Mol. Biol. 200, 501–511 (1988)
Hvorup, R. N. et al. Asymmetry in the structure of the ABC transporter binding protein complex BtuCD-BtuF. Science 317, 1387–1390 (2007)
Dassa, E. & Hofnung, M. Sequence of gene malG in E. coli K12: homologies between integral membrane components from binding protein-dependent transport systems. EMBO J. 4, 2287–2293 (1985)
Saurin, W., Koster, W. & Dassa, E. Bacterial binding protein-dependent permeases: characterization of distinctive signatures for functionally related integral cytoplasmic membrane proteins. Mol. Microbiol. 12, 993–1004 (1994)
Busch, W. & Saier, M. H. Jr. The transporter classification (TC) system, 2002. Crit. Rev. Biochem. Mol. Biol. 37, 287–337 (2002)
Mourez, M., Hofnung, M. & Dassa, E. Subunit interactions in ABC transporters: a conserved sequence in hydrophobic membrane proteins of periplasmic permeases defines an important site of interaction with the ATPase subunits. EMBO J. 16, 3066–3077 (1997)
Ehrle, R., Pick, C., Ulrich, R., Hofmann, E. & Ehrmann, M. Characterization of transmembrane domains 6, 7, and 8 of MalF by mutational analysis. J. Bacteriol. 178, 2255–2262 (1996)
Steinke, A., Grau, S., Davidson, A., Hofmann, E. & Ehrmann, M. Characterization of transmembrane segments 3, 4, and 5 of MalF by mutational analysis. J. Bacteriol. 183, 375–381 (2001)
Quiocho, F. A. Carbohydrate-binding proteins: tertiary structures and protein–sugar interactions. Annu. Rev. Biochem. 55, 287–315 (1986)
Shuman, H. A. Active transport of maltose in Escherichia coli K-12: role of the periplasmic maltose binding protein and evidence for a substrate recognition site in the cytoplasmic membrane. J. Biol. Chem. 257, 5455–5461 (1982)
Schirmer, T., Keller, T. A., Wang, Y. F. & Rosenbusch, J. P. Structural basis for sugar translocation through maltoporin channels at 3.1 Å resolution. Science 267, 512–514 (1995)
Quiocho, F. A. & Ledvina, P. S. Atomic structure and specificity of bacterial periplasmic receptors for active transport and chemotaxis: variation of common themes. Mol. Microbiol. 20, 17–25 (1996)
Miller, D. M., Olson, J. S., Pflugrath, J. W. & Quiocho, F. A. Rates of ligand binding to periplasmic proteins involved in bacterial transport and chemotaxis. J. Biol. Chem. 258, 13665–13672 (1983)
Nelson, B. D. & Traxler, B. Exploring the role of integral membrane proteins in ATP-binding cassette transporters: analysis of a collection of MalG insertion mutants. J. Bacteriol. 180, 2507–2514 (1998)
Austermuhle, M. I., Hall, J. A., Klug, C. S. & Davidson, A. L. Maltose-binding protein is open in the catalytic transition state for ATP hydrolysis during maltose transport. J. Biol. Chem. 279, 28243–28250 (2004)
Fetsch, E. E. & Davidson, A. L. Vanadate-catalyzed photocleavage of the signature motif of an ATP-binding cassette (ABC) transporter. Proc. Natl Acad. Sci. USA 99, 9685–9690 (2002)
Sharma, S. & Davidson, A. L. Vanadate-induced trapping of nucleotide by the purified maltose transport complex requires ATP hydrolysis. J. Bacteriol. 182, 6570–6576 (2000)
Urbatsch, I. L., Sankaran, B., Bhagat, S. & Senior, A. E. Both P-glycoprotein nucleotide-binding sites are catalytically active. J. Biol. Chem. 270, 26956–26962 (1995)
Urbatsch, I. L., Sankaran, B., Weber, J. & Senior, A. E. P-glycoprotein is stably inhibited by vanadate-induced trapping of nucleotide at a single catalytic site. J. Biol. Chem. 270, 19383–19390 (1995)
Jardetzky, O. Simple allosteric model for membrane pumps. Nature 211, 969–970 (1966)
Neuhard, J. & Nygaard, P. in Escherichia coli and Salmonella typhimurium: Cellular and Molecular Biology (eds Ingraham, J. L. & Neidhardt, F. C.) 445–473 (ASM Press, Washington DC, 1987)
Davidson, A. L., Laghaeian, S. S. & Mannering, D. E. The maltose transport system of Escherichia coli displays positive cooperativity in ATP hydrolysis. J. Biol. Chem. 271, 4858–4863 (1996)
Sun, H. & Nathans, J. Mechanistic studies of ABCR, the ABC transporter in photoreceptor outer segments responsible for autosomal recessive Stargardt disease. J. Bioenerg. Biomembr. 33, 523–530 (2001)
Davidson, A. L. & Nikaido, H. Overproduction, solubilization, and reconstitution of the maltose transport system from Escherichia coli . J. Biol. Chem. 265, 4254–4260 (1990)
Chen, J., Lu, G., Lin, J., Davidson, A. L. & Quiocho, F. A. A tweezers-like motion of the ATP-binding cassette dimer in an ABC transport cycle. Mol. Cell 12, 651–661 (2003)
Dean, D. A., Fikes, J. D., Gehring, K., Bassford, P. J. & Nikaido, H. Active transport of maltose in membrane vesicles obtained from Escherichia coli cells producing tethered maltose-binding protein. J. Bacteriol. 171, 503–510 (1989)
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Macro Crystallogr. A 276, 307–326 (1997)
Project, C. C. The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994)
Jones, T. A., Zou, J. Y., Cowan, S. W. & Kjeldgaard, M. Improved methods for building protein models in electron-density maps and the location of errors in these models. Acta Crystallogr. A 47, 110–119 (1991)
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004)
Brunger, A. T. et al. Crystallography & NMR system: A new software suite for macromolecular structure determination. Acta Crystallogr. D 54, 905–921 (1998)
Painter, J. & Merritt, E. A. TLSMD web server for the generation of multi-group TLS models. J. Appl. Cryst. 39, 109–111 (2006)
Howlin, B., Butler, S. A., Moss, D. S., Harris, G. W. & Driessen, H. P. C. TLSANL—TLS parameter-analysis program for segmented anisotropic refinement of macromolecular structures. J. Appl. Cryst. 26, 622–624 (1993)
Ehrmann, M. & Beckwith, J. Proper insertion of a complex membrane protein in the absence of its amino-terminal export signal. J. Biol. Chem. 266, 16530–16533 (1991)
DeLano, W.L. The PyMOL Molecular Graphics System (2002) on the World Wide Web 〈http://www.pymol.org〉
Kleywegt, G. J. & Jones, T. A. Detection, delineation, measurement and display of cavities in macromolecular structures. Acta Crystallogr. D 50, 178–185 (1994)
Acknowledgements
We thank the beamline staff at the Advanced Photon Source beamline 23-ID for assistance with data collection, and R. MacKinnon, H. Shuman, D. Yernool and C. Orelle for discussions. This work was supported by NIH grants (J.C., A.L.D. and F.A.Q.), the Welch Foundation (F.A.Q.), the Pew Scholar Program (J.C.) and a postdoctoral fellowship from American Heart Association (M.L.O).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
The file contains Supplementary Figures 1-5 and Legends, Supplementary Tables 1-2 and additional references. (PDF 762 kb)
Rights and permissions
About this article
Cite this article
Oldham, M., Khare, D., Quiocho, F. et al. Crystal structure of a catalytic intermediate of the maltose transporter. Nature 450, 515–521 (2007). https://doi.org/10.1038/nature06264
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature06264
This article is cited by
-
Enhancing coevolutionary signals in protein–protein interaction prediction through clade-wise alignment integration
Scientific Reports (2024)
-
X-ray crystallography reveals molecular recognition mechanism for sugar binding in a melibiose transporter MelB
Communications Biology (2021)
-
Cryo-EM reveals unique structural features of the FhuCDB Escherichia coli ferrichrome importer
Communications Biology (2021)
-
Bacterial chemotaxis towards polysaccharide pectin by pectin-binding protein
Scientific Reports (2020)
-
Multitask ATPases (NBDs) of bacterial ABC importers type I and their interspecies exchangeability
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.