Abstract
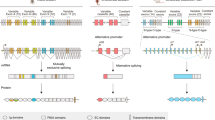
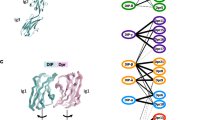
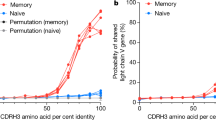
The Dscam gene gives rise to thousands of diverse cell surface receptors1 thought to provide homophilic and heterophilic recognition specificity for neuronal wiring2,3,4 and immune responses5. Mutually exclusive splicing allows for the generation of sequence variability in three immunoglobulin ecto-domains, D2, D3 and D7. We report X-ray structures of the amino-terminal four immunoglobulin domains (D1–D4) of two distinct Dscam isoforms. The structures reveal a horseshoe configuration, with variable residues of D2 and D3 constituting two independent surface epitopes on either side of the receptor. Both isoforms engage in homo-dimerization coupling variable domain D2 with D2, and D3 with D3. These interactions involve symmetric, antiparallel pairing of identical peptide segments from epitope I that are unique to each isoform. Structure-guided mutagenesis and swapping of peptide segments confirm that epitope I, but not epitope II, confers homophilic binding specificity of full-length Dscam receptors. Phylogenetic analysis shows strong selection of matching peptide sequences only for epitope I. We propose that peptide complementarity of variable residues in epitope I of Dscam is essential for homophilic binding specificity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Schmucker, D. et al. Drosophila Dscam is an axon guidance receptor exhibiting extraordinary molecular diversity. Cell 101, 671–684 (2000)
Wojtowicz, W. M., Flanagan, J. J., Millard, S. S., Zipursky, S. L. & Clemens, J. C. Alternative splicing of Drosophila Dscam generates axon guidance receptors that exhibit isoform-specific homophilic binding. Cell 118, 619–633 (2004)
Schmucker, D. & Flanagan, J. G. Generation of recognition diversity in the nervous system. Neuron 44, 219–222 (2004)
Neves, G., Zucker, J., Daly, M. & Chess, A. Stochastic yet biased expression of multiple Dscam splice variants by individual cells. Nature Genet. 36, 240–246 (2004)
Watson, F. L. et al. Extensive diversity of Ig-superfamily proteins in the immune system of insects. Science 309, 1874–1878 (2005)
Wang, J., Zugates, C. T., Liang, I. H., Lee, C. H. & Lee, T. Drosophila Dscam is required for divergent segregation of sister branches and suppresses ectopic bifurcation of axons. Neuron 33, 559–571 (2002)
Hummel, T. et al. Axonal targeting of olfactory receptor neurons in Drosophila is controlled by Dscam. Neuron 37, 221–231 (2003)
Zhan, X. L. et al. Analysis of Dscam diversity in regulating axon guidance in Drosophila mushroom bodies. Neuron 43, 673–686 (2004)
Zhu, H. et al. Dendritic patterning by Dscam and synaptic partner matching in the Drosophila antennal lobe. Nature Neurosci. 9, 349–355 (2006)
Chen, B. E. et al. The molecular diversity of Dscam is functionally required for neuronal wiring specificity in Drosophila. Cell 125, 607–620 (2006)
Hughes, M. et al. Homophilic Dscam interactions control complex dendrite morphogenesis. Neuron 54, 417–427 (2007)
Matthews, B. et al. Dendrite self-avoidance is controlled by Dscam. Cell 129, 593–604 (2007)
Soba, P. et al. Drosophila sensory neurons require Dscam for dendritic self-avoidance and proper dendritic field organization. Neuron 54, 403–416 (2007)
Dong, Y., Taylor, H. E. & Dimopoulos, G. AgDscam, a hypervariable immunoglobulin domain-containing receptor of the Anopheles gambiae innate immune system. PLoS Biol. 4, e229 (2006)
Ohi, M., Li, Y., Cheng, Y. & Walz, T. Negative staining and image classification—powerful tools in modern electron microscopy. Biol. Proced. Online 6, 23–34 (2004)
Lo Conte, L., Chothia, C. & Janin, J. The atomic structure of protein–protein recognition sites. J. Mol. Biol. 285, 2177–2198 (1999)
Su, X. D. et al. Crystal structure of hemolin: a horseshoe shape with implications for homophilic adhesion. Science 281, 991–995 (1998)
Freigang, J. et al. The crystal structure of the ligand binding module of axonin-1/TAG-1 suggests a zipper mechanism for neural cell adhesion. Cell 101, 425–433 (2000)
Schurmann, G., Haspel, J., Grumet, M. & Erickson, H. P. Cell adhesion molecule L1 in folded (horseshoe) and extended conformations. Mol. Biol. Cell 12, 1765–1773 (2001)
Harpaz, Y. & Chothia, C. Many of the immunoglobulin superfamily domains in cell adhesion molecules and surface receptors belong to a new structural set which is close to that containing variable domains. J. Mol. Biol. 238, 528–539 (1994)
Wang, J. Protein recognition by cell surface receptors: physiological receptors versus virus interactions. Trends Biochem. Sci. 27, 122–126 (2002)
Schneider, T. D. & Stephens, R. M. Sequence logos: a new way to display consensus sequences. Nucleic Acids Res. 18, 6097–6100 (1990)
Otwinowski, Z. & Minor, W. in Macromolecular Crystallography (eds Carter, C. W. Jr & Sweet, R. M.) 307–326 (Academic Press, New York, 1997)
Schneider, T. R. & Sheldrick, G. M. Substructure solution with SHELXD. Acta Crystallogr. D Biol. Crystallogr. 58, 1772–1779 (2002)
La Fortelle, E. & Bricogne, G. in Methods in Enzymology, Macromolecular Crystallography (eds Carter, C. W. Jr & Sweet, R. M.) 472–494 (Academic Press, New York, 1997)
CCP4. The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994)
McRee, D. E. XtalView/Xfit—A versatile program for manipulating atomic coordinates and electron density. J. Struct. Biol. 125, 156–165 (1999)
Cowtan, K., Zhang, K. & Main, P. in International Tables for Crystallography (eds Rossmann, M. & Arnold, E.) 25.2.5 (Kluwer Academic Publishers, Dordrecht, 2001)
Perrakis, A., Morris, R. & Lamzin, V. S. Automated protein model building combined with iterative structure refinement. Nature Struct. Biol. 6, 458–463 (1999)
Murshudov, G. N., Vagin, A. A. & Dodson, E. J. Refinement of macromolecular structures by the maximum-likelihood method. Acta Crystallogr. D Biol. Crystallogr. 53, 240–255 (1997)
Adams, P. D. et al. Recent developments in the PHENIX software for automated crystallographic structure determination. J. Synchrotron Radiat. 11, 53–55 (2004)
Ohi, M., Li, Y., Cheng, Y. & Walz, T. Negative staining and image classification—powerful tools in modern electron microscopy. Biol. Proced. Online 6, 23–34 (2004)
Frank, J. et al. SPIDER and WEB: processing and visualization of images in 3D electron microscopy and related fields. J. Struct. Biol. 116, 190–199 (1996)
Thompson, J. D., Higgins, D. G. & Gibson, T. J. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 22, 4673–4680 (1994)
Acknowledgements
We thank K. Tan for discussion, and E. Reinherz, M. Eck and B. Chen for comments on the manuscript. We also thank R. Zhang and A. Joachimiak at the 19ID beamline of the Advanced Photon Source at the Argonne National Laboratory, and A. Soares at the X25 beamline at the Brookhaven National Synchrotron Light Source for help in X-ray data collection. The molecular electron microscopy facility at Harvard Medical School was established by a generous donation from the Giovanni Armenise Harvard Center for Structural Biology and is maintained by an NIH grant to T.W. We are grateful to S. L. Zipursky and W. Wojtowicz for discussions and the sharing of unpublished results. This work was supported by NIH grants to J.-h.W., D.S. and T.W., and a Pew Scholar Award and John Merck Fund Award to D.S. G.S. is a Damon Runyon fellow, supported by the Damon Runyon Cancer Research Foundation.
Atomic coordinates and structure factors have been deposited in the Protein Data Bank: 2V5M (Dscam D1–D41.34, space group P4222), 2V5S (Dscam D1–D41.34, space group C2221) and 2V5R (Dscam D1–D49.9, space group C2).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures S1-S12 with Legends and Supplementary Table S1. (PDF 8939 kb)
Rights and permissions
About this article
Cite this article
Meijers, R., Puettmann-Holgado, R., Skiniotis, G. et al. Structural basis of Dscam isoform specificity. Nature 449, 487–491 (2007). https://doi.org/10.1038/nature06147
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature06147
This article is cited by
-
The gene structure and hypervariability of the complete Penaeus monodon Dscam gene
Scientific Reports (2019)
-
Revisiting Dscam diversity: lessons from clustered protocadherins
Cellular and Molecular Life Sciences (2019)
-
A chelicerate-specific burst of nonclassical Dscam diversity
BMC Genomics (2018)
-
Improvement of Dscam homophilic binding affinity throughout Drosophilaevolution
BMC Evolutionary Biology (2014)
-
Kassiopeia: a database and web application for the analysis of mutually exclusive exomes of eukaryotes
BMC Genomics (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.